
Int J Curr Pharm Res, Vol 18, Issue 1, 36-41Original Article
RISK FACTORS, MICROBIOLOGICAL SPECTRUM, ANTIBIOTIC SENSITIVITY AND OUTCOMES OF VENITILATOR ASSOCIATED PNEUMONIA IN NEONATES: A STUDY FROM A TERTIARY CARE HOSPITAL IN SOUTHERN ASSAM
VEERANNA SHIVANAND CHIKKAMATH1, M. SWATHI PATIL2, NILANJAN BISWAS3, PRANAB DAS4*, SUJIT NATH CHOUDHURY5
1,2,3Department of Paediatrics, Silchar Medical College and Hospital, Assam, India. 4Department of Pharmacology, Silchar Medical College and Hospital, Assam, India. 5Department of Pharmacology, Pragjyotishpur Medical College and Hospital, Assam, India
*Corresponding author: Pranab Das; *Email: pranabdas2580123@gmail.com
Received: 09 Oct 2025, Revised and Accepted: 28 Nov 2025
ABSTRACT
Objective: To determine the incidence, microbiological profile, antibiotic sensitivity, risk factors, and outcomes of VAP in neonates.
Methods: This prospective observational study, conducted for one year in a tertiary care NICU in Southern Assam, included 101 neonates ventilated for over 48 h. VAP was defined using CDC criteria, integrating clinical, Laboratory, radiological, and microbiological findings. Data on patient characteristics, VAP occurrence, microbiology, and antibiotic susceptibility were collected and analysed.
Results: Thirty-seven (36.7%) neonates developed VAP, resulting in an incidence of 41.11 per 1,000 ventilator days. Key risk factors significantly associated with VAP included prematurity, low birth weight, bronchopulmonary dysplasia, and asphyxia. Common clinical signs were new onset tachypnoea and increased respiratory secretions. VAP led to significantly longer hospital stays (38.1±17.82 vs. 21.4±14.3 d, p=0.0001) and mechanical ventilation durations (13.42±8.74 vs. 5.89±3.56 d, p=0.0001), alongside increased antibiotic use. Gram-negative organisms predominated (68.9% of 45 isolates), notably multi-drug resistant (MDR) Acinetobacter spp. and ESBL-producing Klebsiella pneumoniae/oxytoca. Pseudomonas isolates showed high resistance to Amikacin, Meropenem, and Piperacillin+Tazobactam. MRSA exhibited good Linezolid susceptibility. VAP is associated with increased morbidity and mortality.
Conclusion: Neonatal VAP imposes a substantial burden, driven by prematurity, severe underlying conditions, and a high prevalence of MDR Gram-negative pathogens. Our findings highlight the urgent need for robust infection control, vigilant surveillance, and culture-guided antibiotic therapy to optimize outcomes and combat antimicrobial resistance. Larger, multi-center studies are essential.
Keywords: Neonatal VAP, Antimicrobial resistance, Multi-drug resistant (MDR), NICU, Gram-negative bacteria
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i1.8008 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Ventilator-associated pneumonia (VAP) is a serious, frequent hospital-acquired infection in Neonatal Intensive Care Units (NICUs), notably impacting neonates on prolonged mechanical ventilation. Defined as pneumonia developing after 48 h of intubation, VAP increases morbidity, mortality, hospital stays, and healthcare costs [1, 2]. Neonates, especially preterm or low birth weight infants, are highly susceptible due to immature immune systems, fragile lungs, and invasive procedures [3, 4].
Despite VAP research in pediatrics, specific studies on neonates, particularly low birth weight infants, are limited [5]. Key risk factors include prolonged ventilation, frequent suctioning, reintubation, prematurity, low birth weight, and prior broad-spectrum antibiotic use [5, 6]. The microbiological landscape often features Gram-negative bacilli (e. g., Klebsiella pneumoniae, Pseudomonas aeruginosa, Escherichia coli) and Gram-positive cocci (e. g., Staphylococcus aureus) [7, 8]. Multidrug-resistant (MDR) organisms are increasingly problematic, emphasizing the need for antibiotic stewardship and targeted therapy [9].
Diagnosing neonatal VAP is complex due to non-specific clinical signs and lack of a gold standard. Criteria typically include new respiratory symptoms, radiological infiltrates, and microbiological confirmation from tracheal aspirates [10, 11]. Guidelines like the CDC's aim to standardize definitions for better accuracy and surveillance [12].
Outcomes of neonatal VAP vary from prolonged respiratory support to increased mortality, especially in extremely low birth weight and critically ill infants [13, 14]. Given its substantial clinical and economic burden, understanding risk factors, accurate diagnosis, and local microbial resistance patterns is vital for optimizing NICU protocols.
MATERIALS AND METHODS
Study design
This was a prospective, observational study aimed at determining the incidence, microbiological profile, antibiotic sensitivity, risk factors, and outcomes of VAP in neonates admitted to the NICU. The prospective design enabled real-time data collection and close monitoring of clinical, microbiological, and treatment variables. Standardized diagnostic criteria for VAP were applied.
Study setting
The study was conducted in the Neonatal Intensive Care Unit (NICU) of Silchar Medical College and Hospital, Assam, a tertiary care referral centre serving the southern Assam and Neighbouring Northeastern States. The hospital’s high patient volume, well-equipped NICU, and comprehensive laboratory facilities provided a representative neonatal population and reliable microbiological diagnostics essential for this research.
Study duration
Study was conducted for 1 y (May 2024 to June 2025).
Study population
We included all neonates in the NICU admitted in our unit and who needed over 48 h of mechanical ventilation via an endotracheal tube. However, we excluded those born with major congenital malformations, those with an APGAR score below 4 at five minutes of birth, and any whose guardians didn't provide informed written consent.
VAP definition and identification
Neonatal VAP is defined as pneumonia developing ≥48 h after starting endotracheal mechanical ventilation. Diagnosis, as per CDC guidelines [10], relies on a combination of criteria: clinical symptoms (e. g., worsening respiratory status, temperature instability, altered tracheal secretions, apnea/bradycardia), laboratory findings (e. g., elevated CRP, abnormal white blood cell count, thrombocytopenia), and radiological evidence (new/progressive chest X-ray infiltrates). Isolation of pathogens from respiratory samples (tracheal aspirates, BAL, PSB), especially with purulent secretions, provides supportive microbiological confirmation.
Sample size and collection
Neonates born on the unit’s emergency service days at Silchar Medical College, who met the inclusion criteria, were enrolled in the study after obtaining parental consent, irrespective of their delivery method. Total of 101 babies were included in the study during the study period. Consecutive sampling technique was chosen to minimize selection bias and to ensure that the sample accurately represented the overall population of neonates in the institution.
Data collection and statistical analysis
Data were collected via a structured proforma detailing patient history (gender, gestational age, birth weight, delivery mode, APGAR scores), ventilation specifics (duration, intubation), and VAP occurrence. Microbiological analysis involved tracheal aspirates and blood cultures. SPSS software was used for analysis: continuous variables were presented as means±SD, categorical as frequencies/percentages. Groups were compared using chi-square/Fisher’s exact tests for categorical data and t-tests/Mann-Whitney U tests for continuous data, with p<0.05 considered significant.
Ethical clearance
Upon receiving approval and clearance from the Institutional Ethics Committee (vide no. SMC/ETHICS/MI/2024/34), the research was started.
RESULTS
Out of 101 neonates requiring mechanical ventilation for over 48 h, 37 (36.7%) developed VAP, an incidence rate of 41.11 per 1,000 ventilator days. Although males outnumbered females (62 vs. 39), females had a slightly higher VAP incidence (41% vs. 33.8%). The 64 neonates who didn't develop VAP served as the control group (table 1).
Lower gestational age significantly correlated with higher VAP and mortality rates (p=0.028). Neonates born at ≤28 w gestational age had the highest VAP incidence (41.7%) and mortality (58.3%), contrasting with those>37 w (35% VAP, 25% mortality). Intermediate gestational age groups (34-37 w and 28-34 w) showed a progressive increase in VAP and mortality as gestational age decreased.
Lower birth weight was significantly associated with higher VAP and mortality (p=0.035). Neonates weighing less than 1000g showed the highest mortality rate (37.5%), despite varying VAP percentages across weight groups. The VAP group, overall, had a lower mean birth weight (1851grams).
Several clinical conditions and interventions were significantly more prevalent in the VAP group. Bronchopulmonary dysplasia (BPD) affected 37.83% of VAP patients versus 15.62% of non-VAP (p=0.012). Asphyxia was also notably higher in VAP neonates (62.16% vs. 28.12%, p=0.0008). Additionally, a higher percentage of VAP patients received surfactant therapy (45.94% vs. 26.56%, p=0.012). Mode of delivery, however, showed no significant difference (p=0.321).
Neonates with VAP experienced significantly longer hospital stays (38.1±17.82 d vs. 21.4±14.3 d; p=0.0001) and prolonged mechanical ventilation (13.42±8.74 d vs. 5.89±3.56 d; p=0.0001). The VAP group also had a higher reintubation rate (29.7% vs. 12.5%), though this difference was not statistically significant (p=0.33).
Antibiotic utilization was significantly higher in the VAP group. Neonates with VAP received antibiotics for a much longer duration (34.34±11.73 d vs. 18.26±11.22 d; p<0.000001) and were administered a significantly greater number of different antibiotics (5.43±1.34 vs. 3.13±1.22; p<0.000001). Despite this increased antibiotic exposure and overall morbidity in the VAP group, mortality rates did not differ significantly between the VAP (37.8%) and non-VAP (39.1%) cohorts (p=0.90).
Table 1: Baseline and clinical characteristics of VAP vs non-VAP neonates
| Variables | VAP(n=37) | Non VAP(n=64) | p-Value |
| Gender | |||
| Male | 21 (56.75%) | 41 (64.06%) | 0.46 |
| Female | 16 (43.24%) | 23 (35.93%) | |
| Gestational Age | |||
| >37 w | 14 (37.8%) | 26 (40.6%) | 0.028 |
| 37-34 w | 12 (32.4%) | 21 (32.8%) | |
| 34-28 w | 6 (16.4%) | 10 (15.6%) | |
| <28 w | 5 (13.6%) | 7 (11%) | |
| Birth weight | |||
| >2500g | 10(27.1%) | 21(32.8%) | 0.035 |
| 1500-2500 g | 11 (29.7%) | 17 (26.5%) | |
| 1000-1500g | 9 (24.3%) | 17 (26.5%) | |
| <1000 g | 7(18.9%) | 9 (14.1%) | |
| Mode of delivery | |||
| Caesarean section | 20 (54.05%) | 28 (43.75%) | 0.321 |
| Normal Vaginal delivery | 17 (45.95%) | 36 (56.25%) | |
| APGAR score | |||
| At 1 min | 4.9±2.8 | 5.1±2.7 | 0.73 |
| At 5 min | 7.2±2.6 | 7.9±3.1 | 0.23 |
| Other parameters | |||
| BPD | 14 (37.83%) | 10 (15.62%) | 0.012 |
| Asphyxia | 23 (62.16%) | 18 (28.12%) | 0.0008 |
| Received Surfactant | 19 (45.94%) | 17 (26.56%) | 0.012 |
| Length of Hospital stay (days) | 38.1±17.82 | 21.4±14.3 | 0.0001 |
| Mechanical Ventilation (days) | 13.42±8.74 | 5.89±3.56 | 0.0001 |
| Antibiotics Treatment (days) | 34.34±11.73 | 18.26±11.22 | 0.000001 |
| No of Antibiotics used | 5.43 ±1.34 | 3.13 ±1.22 | 0.000001 |
| Reintubation (n) | 11 (29.72%) | 8 (12.5%) | 0.33 |
| Deaths | 14 (37.8%) | 25 (39.1%) | 0.90 |
Among 37 VAP cases, new onset tachypnea (45.95%) and increased respiratory secretions (40.54%) were the most common clinical signs, followed by tachycardia (32.43%) and new onset apnea (24.32%), Both increased temperature and color changes in respiratory secretions (R. S) were observed in 16.22% of cases. Lab results frequently showed elevated C-reactive protein (70.27%), an I/T ratio>0.2 (62.16%), and thrombocytopenia (48.65%). Radiologically, new patches on X-rays (56.76%) were typical, with progressive patches in 29.73%. Microbiologically, tracheal aspirate cultures were positive in 75.68%, and blood cultures (CS) in 56.76% of VAP patients (fig. 1).
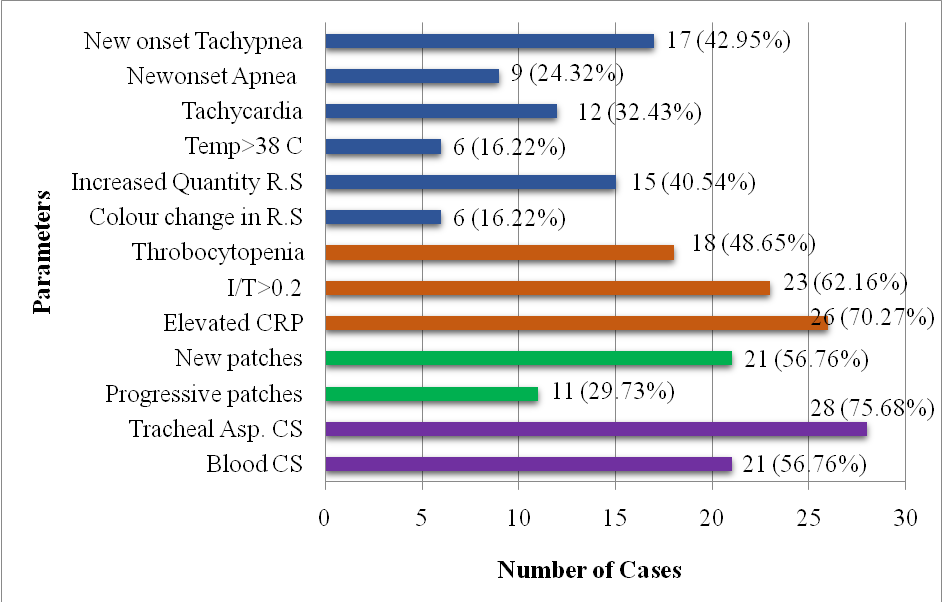
Fig. 1: Clinical, laboratory and radiological findings in VAP cases
From 37 VAP cases, 45 bacterial isolates were recovered, primarily from tracheal aspirates (24 isolates) and blood cultures (21 isolates). Gram-negative organisms predominated (68.9%), with Acinetobacter spp. (n=9), Klebsiella pneumoniae (n=9), and Klebsiella oxytoca (n=8) being the most common. Less frequent Gram-negative isolates included E. coli (n=2) and Pseudomonas (n=3). Gram-positive organisms made up 31.1% of isolates, mainly Staphylococcus species MRSA (Methicillin-Resistant Staphylococcus aureus) (n=6); and MSSA (Methicillin-Sensitive Staphylococcus aureus) (n=5) and Enterococcus spp. (n=3). This overall profile indicates a substantial burden of Gram-negative pathogens, particularly Acinetobacter and Klebsiellasp. in VAP among this neonatal cohort (fig. 2).
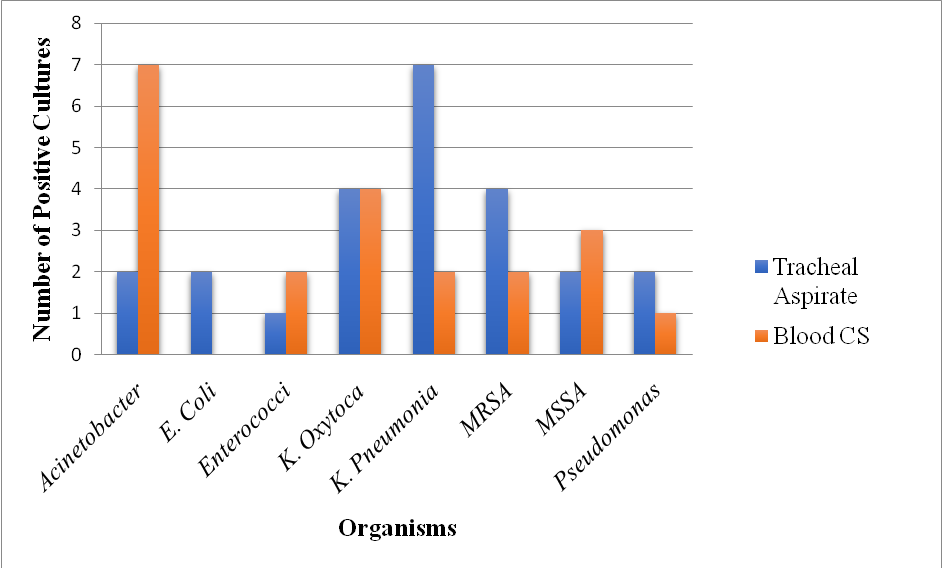
Fig. 2: Distribution of bacterial isolates from tracheal aspirate and blood cultures in neonates with VAP
Analysis of antibiotic susceptibility patterns for the 45 bacterial isolates revealed varied effectiveness. Among Gram-negative pathogens, Acinetobacter spp. (n=9) showed significant multi-drug resistance, with less than 50% susceptibility to Amikacin (44.4%), Amoxicillin+Clavulanic Acid (22.2%), Ceftriaxone (44.4%), Ciprofloxacin (33.3%), and Piperacillin+Tazobactam (44.4%). Resistance to Meropenem was lower, with 6 out of 9 isolates being susceptible.
K. pneumoniae (n=9) exhibited concerning resistance to third-generation cephalosporins, with 66.7% resistant to both Cefixime and Ceftriaxone, highly suggestive of ESBL production. It maintained better susceptibility to Meropenem (66.7% S) and Amikacin (66.7% S). K. oxytoca (n=8) displayed similar patterns to K. pneumoniae, with notable resistance to Cefixime (37.5% R) and Ceftriaxone (25% R, 12.5% I), but 62.5% susceptibility to Meropenem and 75% to Piperacillin+Tazobactam.
In contrast, the few E. coli (n=2) and Pseudomonas (n=3) isolates showed mixed susceptibility. For E. coli, 100% were susceptible to Meropenem, Piperacillin+Tazobactam, and Doxycycline, though one isolate was resistant to Ceftriaxone and Cotrimoxazole. For Pseudomonas, all isolates were resistant to Amikacin, and two out of three were resistant to Meropenem and Piperacillin+Tazobactam.
Regarding Gram-positive organisms, MRSA (n=6) showed good susceptibility to Linezolid (83.3%) but significant resistance to Amoxicillin+Clavulanic Acid (66.7% R), Azithromycin (66.7% R), Cefixime (50% R), and Ceftriaxone (83.3% R). MSSA (n=5) was highly susceptible to Linezolid, Ciprofloxacin, and Doxycycline (all 100% S), and showed good susceptibility to Amoxicillin+Clavulanic Acid, Cefixime, Ceftriaxone, and Levofloxacin (all 80% S). Enterococcus spp. (n=3) were 100% susceptible to Linezolid and Doxycycline. However, one out of three tested isolates was resistant to Amoxicillin+Clavulanic Acid and Ciprofloxacin, while no data was provided for Amikacin (table 2).
Table 2: Antibiotic susceptibility patterns of bacterial isolates from neonatal VAP cases
| Antibiotics (µg) | Organisms | |||||||
| Acinetobacter (n = 9) | E. coli (n = 2) | K. pneumoniae (n =9) | K. oxytoca (n = 8) | MRSA (n =6) | MSSA (n =5) | Pseudomonas (n = 3) | Enterococcus (n =3) | |
| SIR | SIR | SIR | SIR | SIR | SIR | SIR | SIR | |
| Amikacin (30) | 4 2 2 | 2-- | 6-3 | 4 2 2 | 4-2 | 3-2 | --3 | - |
| Amoxicillin+Clavulanic Acid (20/10) | 2-4 | --1 | 2-2 | 2-- | 2-4 | 4-1 |
|
|
| Azithromycin(15) | 1-4 | 1-4 | ||||||
| Cefixime(5) | 2-- | 3-6 | 1-3 | 1-3 | 4-- | 2-1 | ||
| Ceftriaxone(30) | 4-3 | 1-1 | 3-6 | 1 1 2 | 1-5 | 4-- | ||
| Ciprofloxacin(5) | 3 2 2 | 2-- | 4 1 2 | 3-1 | 2 1 3 | 5-- | 2-- | 2-1 |
| Cotrimoxazole (1.25/23.75) | 5 1 1 | 1-1 | 5 3- | 2-2 | 3 1 2 | 3-2 |
|
|
| Doxycycline(30) | 4-1 | 2-- | 6-- | 4-2 | 4-2 | 5-- | 1-2 | 3-- |
| Linezolid(30) | - | 1-- | 5-- | 5-- | 1-2 | 3-- | ||
| Levofloxacin(5) | 4-1 | 2-- | 4 3 1 | 4-2 | 4-2 | 4-1 | 1-2 | |
| Meropenem(10) | 6-1 | 2-- | 6 1 1 | 5 2 1 | 3-- | 2-- | 1-2 | |
| Nitrofurantoin(300) | 1-- | 3-- | 2-- | 1-- | 3-- | |||
| Piperacillin+Tazobactam(100/10) | 4-- | 2-- | 4-4 | 6-- | 1-- | 2-- | 1-- | |
| S-Susceptible; I-Intermediate; R – Resistant | ||||||||
DISCUSSION
The VAP incidence in this study was 41.11 per 1,000 ventilator days, it aligns with rates seen in developing countries (16.1 to 89 per 1,000 ventilator days), as reported by Mohamed et al. [2024] and Al-Kadri et al. [2024] [15]. Specifically, our findings are very similar to rates of 36.9 per 1,000 ventilator days in India (Mir et al., 2017) [16] and 35.06 in Bulgaria (Georgieva et al., 2022) [17], suggesting comparability with studies from similar resource settings.
This study highlights several critical risk factors for VAP in neonates, consistent with findings in other cohorts. Lower gestational age (p=0.028) and lower birth weight (p=0.035) were significantly linked to a higher incidence of VAP. This finding is consistent with a study by Singh et al. [2018] [20], which also observed that prematurity and low birth weight are significant risk factors for VAP due to immature immune systems and underdeveloped lungs in neonates. Similarly, Costa et al. [2020] [19] highlighted in their meta-analysis the strong correlation between lower gestational age and increased VAP risk.
Our study found bronchopulmonary dysplasia (BPD) (p=0.012) and a history of asphyxia (p=0.0008) were significantly more prevalent in neonates who developed VAP. These findings align with Singh et al. [2015] [21] and Van Der Pol et al. [2017] [22], who suggest that conditions prolonging ventilation compromise lung defenses, increasing VAP risk. Neonates with VAP also received surfactant therapy significantly more often (p=0.012), likely reflecting severe underlying respiratory distress, where invasive ventilation itself elevates VAP risk, as observed by Modi et al. [2017] [23].
Neonates who developed VAP had significantly longer hospital stays (averaging 38.1±17.82 d compared to 21.4±14.3 d for those without VAP; p=0.0001). They also required mechanical ventilation for a considerably longer duration (13.42±8.74 d versus 5.89±3.56 d; p=0001). These observations are consistent with research by El-Masri et al. [2020] [24], which similarly found that VAP leads to extended hospitalization and increased healthcare expenses.
Additionally, antibiotic use was much greater in the VAP group. These neonates received antibiotic therapy for significantly longer periods (34.34±11.73 d compared to 18.26±11.22 d in the non-VAP group; p=0.000001) and were given a higher average number of different antibiotics (5.43±1.34 versus 3.13±1.22; p=0.000001). While increased antibiotic use is expected for VAP, this trend, as noted by Kumar et al. [2016] [25], raises concerns about the potential for antibiotic resistance and adverse effects in this vulnerable patient population.
In this study, new onset tachypnea (45.95%) and increased respiratory secretions (40.54%) were the most common clinical VAP symptoms, consistent with findings from Georgieva et al. [2022] [17] and Al-Kadri et al. [2025] [15]. Laboratory tests frequently showed elevated C-reactive protein (70.27%) and an I/T ratio>0.2 (62.16%), indicating systemic inflammation, as supported by Al-Kadri et al. [2025] [15]. While challenging to interpret in neonates, chest X-ray changes like new infiltrates were crucial for diagnosis, a point highlighted by Georgieva et al. [2022] [17] and Li et al. [2022] [26].
Tracheal aspirate cultures had a high yield (75.68%), underscoring their importance for pathogen identification and targeted therapy (Tuteja et al., 2022 [18]; Dey et al., 2021) [27]. The significant rate of positive blood cultures (56.76%) suggests associated bacteremia, emphasizing the need for both local and systemic samples to guide antimicrobial management (Tuteja et al., 2022) [18].
Analysis of antibiotic susceptibility revealed concerning Gram-negative resistance. Acinetobacter spp. showed broad multi-drug resistance to Amikacin, Amoxicillin+Clavulanic Acid, Ceftriaxone, Ciprofloxacin, and Piperacillin+Tazobactam, with Meropenem being relatively more effective. This high resistance in Acinetobacter is a recognized challenge in neonatal infections, limiting options and demanding potent empirical therapies, as Al-Kadri et al. [2025] [15] also reinforce. Similarly, K. pneumoniae and K. oxytoca exhibited substantial resistance to Cefixime and Ceftriaxone, strongly suggesting high ESBL production. Their good susceptibility to Meropenem highlights an increasing reliance on carbapenems for ESBL-producing strains, a trend noted by Mir et al. [2017] [16] in other NICU settings. Our Pseudomonas isolates showed alarming resistance, with 100% resistant to Amikacin and 66.7% resistant to Meropenem and Piperacillin+Tazobactam. This high-level resistance, common in Pseudomonas aeruginosa, complicates treatment and necessitates rapid susceptibility testing for effective neonatal VAP management [28, 29].
Among Gram-positive isolates, MRSA (n=6) exhibited good susceptibility to Linezolid, indicating its continued effectiveness against this resistant pathogen, consistent with general literature on Linezolid's efficacy. MSSA (n=5) showed broad susceptibility to most tested antibiotics, reflecting its generally less resistant nature. Enterococcus spp. (n=3) were entirely susceptible to Linezolid and Doxycycline but notably resistant to Amoxicillin+Clavulanic Acid, Ciprofloxacin, and Amikacin. The presence of MRSA, even in smaller numbers, underscores the ongoing need for vigilance and infection control, as discussed by Shah et al. [2023] [30]. The diverse susceptibility patterns observed across both Gram-negative and Gram-positive pathogens, particularly the multidrug resistance in Acinetobacter and ESBL-producing Klebsiella, strongly emphasizes the critical importance of culture-guided antibiotic therapy in neonatal VAP management. This approach optimizes patient outcomes and mitigates further antimicrobial resistance development, a strategy supported by Smith et al. [2024] [29].
A recent study by Biswas N et al. [2025] [31] on pediatric UTIs demonstrated a similar pattern of MDR, with Escherichia coli, Klebsiella spp., and MRSA emerging as predominant pathogens. The high resistance to Cephalosporins and Fluoroquinolones in both infections highlights the urgent need for continuous surveillance and antibiotic stewardship to guide effective empirical therapy.
LIMITATIONS
The relatively small sample size and its single-center origin mean the results may not be broadly applicable to all neonatal populations or different healthcare settings. Diagnosing VAP in neonates is inherently challenging due to the subjective nature of clinical, laboratory, and radiological criteria in this vulnerable group. Furthermore, our analysis of antibiotic susceptibility was confined to a specific set of agents, and we didn't explore broader resistance patterns or the underlying genetic mechanisms of resistance. Despite higher morbidity and antibiotic use in the VAP group, we found no significant difference in mortality rates compared to non-VAP neonates. However, without detailing the specific causes of death in either group, it's difficult to fully interpret this finding.
CONCLUSION
This study found neonatal VAP was linked to prematurity, low birth weight, bronchopulmonary dysplasia, and asphyxia. VAP led to longer hospital stays and ventilation durations, plus higher antibiotic use. We identified predominantly Gram-negative bacteria, like Acinetobacter and Klebsiella, with significant drug resistance. Despite the increased morbidity and mortality in this small, single-center study. Further research with larger cohorts is needed to confirm these findings and guide VAP management.
ACKNOWLEDGMENT
The authors expressed their appreciation to the Silchar Medical College and Hospital's Department of Microbiology for their significant contribution.
FUNDING
This study was entirely self-funded.
AUTHORS CONTRIBUTIONS
Dr. Veeranna Shivanand Chikkamath and Dr. M. Swathi Patil were involved in the conception and design of the study, patient recruitment, data collection, analysis, interpretation of results, and drafting of the manuscript. Dr. Nilanjan Biswas assisted with statistical support and contributed to the results section. Dr. Pranab Das performed literature review and provided inputs during revision of the discussion. Dr. Sujit Nath Choudhury offered overall supervision, critically reviewed the manuscript for important intellectual content. All authors have read and approved the final manuscript and agree to be accountable for all aspects of the work.
CONFLICT OF INTERESTS
There is no conflict of interests
REFERENCES
Shane AL, Sanchez PJ, Stoll BJ. Neonatal sepsis. Lancet. 2017 Nov 18;390(10104):1770-80. doi: 10.1016/S0140-6736(17)31002-4, PMID 28434651.
Azab SF, El-Sayed ZM, El-Sayed ZM, Abdelrahman AM, Sherief LM, Osman NH. Impact of a ventilator-associated pneumonia prevention bundle on neonatal intensive care unit patients. J Infect Public Health. 2015 May-Jun;8(3):268-74. doi: 10.1016/j.jiph.2014.09.001.
Rosenthal VD, Al-Abdely HM, El-Kholy AA, AlKhawaja SA, Leblebicioglu H, Mehta Y. International nosocomial infection control consortium report data summary of 50 countries for 2010-2015: device-associated module. Am J Infect Control. 2016;44(12):1495-504. doi: 10.1016/j.ajic.2016.08.007, PMID 27742143.
Zhou Q, Lee SK, Jiang SY, Chen C, Kamaluddeen M, Hu XJ. Efficacy of an infection control program in reducing ventilator-associated pneumonia in a Chinese neonatal intensive care unit. Am J Infect Control. 2013;41(11):1059-64. doi: 10.1016/j.ajic.2013.06.007, PMID 24041863.
Tripathi S, Malik GK. Neonatal ventilator-associated pneumonia: an emerging problem. Indian Pediatr. 2010 Oct;47(10):867-71. doi: 10.1007/s12098-014-1561-x.
Sharma R, Goyal A, Ghosh S, Kapil A, Ray P, Taneja N. Microbiological spectrum and antibiotic sensitivity pattern in neonatal intensive care unit. Indian J Pediatr. 2012 Apr;79(4):490-3. doi: 10.1007/s12098-011-0594-6.
Erichsen Andersson A, Petzold M, Bergh I, Karlsson J, Eriksson BI, Nilsson K. Comparison between mixed and laminar airflow systems in operating rooms and the influence of human factors: experiences from a Swedish orthopedic center. Am J Infect Control. 2014;42(6):665-9. doi: 10.1016/j.ajic.2014.02.001, PMID 24713595.
Dudeck MA, Horan TC, Peterson KD, Allen Bridson K, Morrell GC, Pollock DA. National Healthcare Safety Network (NHSN) report data summary for 2009, device-associated module. Am J Infect Control. 2011;39(5):349-67. doi: 10.1016/j.ajic.2011.04.011, PMID 21774120.
Hogue AN, Temple Cooper ME, Lagzdins M, Worley S, Scwersenski J, Floyd R. Effects of in-utero exposure to selective serotonin reuptake inhibitors and venlafaxine on term and preterm infants. J Neonatal Perinat Med. 2017;10(4):371-80. doi: 10.3233/NPM-16133, PMID 29286926.
Centers for Disease Control and Prevention. Ventilator-associated event (VAE) surveillance protocol. In: CDC NHSN Patient Safety Manual, Chapter 10. Atlanta (GA): CDC; 2025 Jan. Available from: https://www.cdc.gov/nhsn/pdfs/pscmanual/10-vae_final.pdf.
Kalanuria AA, Ziai W, Mirski M. Ventilator-associated pneumonia in the ICU. Crit Care. 2014;18(2):208. doi: 10.1186/cc13775, PMID 25029020.
German National Nosocomial Infection Surveillance System (KISS). NEO-KISS module for very low birth weight infants. National Reference Center for Surveillance of Nosocomial Infections (NRZ) Website; 2020. Available from: https://www.nrz-hygiene.de/en/KISS-Modul/KISS/NEO.
Gupta A, Nayak A, Gopinath KG, Rathod DA, Krishnan R, Kumar A. Clinical outcomes of ventilator-associated pneumonia in mechanically ventilated neonates. Indian Pediatr. 2013 Oct;50(10):899-903. doi: 10.1007/s12098-013-1147-x.
Sulek K, Mc Donald LC, Balchunas L, Jarvis WR, Kurmasheva RT, Khudyakova ND. Incidence risk factors and outcomes of ventilator-associated pneumonia in neonates. J Perinatol. 2005 Aug;25(8):533-9.
Al-Kadri H, Al-Anazi A, Al-Burayh N, Al-Kadi M. The association between neonatal mortality and ventilator-associated pneumonia. Alexandria Journal of Medicine; 2024. Available from: https://doaj.org/article/6e0193e2ae4f46a09b2cc08cd979d89a.
Mir Z, Ali I, Qureshi O, Wani G. Risk factors pathogen profile and outcome of ventilator-associated pneumonia in a neonatal intensive care unit. Int J Contemp Pediatr. 2015;2(1):17. doi: 10.5455/2349-3291.ijcp20150204.
Rangelova VR, Raycheva RD, Kevorkyan AK, Krasteva MB, Kalchev YI. Ventilator-associated pneumonia in neonates admitted to a tertiary care NICU in Bulgaria. Front Pediatr. 2022;10:909217. doi: 10.3389/fped.2022.909217, PMID 35837238.
Tuteja A, Pournami F, Nandakumar A, Prabhakar J, Jain N. Endotracheal aspirate and ventilator-associated pneumonia in neonates: revisiting an age-old debate. Indian J Pediatr. 2022 Dec;89(12):1202-8. doi: 10.1007/s12098-022-04142-y, PMID 35503590.
Ghobrial EE, Farag YM, Abdul-Aziz DM, Omar MA. Assessment of epicardial adipose tissue thickness in children with familial mediterranean fever. J Trop Pediatr. 2020;66(2):121-8. doi: 10.1093/tropej/fmz040, PMID 31257436.
Kulkarni S, Gilbert C, Zuurmond M, Agashe S, Deshpande M. Blinding retinopathy of prematurity in Western India: characteristics of children reasons for late presentation and impact on families. Indian Pediatr. 2018;55(8):665-70. doi: 10.1007/s13312-018-1355-8, PMID 30218512.
Singh DP, Yadav DK, Sharma M, Singh S. Risk factors and outcome of ventilator-associated pneumonia in a tertiary care neonatal intensive care unit. J Clin Neonatol. 2015;4(4):214-8. doi: 10.4103/2249-4847.170991.
Vu EL, Rusin CG, Penny DJ, Kibler KK, Easley RB, Smith B. A novel electrocardiogram algorithm utilizing ST-segment instability for detection of cardiopulmonary arrest in single ventricle physiology: a retrospective study. Pediatr Crit Care Med. 2017;18(1):44-53. doi: 10.1097/PCC.0000000000000980, PMID 27755397.
Modi KB, Patel SM, Patel KM, Patel J, Patel K. Risk factors for ventilator-associated pneumonia in neonates: a prospective observational study. J Med Sci Clin Res. 2017;5(8):27885-91.
Moreno CS, Rogero SO, Ikeda TI, Cruz AS, Rogero JR. Resveratrol and radiation biological effects. Int J Nutrology. 2020;5(1):28-33. doi: 10.1055/s-0040-1701425.
Samayam P, Ranganathan PK, Balasundaram R. Study of weight patterns in exclusively breast fed neonates does the route of delivery have an impact? J Clin Diagn Res. 2016;10(1):SC01-3. doi: 10.7860/JCDR/2016/17889.7025, PMID 26894138.
Jiang P, Wei J. The application of pulmonary ultrasound in neonatal ventilator-associated pneumonia. Front Pediatr. 2022;10:882056. doi: 10.3389/fped.2022.882056, PMID 35844761.
Dey S, Sharma D, Kohli M, Mishra A. Utility of quantitative tracheal aspirate cultures in predicting ventilator-associated pneumonia in neonates. Indian J Pediatr. 2021;88(3):250-4. doi: 10.1007/s12098-020-03441-4.
Morales Lopez J, Ortiz Cartagena C, Alonso Bello CD, Gonzalez Serrano D, Serna Gallego A, Cortes Lopez HJ. Antibiotic-Resistant Pseudomonas aeruginosa: Current Challenges and emerging alternative therapies. Antibiotics (Basel). 2024;13(4):913. doi: 10.3390/antibiotics13040913.
Rallis D, Giapros V, Serbis A, Kosmeri C, Baltogianni M. Fighting antimicrobial resistance in neonatal intensive care units: rational use of antibiotics in neonatal sepsis. Antibiotics (Basel). 2023;12(3):508. doi: 10.3390/antibiotics12030508, PMID 36978375.
S Karger Gmb H, Freiburg. PharmaNews. Kompass Pneumol. 2022;10(6):322-4. doi: 10.1159/000528268.
Biswas N, Roy D, Borah M, Chanda DD, Das P, Datta K. Antibiotic resistance pattern in isolated pathogens of urinary tract infections in pediatric patients at a Tertiary Care Hospital. Int J Curr Pharm Sci. 2025 Mar 15;17(2):79-85. doi: 10.22159/ijcpr.2025v17i2.6083.