
Int J Curr Pharm Res, Vol 18, Issue 1, 23-29Original Article
EFFICACY OF PCSK9 INHIBITORS IN ADDITION TO BACKGROUND STATIN THERAPY COMPARED WITH PLACEBO IN ACHIEVING LDL-C TARGETS IN ADULTS WITH HYPERLIPIDAEMIA: A SYSTEMATIC REVIEW AND META-ANALYSIS OF RANDOMIZED CONTROLLED TRIALS
PRANAB DAS1*, MUKUNDAM BORAH2
1Department of Pharmacology, Pragjyotishpur Medical College and Hospital, Guwahati, Assam, India. 2Department of Pharmacology, Gauhati Medical College and Hospital, Guwahati, Assam, India
*Corresponding author: Pranab Das; *Email: pranabdas2580123@gmail.com
Received: 07 Oct 2025, Revised and Accepted: 27 Nov 2025
ABSTRACT
Objective: To evaluate the effect of Proprotein Convertase Subtilisin/Kexin type 9 (PCSK9) inhibitors, compared with placebo, when added to background statin therapy, on the proportion of adults with hyperlipidaemia achieving low-density lipoprotein cholesterol (LDL-C)<1.8 mmol/l or ≥50% reduction from baseline.
Methods: A systematic search of PubMed, Scopus, Cochrane Library, and Google Scholar (2015–2025) identified randomized controlled trials (RCTs) enrolling adults (≥18 y) with hyperlipidaemia on background statin therapy. Eligible trials compared any PCSK9 inhibitor to placebo, reporting binary outcomes for LDL-C target attainment. Data extraction and risk of bias assessment followed PRISMA 2020 guidelines. Random-effects meta-analysis was performed to calculate pooled risk ratios (RR) with 95% confidence intervals (CI). Heterogeneity (I²), publication bias, and sensitivity analysis were conducted.
Results: Five RCTs (n>50,000 participants) were included. PCSK9 inhibitors significantly increased the likelihood of achieving LDL-C targets versus placebo (pooled RR = 2.57, 95% CI: 2.52–2.63). Individual study RRs ranged from 1.89 to 31.37. Heterogeneity was high (I² = 99.86%), largely influenced by one outlier study; removal of this trial reduced I² to 61.39%. All studies had low risk of bias, and no evidence of publication bias was detected. The GRADE certainty of evidence was rated as moderate, downgraded for inconsistency.
Conclusion: Adding PCSK9 inhibitors to statin therapy substantially improves LDL-C target attainment in high-risk hyperlipidaemia patients, representing a potent adjunctive strategy when conventional lipid-lowering approaches are insufficient.
Keywords: PCSK9 inhibitors, Statins, Hyperlipidaemia, LDL-C reduction, Meta-analysis, Lipid-lowering therapy, Systematic review, Dyslipidaemia
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i1.7091 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Hyperlipidaemia, characterized by elevated levels of total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), and triglycerides (TG), with or without reduced high-density lipoprotein cholesterol (HDL-C), is a major risk factor for the development of atherosclerotic cardiovascular disease (ASCVD) [1]. The global burden of hyperlipidaemia is increasing, influenced by dietary habits, sedentary lifestyles, and metabolic disorders such as type 2 diabetes mellitus (T2DM) [2]. Untreated lipid abnormalities contribute to endothelial dysfunction, vascular inflammation, and accelerated atherosclerosis, ultimately leading to coronary artery disease, stroke, and peripheral vascular disease [3].
Epidemiological studies have demonstrated that age and gender significantly influence lipid profiles. Postmenopausal women often exhibit higher TC, LDL-C, and TG levels than men of the same age group, which may be attributed to hormonal changes [1]. Additionally, age progression is associated with a proportional increase in lipid parameters, underscoring the importance of early detection and lifestyle interventions to reduce cardiovascular risk [4]. In patients with T2DM, dyslipidaemia presents as elevated TG, small dense LDL particles, and reduced HDL-C, which markedly increases cardiovascular risk [4].
Current first-line pharmacological therapy for hyperlipidaemia involves statins, which inhibit HMG-CoA reductase and effectively lower LDL-C. However, even with maximally tolerated doses, many high-and very-high-risk patients fail to achieve LDL-C targets recommended by international guidelines (<1.8 mmol/l or<70 mg/dl, or ≥50% reduction from baseline) [5]. This treatment gap highlights the need for adjunctive lipid-lowering therapies to optimize cardiovascular risk reduction.
Proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors, such as evolocumab, alirocumab, and inclisiran, have emerged as potent agents that reduce LDL-C by preventing degradation of hepatic LDL receptors. When combined with statin therapy, PCSK9 inhibitors have demonstrated significant additional LDL-C lowering and a higher likelihood of achieving therapeutic targets, with favourable tolerability profiles [6].
While several randomized controlled trials (RCTs) have evaluated the efficacy of PCSK9 inhibitors, no meta-analysis has specifically synthesized evidence on their effect in addition to background statin therapy for achieving LDL-C<1.8 mmol/l or ≥50% reduction from baseline in adults with hyperlipidaemia. Such evidence is essential for guiding clinicians in selecting optimal lipid-lowering regimens for high-risk patients.
The objective of the meta-analysis is to evaluate the effect of PCSK9 inhibitors, compared with placebo, when added to background statin therapy, on the proportion of adults with hyperlipidaemia achieving LDL-C<1.8 mmol/l or ≥50% reduction from baseline.
MATERIALS AND METHODS
Research question
In adults (≥18 y) with hyperlipidaemia receiving stable background statin therapy (with or without ezetimibe), does the addition of PCSK9 inhibitors, compared with placebo, increase the proportion of patients achieving LDL-C<1.8 mmol/l (<70 mg/dl) or ≥50% reduction from baseline LDL-C levels?
Study design and reporting standards
This research utilised a quantitative meta-analytical methodology based on data acquired from qualifying investigations. The execution and documentation of the meta-analysis adhered to the 2020 Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines to ensure transparency, reproducibility, and scientific rigour [7]. This meta-analysis just employed existing literature and did not incorporate any novel data from humans or animals. Consequently, ethical approval or institutional review board authorisation was unnecessary.
Eligibility criteria
This review considered randomized controlled trials enrolling adults (≥18 y), with or without established atherosclerotic cardiovascular disease (ASCVD) or high cardiovascular risk, diagnosed with familial or non-familial hyperlipidaemia and receiving stable background statin therapy, with or without ezetimibe. Eligible studies evaluated any PCSK9 inhibitor, irrespective of type, dose, or regimen, in combination with statin therapy, compared with placebo plus the same background statin regimen. Only articles published between 2015 and 2025 in peer-reviewed, indexed journals written in English language were included. Trials were required to report binary outcomes, that is the proportion of participants achieving LDL-C<1.8 mmol/l (<70 mg/dl) or ≥50% reduction from baseline with a minimum follow-up of four weeks. Exclusion criteria comprised non-randomized designs, observational studies, reviews, or meta-analysis; absence of background statin therapy in both arms; head-to-head comparisons of PCSK9 inhibitors with statin monotherapy; reporting only mean LDL-C change without sufficient data for binary outcome calculation; and duplicate datasets.
Search strategy
After conducting a comprehensive search of electronic databases, the researchers were able to discover research that met the criteria for consideration. A number of databases, such as Google Scholar, Scopus, PubMed, and the Cochrane Library, were searched by the researchers.
Boolean operators, along with the keywords, such as the following, were incorporated into the search method:
("PCSK9 inhibitor" or “evolocumab” or “alirocumab” or “inclisiran” or “tafolecimab”) and ("hyperlipidaemia" or “dyslipidaemia” or "familial hypercholesterolemia") and ("statin" or "HMG-CoA reductase inhibitor") and ("randomized controlled trial" or “RCT” or "clinical trial") and ("LDL-C" or "low-density lipoprotein cholesterol" or "cholesterol reduction")
Study selection process
The research selection method adhered to the PRISMA 2020 paradigm, comprising four principal phases: identification, screening, eligibility, and inclusion. In the identification step, records were obtained through extensive searches of electronic databases. During the screening step, titles and abstracts were evaluated for their pertinence. Subsequently, full-text articles were assessed for eligibility according to established inclusion and exclusion criteria. Studies that fulfilled all eligibility criteria were incorporated into the final meta-analysis. Duplicate records were eliminated, and the rationale for removal was recorded at each phase of the procedure. In conclusion, five studies were chosen for inclusion, each offering binary outcome data appropriate for analysis. Two independent reviewers screened all retrieved titles and abstracts against the eligibility criteria. Any disagreements between the two reviewers were resolved through discussion, and, when necessary, adjudication by a third independent reviewer.
Data extraction process
Data extraction was performed independently by two reviewers using a standardized Microsoft Excel 365 form. Extracted variables included study characteristics (study ID, author, year, sample size, follow-up duration), participant demographics, intervention details (PCSK9 inhibitor type and dose), comparator details, and binary outcome data (number achieving LDL-C<1.8 mmol/l or ≥50% reduction, total number randomized per arm). Discrepancies were resolved by consensus or referral to a third reviewer.
Statistical analysis
All statistical analyses were performed using Microsoft Excel 365. For each included trial, risk ratios (RR) with 95% confidence intervals (CI) were calculated for the primary outcome. A random-effects model was applied to pool effect sizes, accounting for between-study variability. Statistical heterogeneity was assessed using the I² statistic, with thresholds of 25%, 50%, and 75% representing low, moderate, and high heterogeneity, respectively. Publication bias was evaluated visually through funnel plot and Egger’s Test. Sensitivity analyses and subgroup analyses were conducted where applicable to assess the robustness of findings.
RESULTS
Study selection and sample characteristics
A total of 41 records were identified through the initial database search. After the removal of 9 duplicates, 32 unique records were screened based on title and abstract. Of these, 24 records were excluded for not meeting the eligibility criteria, leaving 8 full-text articles for detailed assessment. Following full-text evaluation, 3 studies were excluded for reasons such as ineligible study design, absence of required binary LDL-C outcomes, or intervention not meeting the criteria. Ultimately, 5 randomized controlled trials fulfilled all inclusion criteria and were included in both the qualitative synthesis and quantitative meta-analysis.
The included studies enrolled adult participants (≥18 y) with or without established atherosclerotic cardiovascular disease or high cardiovascular risk, all of whom were on stable background statin therapy, with or without ezetimibe. The trials varied in sample size, follow-up duration, and type of PCSK9 inhibitor used, but all reported the proportion of participants achieving LDL-C<1.8 mmol/l (<70 mg/dl) or ≥50% reduction from baseline levels either directly or indirectly. The detailed PRISMA summary of the selection process is provided in table 1.
Table 1: PRISMA summary table
| Stage | Frequency (n) |
| Records identified through database searching | 41 |
| Duplicates removed | 9 |
| Records screened | 32 |
| Records excluded | 24 |
| Full-text articles assessed for eligibility | 8 |
| Full-text articles excluded | 3 |
| Studies included in qualitative and quantitative synthesis | 5 |
Study characteristics
Details of study attributes which were included in this research are displayed in table 2.
Meta-analysis findings
The effect estimates of the meta-analysis are summarised below in table 3.
Interpretation of table 3
The five included randomized trials showed RRs ranging from 1.89 (S4) to 31.37 (S1). Larger studies (S4, S5) had narrower confidence intervals, indicating greater precision. Trials S2, S3, and S5 reported RRs above 7, highlighting a notably higher achievement of LDL-C targets with PCSK9 inhibitors versus placebo. The pooled RR was 2.57 (95% CI: 2.52–2.63), confirming a significant overall benefit of adding PCSK9 inhibitors to background statin therapy.
Forest plot
Risk ratios (RR) with 95% confidence intervals (CIs) are shown in the below forest plot for each included randomized controlled trial, along with the pooled effect estimate from the random-effects meta-analysis (fig. 1).
Table 2: Attributes of included randomized controlled trials (RCTs)
| Study ID | First Author (Year) | Study design | Population | Intervention (PCSK9i) | Comparator | Intervention dose | Comparator dose | Primary Outcome | Proportion of patients achieving target outcome in Intervention group* | Proportion of patients achieving target outcome in Comparator group* | Duration (weeks) |
| S1 | Li Q et al. (2023) [8] | RCT | Heterozygous familial hypercholesterolemia (HeFH) | Tafolecimab | Placebo | 450 mg SC every 4 w | Matching placebo SC | LDL-C<1.8 mmol/l or ≥50% reduction | 193/201 | 3/98 | 12 |
| S2 | Ray et al. (2020) (Orion-10 Trial) [9] | RCT | ASCVD with elevated LDL-C | Inclisiran | Placebo | 300 mg SC on day 1, day 90, then every 6 mo | Matching placebo SC | LDL-C<1.8 mmol/l or ≥50% reduction | 605/781 | 84/780 | 73 |
| S3 | Robinson et al. (2015) [10] | RCT | Hyperlipidaemia with high cardiovascular risk | Alirocumab | Placebo | 150 mg SC every 2 w (adjusted if needed) | Matching placebo SC | LDL-C<1.8 mmol/l or ≥50% reduction | 1213/1530 | 62/780 | 24 |
| S4 | Schwartz et al. (2018) [11] | RCT | Acute coronary syndrome with elevated LDL-C | Alirocumab | Placebo | 75 mg SC every 2 w, titrated to 150 mg if LDL-C above target | Matching placebo SC | LDL-C<1.8 mmol/l or ≥50% reduction | 7945/9462 | 4214/9462 | 144 |
| S5 | Sabatine et al. (2017) [12] | RCT | ASCVD with elevated LDL-C | Evolocumab | Placebo | 140 mg SC every 2 w or 420 mg monthly | Matching placebo SC | LDL-C<1.8 mmol/l or ≥50% reduction | 11899/13784 | 1515/13780 | 48 |
*Proportion of patients achieving target outcome in intervention group = Sample size of intervention group showing the desired outcome/Total sample size of intervention group. **Proportion of patients achieving target outcome in Comparator group = Sample size of comparator group showing the desired outcome/Total sample size of comparator group.
Table 3: Summary of effect estimates for included randomized controlled trials
| Study ID | RR | Log(RR) | SE | Lower 95% CI (Individual Study) | Upper 95% CI (Individual Study) | Weight | Weighted Log(RR) | Pooled RR | SE of Pooled Log(RR) | Lower 95% CI (Meta-analysis) | Upper 95% CI (Meta-analysis) |
| S1 | 31.37 | 3.4457 | 0.5686 | 10.29 | 95.61 | 3.09 | 10.6569 | 2.5747 | 0.0109 | 2.5203 | 2.6303 |
| S2 | 7.19 | 1.9731 | 0.1049 | 5.86 | 8.83 | 90.95 | 179.4539 | ||||
| S3 | 9.97 | 2.3000 | 0.1225 | 7.84 | 12.68 | 66.59 | 153.1509 | ||||
| S4 | 1.89 | 0.6341 | 0.0123 | 1.84 | 1.93 | 6587.71 | 4177.4697 | ||||
| S5 | 7.85 | 2.0607 | 0.0245 | 7.48 | 8.24 | 1669.48 | 3440.3732 |
RR: Risk Ratio; SE: Standard Error; CI: Confidence Interval.
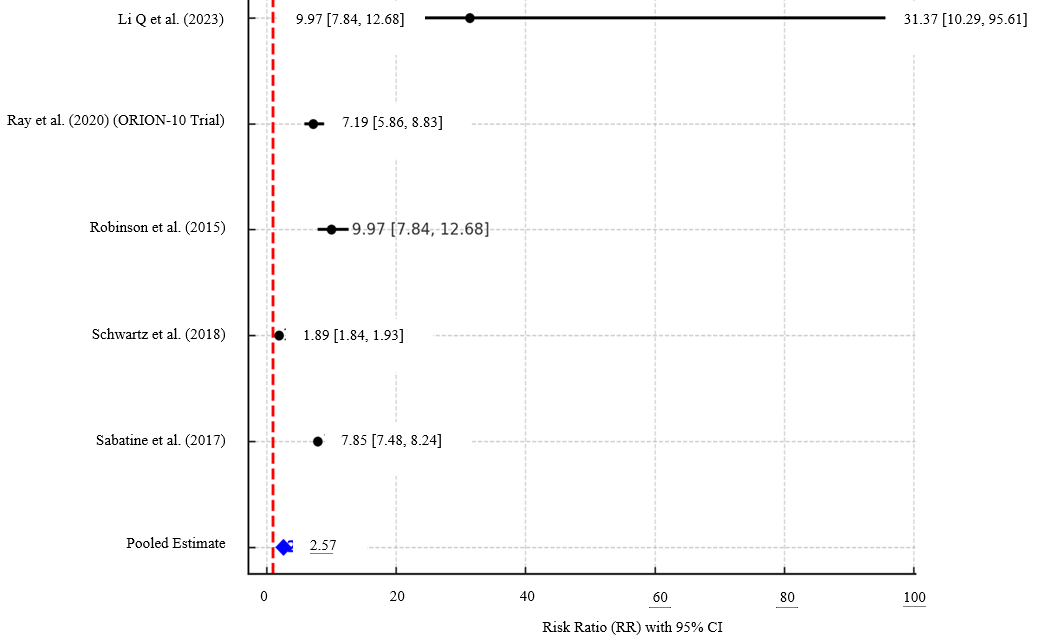
Fig. 1: Forest plot comparing PCSK9 inhibitors versus placebo in achieving LDL-C targets (<1.8 mmol/l or ≥50% reduction) among adults on background statin therapy
Interpretation of forest plot
The forest plot demonstrates that all included trials favoured PCSK9 inhibitors over placebo for achieving LDL-C targets. Individual study RRs ranged from 1.89 to 31.37, with Li et al. (2023) showing the largest effect size but also the widest confidence interval, reflecting greater uncertainty due to smaller sample size. Larger studies such as Sabatine et al. (2017) and Schwartz et al. (2018) yielded narrower CIs, indicating higher precision. The pooled RR was 2.57 (95% CI: 2.52–2.63), confirming a statistically significant and clinically meaningful benefit of adding PCSK9 inhibitors to background statin therapy.
Publication bias assessment
Table 4 shows Egger’s regression test assessing small-study effects among trials included in the meta-analysis of PCSK9 inhibitors versus placebo in the background of statin therapy for LDL-C target achievement. The slope (β₁) and intercept (β₀) were derived using precision as the independent variable and standardized effect size as the dependent variable.
Table 4: Egger’s regression test for publication bias among included randomized controlled trials
| Study ID | Log(RR) | SE | Inverse SE (Precision) | Standardized Effect Size | β₁ (Slope) | β₀ (Intercept) | SE of β₀ (Intercept) | t-value | df | p-value |
| S1 | 3.4457 | 0.5686 | 1.7586 | 6.0598 | 0.6540 | 17.3566 | 16.5812 | 1.0468 | 3 | 0.3721 |
| S2 | 1.9731 | 0.1049 | 9.5367 | 18.8172 | ||||||
| S3 | 2.3000 | 0.1225 | 8.1601 | 18.7682 | ||||||
| S4 | 0.6341 | 0.0123 | 81.1647 | 51.4690 | ||||||
| S5 | 2.0607 | 0.0245 | 40.8592 | 84.2006 |
Interpretation of egger’s regression test
Egger’s regression analysis demonstrated a non-significant intercept (p = 0.372, that is p>0.05), indicating no statistically detectable small-study effects or publication bias. This suggests that the pooled effect estimate is unlikely to be distorted by selective reporting or asymmetry in study results.
Fig. 2 shows the funnel plot assessment of the meta-analysis.
Interpretation of funnel plot
The funnel plot demonstrates an overall symmetrical distribution of studies around the pooled effect size, suggesting minimal risk of publication bias. While one study (S4) appears as an outlier with a wider standard error and effect size deviation, its influence does not substantially distort the plot’s symmetry. This observation is consistent with Egger’s regression test, which yielded a non-significant result (p = 0.372), indicating no statistical evidence of small-study effects. The presence of the outlier may slightly widen the confidence boundaries but is unlikely to meaningfully affect the robustness of the pooled estimate.
Risk of bias assessment
Table 5 shows the Risk of Bias (RoB) assessment of the meta-analysis as per the Cochrane RoB 2 guidance tool [13].
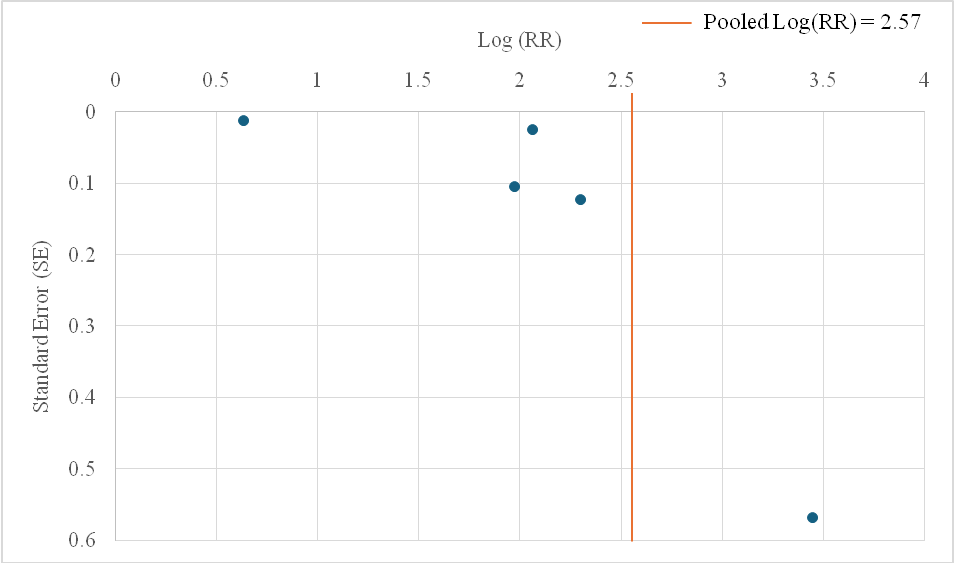
Fig. 2: Funnel plot assessing publication bias among the included studies comparing PCSK9 inhibitors versus placebo for LDL-C target attainment. The vertical red dashed line represents the pooled log(RR), and the blue dashed lines indicate pseudo 95% confidence limits
Table 5: Risk of bias (RoB) assessment for included randomized controlled trials using a domain-based evaluation
| Study ID | Domain 1 Judgment | Domain 2 Judgment | Domain 3 Judgment | Domain 4 Judgment | Domain 5 Judgment | Overall RoB | Justification |
| S1 | Low risk | Low risk | Low risk | Low risk | Low risk | Low | RCT with concealed randomization, double-blind, minimal loss to follow-up, outcome pre-specified. |
| S2 | Low risk | Low risk | Low risk | Low risk | Low risk | Low | Adequate randomization/blinding, outcome data complete, LDL-C endpoints pre-specified. |
| S3 | Low risk | Low risk | Low risk | Low risk | Low risk | Low | Randomized, double-blind, ITT analysis, near-complete follow-up, outcome definition standard. |
| S4 | Low risk | Low risk | Low risk | Low risk | Low risk | Low | Large double-blind RCT, balanced arms, pre-specified analysis, minimal missing data. |
| S5 | Low risk | Low risk | Low risk | Low risk | Low risk | Low | Well-conducted RCT with clear allocation concealment, blinding, and pre-registered outcomes. |
Interpretation of RoB assessment
All five studies included in this meta-analysis were judged to have a low risk of bias across all evaluated domains, reflecting strong methodological rigor and reliable outcome reporting. Adequate randomization, allocation concealment, blinding procedures, and adherence to pre-specified endpoints were consistently observed. Minimal loss to follow-up and clear outcome definitions further enhances the validity of the findings, thereby increasing confidence in the pooled effect estimates generated from these trials.
Heterogeneity assessment
Table 6 shows Cochrane’s Q statistic and I2statistic calculation for the heterogeneity assessment of the meta-analysis.
Interpretation of heterogeneity assessment
The heterogeneity assessment indicates a very high degree of variability among the included studies, with an I² value of approximately 99.86%, suggesting that most of the observed variance is due to differences between studies rather than chance. The Cochran's Q statistic further supports the presence of significant heterogeneity, implying that caution should be exercised when interpreting the pooled results. This level of heterogeneity underscores the need to explore potential sources of variability, such as differences in study populations, interventions, or methodological designs.
Leave-one-out sensitivity analysis
A leave-one-out sensitivity analysis was conducted to evaluate the influence of individual studies on the overall pooled effect and heterogeneity (table 7).
Interpretation
The sensitivity analysis revealed that removal of S4 substantially reduced heterogeneity (I² = 61.39%), indicating that this study contributed significantly to the observed variability. However, exclusion of any other single study did not materially alter the high heterogeneity (>98%) or the pooled effect estimate, suggesting overall robustness of the results despite the presence of outlier influence.
Subgroup analysis
A subgroup analysis was performed to assess whether study duration (≤48 w vs.>48 w) influenced the pooled effect estimates and heterogeneity (table 8).
Table 6: Heterogeneity assessment of included studies
| Study ID | ((Log RR-Pooled log RR)^2)*weight | Cochran's Q statistic | Degrees of freedom (df) | I2 statistic |
| S1 | 19.3297104 | 2952.68869 | 4 | 99.8645302 |
| S2 | 95.998525 | |||
| S3 | 122.12033 | |||
| S4 | 639.687637 | |||
| S5 | 2075.55249 |
Table 7: Leave-one-out sensitivity analysis of included studies
| Removed study ID | Pooled log(RR) | Q statistic | I² value (%) |
| S1 | 0.9448 | 2933.3519 | 99.90 |
| S2 | 0.9345 | 2855.6416 | 99.86 |
| S3 | 0.9349 | 2829.5946 | 99.86 |
| S4 | 2.0674 | 10.3600 | 61.39 |
| S5 | 0.6699 | 363.6638 | 98.90 |
Table 8: Subgroup analysis by study duration
| Study ID | Duration category | Pooled RR | Lower 95% CI | Upper 95% CI | I2value (%) |
| S1 | ≤48 w | 7.9436 | 7.578935138 | 8.325892729 | 78.97 |
| S3 | |||||
| S5 | |||||
| S2 | >48 w | 1.9201 | 5.23875E-70 | 7.03735E+69 | 99.97 |
| S4 |
Interpretation of subgroup analysis based on duration of treatment
The subgroup analysis indicated that studies with shorter follow-up durations (≤48 w) showed consistently high relative risk estimates, with high heterogeneity (I² = 78.97%). In contrast, longer-duration studies (>48 w) demonstrated substantial heterogeneity (I² = 99.97%), largely driven by variation in study design and outcome magnitude. This suggests that follow-up length may partially contribute to variability in effect size, but high heterogeneity persists across both subgroups.
GRADE assessment
The GRADE assessment of the meta-analysis is explained in table 9.
Table 9: GRADE assessment of certainty of evidence for LDL-C reduction with PCSK9 inhibitors
| Outcome | Risk of bias | Inconsistency | Indirectness | Imprecision | Publication bias | Overall certainty level | Justification |
| LDL-C reduction (RR = 2.57, 95% CI: 2.52–2.63) | No | Yes | No | No | No | Moderate | All included RCTs had low RoB. Very high heterogeneity (I² = 99.86%) downgraded by one level for inconsistency. Populations, interventions, comparators, and outcomes directly aligned with PICO. CI was narrow and distant from the null line (no effect line), so no downgrade for imprecision. Funnel plot was symmetric and Egger’s p = 0.249, indicating no downgrade for publication bias. |
Interpretation of GRADE assessment
The certainty of evidence for LDL-C reduction with PCSK9 inhibitors was rated as moderate. Although the body of evidence consisted of well-conducted RCTs with minimal risk of bias, the presence of substantial heterogeneity (I² = 99.86%) warranted a downgrade for inconsistency. No downgrades were made for indirectness, imprecision, or publication bias, as the included studies directly addressed the research question, produced precise effect estimates, and showed no signs of small-study effects.
DISCUSSION
This meta-analysis combined data from five rigorously conducted randomised controlled trials (RCTs) to evaluate the efficacy of PCSK9 inhibitors, added to stable statin therapy with or without ezetimibe, in achieving clinically relevant LDL-C targets (<1.8 mmol/l or ≥50% reduction from baseline). The pooled analysis demonstrated a robust and statistically significant benefit, with a pooled risk ratio (RR) of 2.57 (95% CI: 2.52–2.63). This indicates that patients treated with PCSK9 inhibitors were more than twice as likely to reach LDL-C goals compared with placebo. These findings closely parallel results from major cardiovascular outcome trials, such as Fourier (evolocumab) and Odyssey Outcomes (alirocumab), which also reported high target attainment rates and marked LDL-C reductions when PCSK9 inhibitors were added to maximally tolerated statins [11, 12].
The effect sizes observed in this analysis align with the Fourier trial, where evolocumab achieved a ~59% LDL-C reduction from baseline, with 87% of patients reaching<1.8 mmol/l [12], and with Odyssey Outcomes, where alirocumab lowered LDL-C by ~54% and enabled the majority to meet stringent lipid targets [11]. High RRs in several included trials (e. g., Li et al., 2023; Robinson et al., 2015) were driven by the low attainment of LDL-C goals in placebo arms, reflecting the challenge of achieving these levels with statin±ezetimibe alone in high-risk populations.
Previous meta-analyses have reported similar magnitudes of LDL-C lowering with PCSK9 inhibitors, though often expressed as mean percentage change rather than binary target achievement. For instance, Navarese et al. (2015) observed a pooled LDL-C reduction of −49.5% [14]. Our focus on binary outcomes makes the findings directly applicable to current guideline-defined treatment targets, enhancing their clinical relevance.
Heterogeneity in the present analysis was substantial (I² = 99.86%), which persisted even after subgrouping by follow-up duration, although removal of Schwartz et al. (2018) reduced I² to 61.39%. The prolonged follow-up (144 w), titration strategies, and differences in baseline risk in that trial likely contributed to its outlier influence. Persistent high heterogeneity across subgroups suggests that factors such as baseline LDL-C, ASCVD burden, and PCSK9 inhibitor type or dosing regimen may also underlie variability in effect estimates. This pattern is consistent with prior meta-analyses that found high I² values when pooling diverse PCSK9 inhibitor RCTs [14, 15].
The GRADE assessment rated the certainty of evidence as moderate, with a single downgrade for inconsistency due to heterogeneity. All included RCTs had low risk of bias in every Cochrane RoB 2 domain, and neither Egger’s test (p = 0.372) nor the funnel plot suggested publication bias. The leave-one-out sensitivity analysis further confirmed that no single trial materially altered the pooled RR, indicating robustness of the overall effect estimate.
While the LDL-C lowering efficacy of PCSK9 inhibitors is established, its translation into cardiovascular event reduction warrants emphasis. In FOURIER and ODYSSEY OUTCOMES, relative risk reductions in major adverse cardiovascular events (MACE) were modest over 2–3 y, but absolute event reductions were greater in patients achieving very low LDL-C (<0.78 mmol/l) [11, 12]. This supports the likelihood that the degree of LDL-C lowering observed in our pooled analysis would translate into meaningful cardiovascular benefit, especially in very high-risk groups, provided therapy is maintained long-term.
This meta-analysis adhered to PRISMA 2020 methodology and included only high-quality RCTs with low risk of bias, consistent background statin therapy, and directly relevant binary endpoints aligned with clinical guidelines. Multiple PCSK9 inhibitors were represented, enhancing generalisability. The narrow-pooled confidence interval and absence of publication bias increase confidence in the validity of results.
The analysis was limited by the small number of included trials, which restricted the ability to conduct detailed subgroup analyses or meta-regression. Only trial-level data were available, preventing adjustment for patient-level factors such as age, comorbidities, or baseline LDL-C. The very high heterogeneity reduces the precision of pooled effect size interpretation, although robustness was supported by sensitivity analyses. The binary outcome approach, while clinically intuitive, may not capture the full range of LDL-C reductions achieved.
Future research should employ individual participant data (IPD) meta-analyses to identify predictors of LDL-C goal attainment with PCSK9 inhibitors, evaluate the long-term sustainability of lipid lowering beyond five years, and quantify cardiovascular event reduction in relation to achieved LDL-C thresholds. Real-world studies assessing cost-effectiveness, adherence, and outcomes in broader patient populations will be essential to guide clinical and policy decisions.
CONCLUSION
In summary, this meta-analysis demonstrates that adding PCSK9 inhibitors to statin therapy substantially increases the likelihood of achieving stringent LDL-C targets, offering a powerful therapeutic option for high-risk patients where conventional lipid-lowering strategies are insufficient.
ACKNOWLEDGEMENT
The authors express their sincere gratitude to all the researchers whose original work formed the basis of this analysis and to the peer reviewers for their constructive insights.
FUNDING
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
AUTHORS CONTRIBUTIONS
Pranab Das conceived and designed the study, conducted the literature search, extracted and analyzed the data, and drafted the manuscript, while Mukundam Borah contributed to data validation, interpretation of findings, and critical revision of the manuscript; both authors approved the final version and take full responsibility for the integrity and accuracy of the work.
CONFLICT OF INTERESTS
None of the authors have any conflicts of interest to declare.
REFERENCES
Babu A, Rani S, Revikumar KG, Mattumal S, Unnikrishnan UG. Assessment of correlation in gender and age with lipoprotein levels in hyperlipidemia patients. Asian J Pharm Clin Res. 2016;9(2):197-9. doi: 10.22159/ajpcr.2016.v9i2.10373.
Singh P, Nanda A. Recent patents on solid dispersions of antihyperlipidemic drugs. Int J App Pharm. 2023;15(2):23-31. doi: 10.22159/ijap.2023v15i2.42402.
Aly HF, Abd-Alla HI, Ali SA, Mamdooh MM, Abo Alez R, Abu-Krisha MT. Statistical data analysis which result from the bio-diagnosis and bio-treatment of injured rats with the hyperlipidemia and hyperglycemia diseases. Asian J Pharm Clin Res. 2016;9(Suppl 3):152-8. doi: 10.22159/ajpcr.2016.v9s3.14439.
Khan NA, Venkatachalam VV, Al-Akhali KM, Alavudeen SS, Dhanapal CK, Ansari SM. Therapeutic management of hypertension and hyperlipidemia in type-2 diabetes mellitus patients in southwestern region of Saudi Arabia: a pharmacist perspective. Asian J Pharm Clin Res. 2014;7(5):241-5.
Grundy SM, Cleeman JI, Merz CNB, Brewer HB JR, Clark LT, Hunninghake DB, Pasternak RC, Smith SC, Stone NJ. National cholesterol education program (NCEP) expert panel on detection evaluation and treatment of high blood cholesterol in adults (adult treatment panel iii). Third report of the national cholesterol education program (NCEP) expert panel on detection, evaluation and treatment of high blood cholesterol in adults (adult treatment panel iii) final report. Circulation. 2002;106(25):3143-421. doi: 10.1161/circ.106.25.3143, PMID 12485966.
Akash V, Shailaja P. In vitro and in vivo evaluation of ezetimibe fast-dissolving films. Int J App Pharm. 2023;15(6):108-17. doi: 10.22159/ijap.2023v15i6.49110.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021 Mar 29;372:n71. doi: 10.1136/bmj.n71, PMID 33782057.
Chai M, He Y, Zhao W, Han X, Zhao G, Ma X. Efficacy and safety of tafolecimab in Chinese patients with heterozygous familial hypercholesterolemia: a randomized double-blind placebo-controlled phase 3 trial (CREDIT-2). BMC Med. 2023 Feb 28;21(1):77. doi: 10.1186/s12916-023-02797-8, PMID 36855099.
Ray KK, Wright RS, Kallend D, Koenig W, Leiter LA, Raal FJ. Two phase 3 trials of inclisiran in patients with elevated LDL cholesterol. N Engl J Med. 2020 Apr 16;382(16):1507-19. doi: 10.1056/NEJMoa1912387, PMID 32187462.
Robinson JG, Farnier M, Krempf M, Bergeron J, Luc G, Averna M. Efficacy and safety of alirocumab in reducing lipids and cardiovascular events. N Engl J Med. 2015 Apr 16;372(16):1489-99. doi: 10.1056/NEJMoa1501031, PMID 25773378.
Schwartz GG, Steg PG, Szarek M, Bhatt DL, Bittner VA, Diaz R. Alirocumab and cardiovascular outcomes after acute coronary syndrome. N Engl J Med. 2018 Nov 29;379(22):2097-107. doi: 10.1056/NEJMoa1801174, PMID 30403574.
Sabatine MS, Giugliano RP, Keech AC, Honarpour N, Wiviott SD, Murphy SA. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med. 2017 May 4;376(18):1713-22. doi: 10.1056/NEJMoa1615664, PMID 28304224.
Sterne JAC, Savovic J, Page MJ, Elbers RG, Blencowe NS, Boutron I. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. doi: 10.1136/bmj.l4898.
Navarese EP, Kolodziejczak M, Schulze V, Gurbel PA, Tantry U, Lin Y. Effects of proprotein convertase subtilisin/kexin type 9 antibodies in adults with hypercholesterolemia: a systematic review and meta-analysis. Ann Intern Med. 2015 Jul 7;163(1):40-51. doi: 10.7326/M14-2957, PMID 25915661.
Zhang XL, Zhu QQ, Zhu L, Chen JZ, Chen QH, Li GN. Safety and efficacy of anti-PCSK9 antibodies: a meta-analysis of 25 randomized controlled trials. BMC Med. 2015 Jun 23;13:123. doi: 10.1186/s12916-015-0358-8, PMID 26099511.