
Int J Curr Pharm Res, Vol 18, Issue 1, 14-22Original Article
TO EVALUATE THE NEUROPROTECTIVE ACTIVITY OF STRAWBERRY AND BLUEBERRY EXTRACT IN MPTP-INDUCED PARKINSONISM IN MICE
LALIT KUMAR*, MEENAKSHI MALHOTRA, AJEET PAL SINGH, AMAR PAL SINGH
St. Soldier Institute of Pharmacy, Lidhran Campus Behind NIT, Jalandhar-Amritsar By-Pass, Jalandhar, Punjab 144011, India
*Corresponding author: Lalit Kumar; *Email: railucky193@gmail.com
Received: 06 Oct 2025, Revised and Accepted: 26 Nov 2025
ABSTRACT
Objective: This study aimed to evaluate the neuroprotective effects of ethanolic extracts of strawberry and blueberry in an MPTP-induced mouse model of Parkinsonism.
Methods: Swissmice (30–35 g) were divided into five groups: control, MPTP-treated, Syndopa (Levodopa+Carbidopa)-treated, low-dose extract-treated, and high-dose extract-treated groups. Parkinsonism was induced using MPTP (20 mg/kg) for seven consecutive days. Mice were then administered either Syndopa (10 mg/kg) or strawberry and blueberry extracts at low (100 mg/kg) or high (200 mg/kg) doses for Seven days. Behavioural assessments, including the Rotarod test, Grip test/Hang test, Open-field test, Actophotometer test, Forced Swim test, Elevated plus maze test, and Catalepsy/Wooden bar test, were conducted to evaluate motor function, anxiety-like behaviour, and catalepsy.
Results: MPTP administration led to significant motor impairments, increased catalepsy, and reduced locomotor activity. Treatment with strawberry and blueberry extracts significantly improved motor coordination, reduced cataleptic symptoms, and enhanced locomotor activity in a dose-dependent manner. The observed neuroprotective effects were comparable to those of Syndopa (Levodopa+Carbidopa)-treated, suggesting that bioactive compounds in these fruits may mitigate neurodegeneration.
Conclusion: The results support the potential of strawberry and blueberry extracts as natural therapeutic agents in Parkinson’s disease. Their antioxidant and neuroprotective properties warrant further investigation into the underlying mechanisms and potential clinical applications in PD management.
Keywords: Parkinson’s disease, MPTP, Neuroprotection, Strawberry extract, Blueberry extract, Behavioural assessment, Oxidative stress, Syndopa (Levodopa+Carbidopa)
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i1.7089 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Brain metabolism, accounting for 20% of total oxygen consumption, responds dynamically to energy needs, triggering compensatory mechanisms regionally and within neurons [1]. Functional brain imaging techniques, alongside PET, revolutionize the understanding of movement disorders. Specialized network analysis explores brain-behaviour changes in PD, revealing widespread effects beyond dopamine loss [2].
Deep brain stimulation (DBS) is a neurosurgical technique involving continuous electrical stimulation delivered to neural brain structures via implanted electrodes. It is programmable and adjustable, offering reversible effects and precise targeting compared to ablative methods. High-frequency stimulation (HFS) has emerged as an alternative to lesioning, particularly in movement disorders like Parkinson's disease, with expanding applications in various pathologies. Its mechanisms and effects continue to be studied extensively [3].
Parkinson's disease causes motor symptoms due to dopaminergic neuron loss. It overlaps with other neurodegenerative disorders like Alzheimer's. Challenges include early diagnosis and developing neuron-protective treatments [4]. Parkinson's disease exhibits cognitive deficits beyond motor impairments, attributed to complex interactions between neurotransmitter abnormalities and Lewy’s body pathology, delineated in the 'Dual Syndrome' hypothesis [5]. Parkinson's disease (PD) affects 1-3% of people over 60 globally. It has familial and sporadic forms, with familial cases accounting for 10-15%. Seven genes, including SNCA and LRRK2, are linked to familial PD. Early diagnosis is crucial due to 70% neuronal loss before symptoms appear [6].
Current pharmacological treatments for PD, such as Levodopa combined with Carbidopa, provide symptomatic relief but fail to halt or reverse the neurodegenerative process. Moreover, long-term use of these drugs is often associated with diminished efficacy and adverse effects. This has led to growing interest in alternative and adjunctive therapies, particularly those derived from natural sources, which may offer neuroprotection with fewer side effects.
Dietary antioxidants have emerged as promising candidates in neurodegenerative disease research due to their ability to scavenge reactive oxygen species and modulate inflammatory pathways. Berries, especially strawberries (Fragaria ananassa) and blueberries (Vaccinium corymbosum), are rich in polyphenols, flavonoids, anthocyanins, and other bioactive compounds known for their potent antioxidant and anti-inflammatory properties. Preclinical studies suggest that these phytochemicals can attenuate neuronal damage and improve cognitive and motor functions in various models of neurodegeneration.
In this context, the present study aims to investigate the neuroprotective potential of ethanolic extracts of strawberry and blueberry in an MPTP-induced mouse model of Parkinsonism. MPTP (1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine) is a well-established neurotoxin that selectively targets dopaminergic neurons and replicates key features of human PD in animal models. By evaluating behavioural parameters such as motor coordination, locomotor activity, anxiety-like behaviour, and catalepsy, this study seeks to explore whether these fruit extracts can mitigate PD-like symptoms and provide a basis for their possible therapeutic use in the management of Parkinson’s disease.
Neurodegenerative disease
Neurodegenerative diseases mean that your brain cells are slowly dying. This can lead to memory loss, trouble moving, thinking problems, changes in emotions, and behavior issues [7]. Neurodegenerative diseases are a big problem for health. They happen more as people get older, and because more people are living longer now, these diseases are becoming more common [8].
Herbal therapy
Berries like blueberries and strawberries are highly sought in the nutraceutical and functional food market. They are popular both in fresh and frozen forms, as well as in various processed and derived food products [9]. Blueberries contain a high percentage of phenolics, constituting 50–80% of the total polyphenol content, with concentrations reaching up to 3000 mg/kg fresh weight [10]. Berries, rich in flavonoids and antioxidants, exhibit potential preventive and positive effects in conditions like cancer, cardiovascular disease (CVD), and age-related brain diseases. The neuroprotective properties of berry components, particularly in strawberries and blueberries, are associated with improved cognition, motor behavior, signal transduction, and memory. Research suggests that polyphenolic compounds in berries may mitigate oxidative stress and inflammation, potentially lowering the risk of age-related neurodegenerative diseases [11].
MATERIALS AND METHODS
Chemical requirements
MPTP (1-methyl, 4-phenyl-1, 2, 3, 6-tetrahydropyridine) was purchased from Rajesh Enterprises, Ambala Cantt. All other chemicals and reagents used were analytical grade and procured from approved chemical suppliers.
Plant material
The Ethanolic extracts of blueberry and strawberry fruits were purchased from Shreedha Phyto Extracts, Jaipur.
Standard drug
Syndopa (Levodopa+Carbidopa) is used as a standard drug.
Test component
A combination of fruits of strawberry and blueberry extract was used as the test component.
Extraction of herbal preparation
Preparation of strawberry extract (SBE)
Strawberry fruits weighing 100 g were directly macerated in 80 ml of Ethanol and allowed to stand for 12 h without agitation. Subsequently, the macerated mixture was subjected to homogenization to obtain a clear juice. This juice underwent filtration using Whatman filter paper and was then subjected to incubation at 45 °C for 24 h to facilitate the formation of strawberry extract powder (SBE) [12].
Preparation of blueberry extract (BBE)
Blueberries (Vaccinium virgatum) were frozen at-20 °C until extraction. To extract the compounds, 30 g of fresh blueberries were sonicated for 30 min at 25 °C in a solution of 70% ethanol and 30% water (pH 1.0). The resulting extracts were filtered, and the ethanol was removed under reduced pressure before being freeze-dried. This process was repeated three times and conducted away from light. Our research group previously analyzed this study's blueberry extract and identified fifteen anthocyanins[13].
Preparation and administration of MPTP solution
MPTP was procured from Rajesh Enterprises, Ambala cantt, and stored at 37 °C in accordance with the manufacturer’s guidelines to prevent degradation. A fresh MPTP solution was prepared at a concentration of 20 mg/kg by dissolving it in 0.9% sodium chloride solution. The solution was administered intraperitoneally (i. p.) at a dosage of 20 mg/kg body weight once daily for 7 consecutive days. Notably, the prepared MPTP solution remains stable for only 24 h when stored at 4 °C.
Preparation and administration of levodopa and carbidopa
Levodopa (100 mg/kg) and Carbidopa (10 mg/kg) were accurately weighed and dissolved in distilled water to prepare the dosing solution. The solution was freshly prepared each day to maintain chemical stability and therapeutic efficacy. The formulation was administered orally (p. o.) to the standard treatment group.
Procurement of animals
SwissMice, weighing between-30 to 35g procured from Lovely Professional University, Phagwara, and kept in the Animal House Facility, St. Soldier Institute of Pharmacy, Jalandhar, Punjab, India. The animals were kept in groups of five under standard lab conditions, with the temperature set at 23±1 °C and humidity at 55±5%. They had unlimited access to pellet food (from Sanjay Biological Museum, Amritsar, India) and water. Before the study, they were given 7 days to get used to the lab environment [14].
The experimental protocol was reviewed and approved by the Institutional Animal Ethics Committee (IAEC) under letter number IAEC/SSIP/2024/PR-042. All experimental procedures were carried out in strict accordance with the guidelines established by the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Environment and Forests, Government of India (Registration No. 2011/PO/Re/S/18/CPCSEA; Date of Registration: 01/05/2018), for the ethical care and use of laboratory animals.
Experimental design
Experimental work
The study involves five groups of subjects to investigate the effects of different treatments on Parkinson's disease induced by MPTP. Group I served as the control group and received 0.9% w/v NaCl (10 ml/kg) orally once daily for seven days. Group II, the negative control group, was administered MPTP intraperitoneally (i. p.) at a dose of 20 mg/kg once daily for seven consecutive days to induce PD-like symptoms. Group III, the standard treatment group, received SYNDOPA (Levodopa+Carbidopa) at a dose of 10 mg/kg orally once daily for seven days, followed by MPTP (20 mg/kg, i. p.) once daily for seven days to assess the effects of a known PD treatment. Group IV, the test preparation (low dose) group, received an ethanolic extract of strawberry and blueberry fruits at a low dose (100 mg/kg) orally once daily for seven days, followed by MPTP administration (20 mg/kg, i. p.) once daily for seven days. Similarly, Group V, the test preparation (high dose) group, received a higher dose (200 mg/kg) of the same fruit extract orally for seven days, followed by MPTP administration (20 mg/kg, i. p.) once daily for seven days. Seven days after the last MPTP injection, behavioral tests such as the Rotarod test, Hanging/Grip Test, Open-field test, Actophotometer test, Forced Swim test, Elevated plus Maze test, and Catalepsy/wooden bar test were conducted.
Table 1: Grouping of animals
| Groups | Treatment | Dose (mg/kg) | Animal required | Days |
| I | Vehicle only | 0.9% w/v NaCl | 6 | 7 |
| II | MPTP | 20 mg/kg i. p. | 6 | 7 |
| III | Syndopa (Levodopa (100 mg) and Carbidopa (10 mg)/kg)+MPTP | 10 mg/kg p. o.+20 mg/kg i. p. | 6 | 7 |
| IV | Ethanolic extract of Fruit of Strawberry and blueberry, low dose (100 mg/kg b. wt)+MPTP | 100 mg/kg p. o.+20 mg/kg i. p. | 6 | 7 |
| V | Ethanolic extract of Fruit of Strawberry and Blueberry High dose (200 mg/kg b. wt)+MPTP | 200 mg/kg p. o.+20 mg/kg i. p. | 6 | 7 |
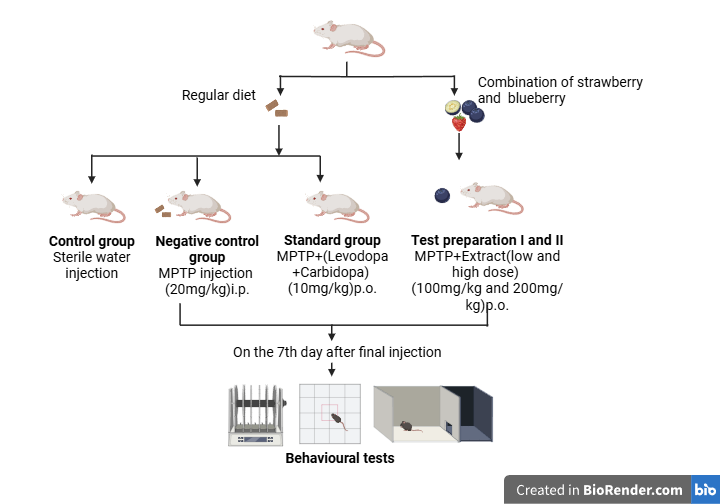
Fig. 1: Treatment schedule and experimental design
Total No. of animals required
No. of the animal in each group (n) = 06
No. of groups (N) = 5
Total no. of animals required= 30
Various test and procedures to be performed in in vivo studies in detail
Body weight Analysis
Feed Intake Analysis
Water Intake Analysis
Behaviour Test:
Rotarod Test
Hanging Test
Forced Swim Test
Open field Test
Actophotometer Test
Elevated Plus Maze Test
Catalepsy bar Test
a. Body weight analysis: Body weight analysis was conducted for each group of animals at the start and at the end of the study, and changes in body weight were recorded for each group.
b. Feed intake analysis: Feed intake was measured for each group of animals at the beginning and end of the study, and any changes in feed intake were recorded accordingly.
c. Water intake analysis: Water intake was monitored for each group of animals at both the start and the end of the study, and any variations in consumption were documented accordingly.
Behaviour test
Rotarod test
In the rotarod experiments, animals were trained for three days before the actual test. During the test, they walked on a rotating rod set at a constant speed of 5 revolutions per minute (rpm). The researchers measured how long the animals could stay on the rod without falling, up to a maximum of 5 min. Each animal went through the test four times, and the average time they stayed on the rod was calculated [15].
Hanging/Grip test
The Bar test has been used to evaluate motor coordination by testing the ability of mice to remain on the rod Deacon, (2013). The bars are made of brass, 38 cm long, held 49 cm above the bench surface by a wooden support column at each end. The columns are secured to a heavy wooden base. Three bar/wire diameters are available: 0.5, 2, and 4 mm. larger diameter bars have been included to refine the test, as mice cannot grip these so well. The experiment was performed as described by Deacon (2013), with some modifications to evaluate fore and hind limb motor coordination. The mouse was lifted by the tail and placed on the bench in front of the apparatus. The mouse was slid backward quickly about 20 cm (aligning it perpendicular to the bar), rapidly raised, and allowed to grasp the horizontal bar at the central point with its forepaws only. The tail was released, and the stopwatch was started simultaneously. Some mice may fail to grip better if the tail is released suddenly. If the mouse failed to grasp the bar properly the first time, the experiment was performed again after a brief rest and repeated a maximum of 3 attempts. The best score was taken. The scoring system for 0.5, 2 mm, and 4 mm bars was the same throughout the protocol, and the final score was recorded [16].
Forced swim test
Mice were put one by one into a tall glass filled with room temperature water (23–25 °C). We measured how long they stayed still in the water as a sign of feeling down or hopeless. The stillness was when they were just floating without trying to escape or move around. Each mouse went through a 15 min training session in the water, and afterward, they were dried using a heating pad [17].
Open field test
Each mouse was placed by itself in a clear box (30 cm × 30 cm × 15 cm) with the floor divided into 6 × 6 cm squares for exploration. We counted how many times they crossed these squares (defined as having at least three paws in a square) during 5 min. Experimenters, who were unaware of the treatment each mouse received, sat quietly about 0.5 meters away, and their positions and tasks remained constant throughout the study. After testing each mouse, we cleaned the box with a 70% ethanol solution and let it dry to prevent the influence of smells [18].
Actophotometer test
We measured how much the animals moved using a device called an actophotometer (IMCORP, India). This device includes a cage that is 30 cm long and 30 cm deep with a wire mesh at the bottom. Six lights were set up to shine on corresponding photoelectric cells. When an animal passes through the light beam, the photoelectric cell is activated by blocking the light. We counted these blockages for 5 min as a way to gauge how much the animal moved, which we call locomotor activity. Before starting the activity, the animal had 2 min to get used to the surroundings. Then, we used the actophotometer to record the animal's movement for another 5 min. The recorded activity is expressed as the total number of times the light beam was blocked in 5 min, and this count is related to how far the animal traveled [19].
Elevated plus maze test
The elevated plus maze test was used to evaluate anxiety-like behavior in mice. Before the test, the mice were allowed to adapt to the experimental room for 30 min. They were then placed in a cross-shaped maze with open arms (5 cm × 50 cm) and closed arms (5 cm × 50 cm × 10 cm), with a central area of 5 cm × 5 cm. A video camera recorded their movement for 1 min as they explored freely. During the test, their movements under dim light were tracked for 5 min using SuperMaze software. The maze was cleaned with 75% ethanol between tests to remove animal odors[20].
Catalepsy bar test
The test followed Hoffman's method to measure catalepsy using a standard bar test. The animal's front paws were placed on a 3 cm high wooden bar (0.9 cm in diameter), and the time it stayed in this position was recorded. Catalepsy ended when the animal either removed its paws from the bar or moved its head to explore. The typical catalepsy test consists of placing an animal into an unusual posture and recording the time taken to correct this posture. This time is regarded as an index of the intensity of catalepsy. Catalepsy is a robust behavior, and the lack of standardization does not usually hinder its actual detection. However, the intensity of the cataleptic effect is influenced by minor methodological differences, and thus, interpretation and comparison of results across laboratories are difficult. The behavioral catalepsy test can use several different apparatuses, including wire grids, parallel bars, platforms, or pegs, to situate the animals in unusual positions. The most common, however, is the "bar test," despite its wide use in psychopharmacological research; even the parameters of this test are not standardized. The present article reviews the wide variety of parameters chosen by investigators that measure catalepsy. The methodological issues of repeated testing, scaling of scores, apparatus, animal weight, maximal test duration, behavioral criteria, and other influences are discussed. In addition, a brief review of the neuropharmacological basis of catalepsy is also included. Finally, it is argued that researchers adopt a universal, standardized bar test. New data on a novel automated bar test in the Digi scan activity monitoring system is presented [21].
Statistical analysis
All the results were expressed as Mean±SEM. The data of all the groups were analyzed by one-way ANOVA followed by Tukey’s test using software GraphPad Prism In Stat (Graph Pad Software Inc., USA). A value of p<0.05 was considered to be significant.
RESULTS AND DISCUSSION
Effect of ethanolic extract of fruit of strawberry and blueberry on body weight (g) of mice
In both test groups, mice of 6 no. in each group Treated with Ethanolic extract of Fruit of Strawberry and Blueberry (100 and 200 mg/kg/p. o.), respectively showed significant (p<0.05) increase in body weight as compared to the control group. Treatment with SYNDOPA (10 mg/kg p. o.) the body weight significantly increased as compared to the normal group.
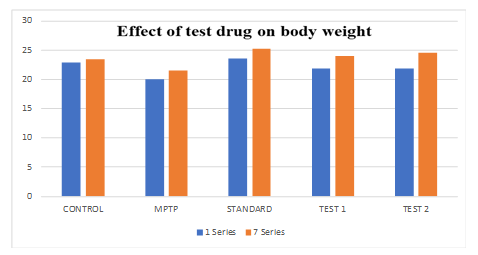
Graph 1: Showing the effect of ethanolic extract of fruit of strawberry and blueberry on body weight (g) of mice
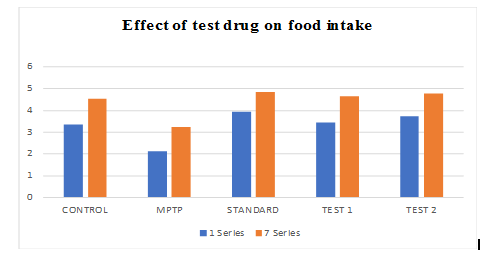
Graph 2: Effect of ethanolic extract of fruit of strawberry and blueberry on feed intake (g) of mice
Effect of ethanolic extract of fruit of strawberry and blueberry on feed intake (g) of mice
The mice of Ethanolic extract of Fruit of Strawberry and Blueberry (100and200 mg/kg/p. o.) treated group showed a significant (p<0.05) increase in feed intake as compared to the control group. In treatment with SYNDOPA (10 mg/kg p. o.), the feed intake significantly (p<0.05) increased as compared to the control group.
Effect of ethanolic extract of fruit of strawberry and blueberry on water intake (ml) of mice
The mice of Ethanolic extract of Fruit of Strawberry and Blueberry (100 and 200 mg/kg/p. o) treated group showed a significant (p<0.05) increase in body water intake as compared to the control group. In treatment with SYNDOPA (10 mg/kg p. o.), the water intake significantly (p<0.05) increased as compared to the test group.
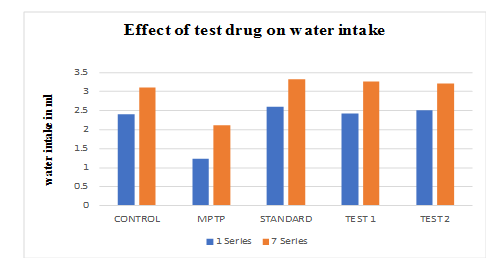
Graph 3: Effect of ethanolic extract of fruit of strawberry and blueberry on water intake (ml) of mice
Behaviour test
Rotarod test
In the rotarod test, the time spent on the rotating rod was measured in Groups I–V to evaluate motor coordination, balance, physical condition, and motor planning. Group I exhibited the best performance, staying on the rod the longest, while Group II had the poorest performance, remaining on the rod for the shortest duration. Groups III, IV, and V performed better than Group II but did not match Group I. Among these, Group III stayed on the rod the longest, followed by Group V, which performed better than Group IV. Notably, Group V's performance was close to that of Group III.
Graph 4: Effect of ethanolic extract of fruit of strawberry and blueberry on rota rod test

Fig. 2: Hanging/grip test
Hanging/grip
The hanging/grip test was used to assess motor coordination by measuring how long mice could balance on a horizontal bar. Three different bar diameters—0.5 mm, 2 mm, and 4 mm—were tested, with larger diameters making it more difficult due to reduced grip efficiency. Group I maintained its balance the longest, except for Group V, while Group II had the shortest time, indicating the weakest motor coordination. Groups III, IV, and V performed better than Group II but did not match Group I, except for Group V, which showed the highest performance. Among these, Group III stayed on the bar longer than Group IV, while Group V outperformed both, demonstrating the best motor coordination overall.
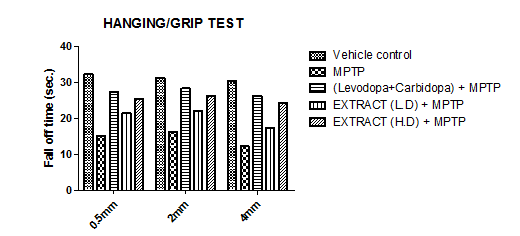
Graph 5: Effect of ethanolic extract of fruit of the strawberry and blueberry on grip test
Forced swim test
The forced swim test was used to measure the time mice took to stop moving and their duration of immobility, which indicates depression-like behavior. Group II exhibited the longest immobility time, suggesting the highest level of depression-like behavior, while Group I had the shortest immobility period, indicating the least depression-like tendencies. Groups III, IV, and V remained immobile for longer than Group I but for a shorter duration than Group II, suggesting moderate levels of depression-like behavior. Among them, Group III had the shortest immobility time, followed by Group V, while Group IV displayed the longest immobility period. Notably, Group V's immobility duration was similar to that of Group III.
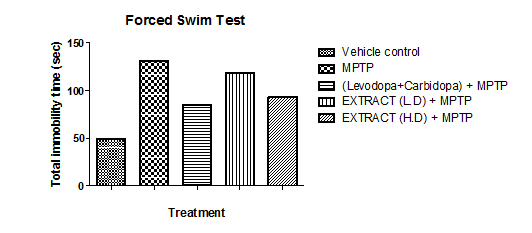
Graph 6: Effect of ethanolic extract of fruit of strawberry and blueberry on FST
Open field test
The open field test was conducted to assess locomotor behaviour by measuring the time spent in the center, grooming duration, and the number of line crossings. Group III spent the most time in the center, while Group II spent the least. Group I remained in the center longer than Group II, whereas Groups III, IV, and V spent more time in the center than both Groups I and II. Among them, Group III had the longest duration, followed by Group V, then Group IV.
For grooming behavior, Group I spent the most time grooming, while Group II spent the least. Groups III, IV, and V were groomed more than Group II but less than Group I, with Group IV grooming the longest, followed by Group V, and Group III spending the least time grooming.
Regarding the number of line crossings, Group I had the highest count, while Group II had the lowest. Groups III, IV, and V crossed more lines than Group II but fewer than Group I. Among them, Group IV had the highest count, followed by Group V, with Group III crossing the least.
Actophotometer test
The actophotometer test was used to measure locomotor activity in Groups I–V. Group I exhibited the highest locomotor activity, spending the most time in movement, while Group II showed the lowest activity with the least movement. Groups III, IV, and V were more active than Group II but less than Group I. Among them, Group III had the highest activity, followed by Group V, while Group IV displayed the lowest activity. Notably, Group V's locomotor activity was similar to that of Group III.
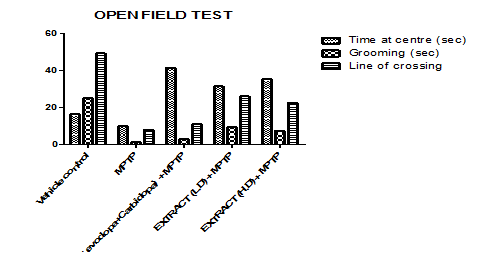
Graph 7: Effect of ethanolic extract of fruit of strawberry and blueberry on OFT
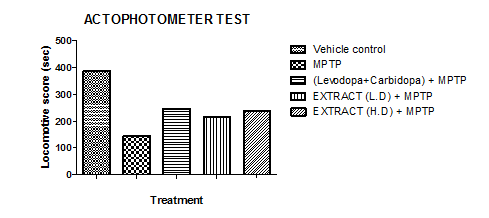
Graph 8: Effect of ethanolic extract of fruit of strawberry and blueberry on actophotometer test

Fig. 3: Mice in actophotometer
Elevated plus maze test
The elevated plus maze test was conducted to assess anxiety-like behaviour by measuring the number of entries and the time spent in open and closed arms. Group I spent the most time in the closed arm, indicating the highest anxiety-like behaviour, while Group II spent the least time in the closed arm, suggesting the lowest anxiety-like behaviour. Groups III, IV, and V spent less time in the closed arm than Group I but more than Group II. Among them, Group IV remained in the closed arm the longest, followed by Group V, while Group III spent the least time there.
Regarding the open arm, Group III spent the most time, reflecting the lowest anxiety-like behaviour, whereas Group II spent the least time. Group I spent more time in the open arm than Group II but less than Groups III, IV, and V. Group V remained in the open arm longer than Group IV but for a shorter duration than Group III. In terms of the number of entries, Group III had the highest count, while Group II had the lowest. Group I made more entries than Group II but fewer than the other groups. Group V had more entries than Group IV but fewer than Group III.

Fig. 4: Elevated plus maze
Catalepsy test
The catalepsy test was used to assess motor coordination by measuring the time animals maintained at least one forepaw on a horizontal bar. The duration was recorded once the animal removed its paw. Group II exhibited the longest time on the bar, indicating the highest catalepsy score and the poorest motor coordination. In contrast, Group IV had the shortest time, reflecting the best motor coordination. Group I remained on the bar for a shorter duration than Group II but longer than the other groups. Group III stayed on the bar longer than Groups IV and V, while Group V spent more time than Group IV but had results close to Group III. A lower catalepsy score indicated better motor coordination.
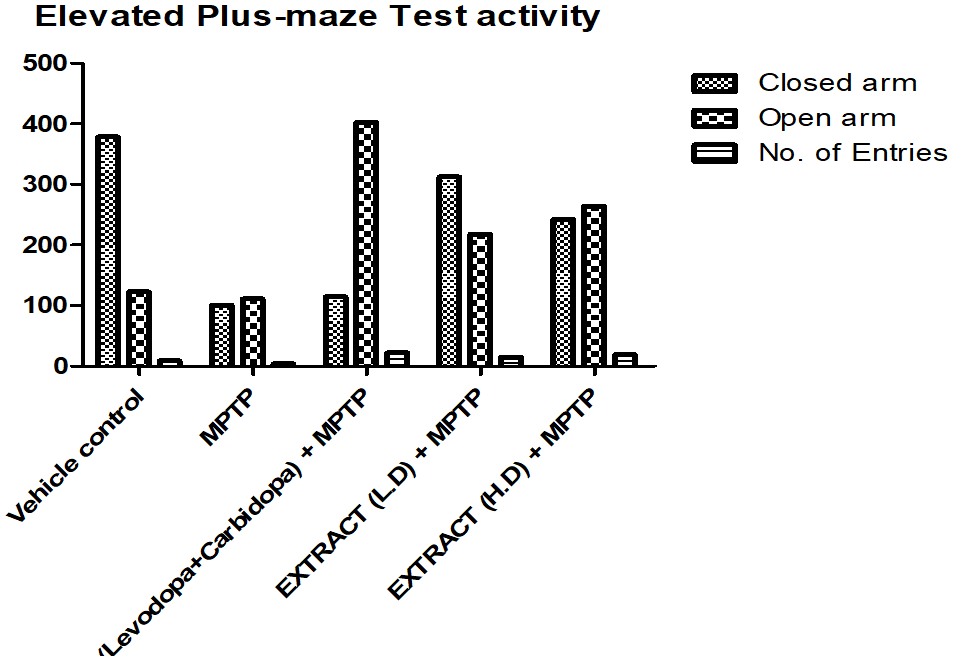
Graph 9: Effect of ethanolic extract of fruit of strawberry and blueberry on EPM test
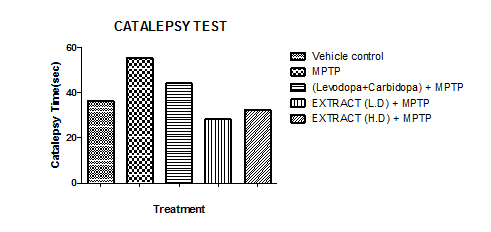
Graph 10: Effect of ethanolic extract of fruit of strawberry and blueberry on catalepsy test
MPTP administration induced Parkinsonian-like symptoms, as observed in the motor and behavioural deficits across various tests. The results demonstrated that treatment with L-DOPA and Carbidopa and ethanolic extracts of blueberries and strawberries significantly alleviated these impairments.
Motor function, assessed through the rotarod and grip tests, showed marked improvement in extract-treated groups. The actophotometer test further confirmed enhanced locomotor activity, supporting the protective role of natural compounds in neurodegeneration.
Behavioural assessments, including the forced swim, elevated plus-maze, and open field tests, revealed that extract treatment reduced depression and anxiety-like behaviour, suggesting possible neuroprotective effects via modulation of neurotransmitter levels.
The significant reduction in catalepsy time in extract-treated groups supports their potential role in mitigating Parkinsonism. The observed benefits may be attributed to the antioxidant and anti-inflammatory properties of blueberries and strawberries, which can counteract MPTP-induced oxidative stress and dopaminergic neuronal damage.
Overall, these findings suggest that ethanolic extracts of blueberries and strawberries exhibit neuroprotective effects comparable to L-DOPA and Carbidopa, making them promising candidates for Parkinson's disease management.
CONCLUSION
The present study evaluates the neuroprotective effects of strawberry and blueberry extracts in MPTP-induced Parkinsonism in mice. The results indicate that MPTP administration successfully induced Parkinson’s disease-like symptoms, as evidenced by significant impairments in motor coordination and behavioural parameters. Treatment with the ethanolic extracts of strawberry and blueberry demonstrated a protective effect by improving motor function, reducing catalepsy, and enhancing locomotor activity. These effects were comparable to the standard treatment, L-DOPA and Carbidopa, suggesting that the bioactive compounds in these fruits may contribute to neuroprotection. The findings support the potential of strawberry and blueberry extracts as natural therapeutic agents in managing Parkinson’s disease, warranting further investigation into their underlying mechanisms and clinical applications.
ACKNOWLEDGMENT
It’s our privilege to express a profound sense of gratitude and cordialthanks to our respected chairman Mr. Anil Chopra; Vice Chairperson, Ms. Sangeeta Chopra, St. Soldier Educational Society, Jalandhar for providing the necessary facilities to complete this research work.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
All authors made substantial contributions to the conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
CONFLICTS OF INTERESTS
Declared none
REFERENCES
Hyder F, Rothman DL, Bennett MR. Cortical energy demands of signaling and nonsignaling components in brain are conserved across mammalian species and activity levels. Proc Natl Acad Sci USA. 2013;110(9):3549-54. doi: 10.1073/pnas.1214912110, PMID 23319606.
Wichmann T, De Long MR. Functional and pathophysiological models of the basal ganglia. Curr Opin Neurobiol. 1996;6(6):751-8. doi: 10.1016/S0959-4388(96)80024-9, PMID 9000030.
Benabid AL. Deep brain stimulation for Parkinson’s disease. Curr Opin Neurobiol. 2003;13(6):696-706. doi: 10.1016/j.conb.2003.11.001.
Schapira AH. Science, medicine and the future: Parkinson’s disease. BMJ. 1999;318(7179):311-4. doi: 10.1136/bmj.318.7179.311, PMID 9924061.
Nombela C, Rowe JB, Winder Rhodes SE, Hampshire A, Owen AM, Breen DP. Genetic impact on cognition and brain function in newly diagnosed Parkinson’s disease: ICICLE-PD study. Brain. 2014;137(10):2743-58. doi: 10.1093/brain/awu201, PMID 25080285.
Ball N, Teo WP, Chandra S, Chapman J. Parkinson’s disease and the environment. Front Neurol. 2019 Mar;10:218. doi: 10.3389/fneur.2019.00218, PMID 30941085.
Mandemakers W, Morais VA, De Strooper B. A cell biological perspective on mitochondrial dysfunction in Parkinson disease and other neurodegenerative diseases. J Cell Sci. 2007;120(10):1707-16. doi: 10.1242/jcs.03443, PMID 17502481.
Heemels MT. Neurodegenerative diseases. Nature. 2016;539(7628):179. doi: 10.1038/539179a, PMID 27830810.
Miller K, Feucht W, Schmid M. Bioactive compounds of strawberry and blueberry and their potential health effects based on human intervention studies: a brief overview. Nutrients. 2019;11(7):1510. doi: 10.3390/nu11071510, PMID 31269727.
Shi M, Loftus H, McAinch AJ, Su XQ. Blueberry as a source of bioactive compounds for the treatment of obesity type 2 diabetes and chronic inflammation. J Funct Foods. 2017;30:16-29. doi: 10.1016/j.jff.2016.12.036.
Kim HG, Ju MS, Shim JS, Kim MC, Lee SH, Huh Y. Mulberry fruit protects dopaminergic neurons in toxin-induced Parkinson’s disease models. Br J Nutr. 2010;104(1):8-16. doi: 10.1017/S0007114510000218, PMID 20187987.
Nellore J, Amarnath K. Development and in vitro evaluation of strawberry fruit extract-loaded biodegradable nanoparticles in therapy for neurodegenerative disorders. In: International Conference on Nanoscience Engineering and Technology (ICONSET 2011). New York: IEEE; 2011. p. 396-400. doi: 10.1109/ICONSET.2011.6167988.
Spohr L, Luduvico KP, Soares MS, Bona NP, Oliveira PS, De Mello JE. Blueberry extract as a potential pharmacological tool for preventing depressive-like behavior and neurochemical dysfunctions in mice exposed to lipopolysaccharide. Nutr Neurosci. 2022;25(4):857-70. doi: 10.1080/1028415X.2020.1819104, PMID 32954970.
Kumar SV, Kumar SP, Rupesh D, Nitin K. Journal of chemical and pharmaceutical research preparations. J Chem Pharm Res. 2011;3(1):675-84.
Manna S, Bhattacharyya D, Mandal TK, Dey S. Neuropharmacological effects of deltamethrin in rats. J Vet Sci. 2006;7(2):133-6. doi: 10.4142/jvs.2006.7.2.133, PMID 16645337.
Deacon RM. Measuring motor coordination in mice. J Vis Exp. 2013;(75):e2609. doi: 10.3791/2609, PMID 23748408.
Liu L, Peritore C, Ginsberg J, Shih J, Arun S, Donmez G. Protective role of SIRT5 against motor deficit and dopaminergic degeneration in MPTP-induced mice model of Parkinson’s disease. Behav Brain Res. 2015;281:215-21. doi: 10.1016/j.bbr.2014.12.035, PMID 25541039.
Wang XL, Xing GH, Hong B, Li XM, Zou Y, Zhang XJ. Gastrodin prevents motor deficits and oxidative stress in the MPTP mouse model of Parkinson’s disease: involvement of ERK1/2-Nrf2 signaling pathway. Life Sci. 2014;114(2):77-85. doi: 10.1016/j.lfs.2014.08.004, PMID 25132361.
Khatri DK, Juvekar AR. Neuroprotective effect of curcumin as evinced by abrogation of rotenone-induced motor deficits, oxidative and mitochondrial dysfunctions in mouse model of Parkinson’s disease. Pharmacol Biochem Behav. 2016;150-151:39-47. doi: 10.1016/j.pbb.2016.09.002, PMID 27619637.
Ruan S, Xie J, Wang L, Guo L, Li Y, Fan W. Nicotine alleviates MPTP-induced nigrostriatal damage through modulation of JNK and ERK signaling pathways in the mice model of Parkinson’s disease. Front Pharmacol. 2023 Feb;14:1088957. doi: 10.3389/fphar.2023.1088957, PMID 36817162.
Harish G Bagewadi, Rajeshwari, Patil Banderao V, Syed Hasan Zahid. Rotarod test and catalepsy bar test: behavioral testing and neuromodulation of Aloe vera in MPTP-induced Parkinson’s disease animal model. Int J Basic Clin Pharmacol. 2018;7(3):494. doi: 10.18203/2319-2003.ijbcp20180663.