
Int J Curr Pharm Res, Vol 18, Issue 1, 47-52Original Article
A RANDOMIZED, DOUBLE-BLIND COMPARISON OF INTRATHECAL NALBUPHINE AND BUPRENORPHINE AS ADJUVANTS TO 1% 2-CHLOROPROCAINE FOR SPINAL ANESTHESIA IN AMBULATORY PERIANAL SURGERY
BEEMAGOUNI GNANESHWAR GOUD, SHOW REDDY EDARA, IAILNENI VENKAT SAI NIKITHA, LAKKAM VAMSEE KIRAN*
Department of anesthesiology, RVM institute of medical sciences and research center, Siddipet, Talangana, India
*Corresponding author: Lakkam Vamsee Kiran; *Email: vamsee.lakkam@gmail.com
Received: 08 Oct 2025, Revised and Accepted: 27 Nov 2025
ABSTRACT
Objective: Perianal surgeries in ambulatory settings require anesthetic agents with rapid onset, predictable duration, and minimal postoperative morbidity. This study compared the efficacy of Nalbuphine and Buprenorphine as intrathecal adjuvants to 1% 2-Chloroprocaine in spinal anesthesia for day-care perianal surgeries.
Methods: In this prospective, randomized, double-blind study, 68 ASA I/II patients aged 18–60 years undergoing elective perianal surgeries were randomized into two groups (n=34 each). Group B received 40 mg 1% 2-Chloroprocaine+Buprenorphine 60 μg; Group N received 40 mg 1% 2-Chloroprocaine+Nalbuphine 0.4 mg. Outcomes included onset and duration of sensory and motor blocks, regression times, postoperative analgesia, hemodynamic stability, and adverse effects.
Results: Sensory block onset was faster in Group B (2.6±1.28 min) compared to Group N (3.71±1.0 min, p<0.001). Motor block onset was also faster with Buprenorphine (2.85±1.23 min vs 3.71±1.0 min, p<0.01). Nalbuphine prolonged regression to S2 (71.29±8.66 min vs 66.82±9.27 min, p<0.001) and duration of motor block (64.41±7.86 min vs 59.65±9.74 min, p<0.05). Time to rescue analgesia was longer with Nalbuphine. Hemodynamics remained stable; adverse effects were minimal.
Conclusion: Intrathecal Buprenorphine and Nalbuphine are safe and effective adjuvants to 1% 2-Chloroprocaine for spinal anesthesia in day-care perianal surgeries. Buprenorphine ensures faster onset of block, while Nalbuphine provides superior and longer-lasting postoperative analgesia. Both support early ambulation and discharge, making them valuable options in ambulatory anesthesia.
Keywords: Spinal anesthesia, 2-Chloroprocaine, Nalbuphine, Buprenorphine, Perianal surgery, Day-care anesthesia
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i1.8011 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
The landscape of surgical practice has undergone a paradigm shift over the past several decades, with a pronounced migration of procedures from traditional inpatient settings to ambulatory or day-care surgical units [1]. This transition, driven by advances in surgical techniques, anesthetic management, and economic pressures, offers substantial benefits, including reduced healthcare costs, lower risks of nosocomial infections, and improved patient satisfaction through quicker return to a familiar home environment [2]. Perianal surgeries, encompassing procedures for conditions such as hemorrhoids, anal fissures, and fistulae, are particularly well-suited for this model. The success of an ambulatory surgical program, however, is critically dependent on an anesthetic technique that not only provides excellent intraoperative conditions but also ensures a rapid, predictable, and complication-free recovery, culminating in a safe and timely discharge [2].
Spinal anesthesia, or subarachnoid block (SAB), has long been established as a premier regional anesthetic technique for surgeries involving the lower abdomen, perineum, and lower extremities. It offers numerous advantages over general anesthesia, including a rapid and reliable onset, a dense and profound sensory and motor blockade, reduced intraoperative blood loss, a lower incidence of postoperative nausea and vomiting (PONV), and the maintenance of a conscious, spontaneously breathing patient [1]. For ambulatory perianal surgery, these characteristics make it an exceptionally attractive option. However, the traditional use of long-acting local anesthetics, such as bupivacaine, for spinal anesthesia often leads to prolonged motor blockade and delayed autonomic recovery (e. g., micturition), which can significantly delay patient discharge and undermine the efficiency of a day-care unit [3, 4].
This challenge has led to a resurgence of interest in preservative-free 2-Chloroprocaine for spinal anesthesia [5]. As an amino-ester local anesthetic, 2-Chloroprocaine is characterized by an extremely rapid metabolism by plasma pseudo cholinesterase, resulting in a very short duration of action. This pharmacokinetic profile is highly desirable in the ambulatory setting, as it facilitates a swift regression of both motor and sensory blockade, allowing for early ambulation and fulfillment of discharge criteria [6]. Numerous studies have affirmed its safety and efficacy, establishing it as a first-line agent for ultra-short surgical procedures [7, 8]. The principal limitation of spinal 2-Chloroprocaine, however, is a direct consequence of its primary advantage: its short duration of action results in a correspondingly brief period of postoperative analgesia. As the anesthetic block recedes, patients can experience a rapid onset of significant pain, which can compromise comfort and delay recovery [9, 10]. This creates a clear clinical need for adjunctive agents that can selectively prolong the duration of analgesia without extending the duration of motor blockade or delaying other aspects of recovery.
The co-administration of intrathecal opioids with local anesthetics is a well-established strategy to enhance the quality and extend the duration of spinal analgesia [11]. By acting directly on opioid receptors within the dorsal horn of the spinal cord, these agents modulate nociceptive signaling, providing synergistic pain relief. The selection of the ideal opioid adjuvant, however, requires a careful balance between analgesic efficacy and the potential for adverse effects. Two agents with distinct pharmacological profiles, Buprenorphine and Nalbuphine, have emerged as promising candidates for this role [12-14].
Buprenorphine is a semi-synthetic, highly lipophilic opioid characterized by a distinctive receptor binding profile. It functions as a partial agonist at the mu-opioid receptor while simultaneously acting as an antagonist at the kappa-opioid receptor. Its high affinity for and slow dissociation from the mu-receptor are thought to contribute to its potent and prolonged analgesic effects, often lasting many hours beyond the resolution of the local anesthetic block. This makes it an attractive option for extending postoperative pain relief. However, its mu-agonist activity also carries the risk of classic mu-mediated side effects, including respiratory depression, pruritus, and, notably, urinary retention [12, 15].
In contrast, Nalbuphine is a synthetic opioid agonist-antagonist. It exerts its analgesic effects primarily through agonism at the kappa-opioid receptor, while simultaneously acting as an antagonist at the mu-opioid receptor. This unique mechanism is advantageous as it provides effective analgesia with a reported ceiling effect on respiratory depression, a feature that enhances its safety profile. Furthermore, its antagonism at the mu-receptor may mitigate other mu-mediated side effects, potentially leading to a more favorable recovery profile compared to traditional mu-agonists [13, 14, 16].
While both Buprenorphine and Nalbuphine have been studied as adjuvants to longer-acting local anesthetics like bupivacaine, their comparative performance when paired with the ultra-short-acting 2-Chloroprocaine remains poorly defined, particularly within the specific context of ambulatory perianal surgery. This gap in the literature establishes a state of clinical equipoise: it is unknown whether the prolonged, potent analgesia of Buprenorphine outweighs its potential for delayed recovery, or if the balanced, safer profile of Nalbuphine provides a more optimal overall experience for the day-care surgical patient. Therefore, this study was undertaken to conduct a direct, randomized, double-blind comparison of intrathecal Nalbuphine versus Buprenorphine as adjuvants to 1% 2-Chloroprocaine, with the aim of elucidating the optimal strategy for enhancing spinal anesthesia in ambulatory perianal surgery.
MATERIALS AND METHODS
Study design and ethical approval
This investigation was conducted as a prospective, randomized, double-blinded, comparative clinical study. The research protocol was designed to rigorously compare the efficacy and safety of two distinct intrathecal opioid adjuvants. All study activities were carried out over a period of one year (April 2023-March 2024) within the operation theatre complex and the postoperative recovery ward of the RVM Institute of Medical Sciences and Research Centre in Laxmakkapally, Telangana, India. The study protocol, including the informed consent documents and patient information sheets, was submitted to and approved by the Institutional Ethics Committee prior to the enrollment of the first patient. The study was conducted in strict adherence to the ethical principles outlined in the Declaration of Helsinki and the guidelines for Good Clinical Practice. Written, informed consent was obtained from every participant after a thorough explanation of the study's purpose, procedures, potential risks, and benefits, ensuring their voluntary participation.
Patient population
The study population consisted of adult patients scheduled for elective, short-duration perianal surgeries under subarachnoid block. A total of 68 patients were enrolled in the study. The inclusion criteria were as follows: (1) age between 18 and 60 years; (2) physical status classified as Grade I or II according to the American Society of Anesthesiologists (ASA) classification system; (3) scheduled for an elective perianal surgical procedure (e. g., hemorrhoidectomy, fistulectomy, fissurectomy); and (4) provision of voluntary, written informed consent.
Patients were excluded from participation if any of the following criteria were met: (1) patient refusal; (2) a known history of allergy or hypersensitivity to any of the study drugs (2-Chloroprocaine, Nalbuphine, Buprenorphine, or other amide/ester local anesthetics or opioids); (3) presence of a coagulopathy or current use of anticoagulant medications; (4) evidence of a localized infection at the proposed site of lumbar puncture; (5) presence of severe systemic illness, including significant cardiac, renal, or hepatic disease (ASA Grade III or higher); (6) pregnancy or lactation; (7) pre-existing neurological or neuromuscular disorders; or (8) spinal deformities or a history of previous spinal surgery that would preclude the safe performance of a subarachnoid block.
Randomization and blinding
Upon meeting the eligibility criteria and providing consent, the 68 participants were randomly allocated into one of two study groups of equal size (n=34). The randomization sequence was generated using a computer-generated random number table to ensure unpredictable allocation. This study employed a double-blind methodology. To maintain blinding, the study drug solutions were prepared and drawn into identical, unlabeled syringes by an independent anesthetist who was not involved in the subsequent anesthetic administration, patient monitoring, or data collection. This process ensured that both the patient and the primary anesthesiologist responsible for patient care and data assessment remained unaware of the specific adjuvant administered, thereby eliminating potential performance and detection bias.
Intervention
All patients received a standardized volume and dose of the primary local anesthetic. The specific adjuvant constituted the only difference between the two groups:
Group N (Nalbuphine group): Patients in this group received an intrathecal injection of a total volume of 4.2 ml, consisting of 4 ml of 1% 2-Chloroprocaine (40 mg) combined with 0.2 ml of Nalbuphine (0.4 mg).
Group B (Buprenorphine group): Patients in this group received an intrathecal injection of a total volume of 4.2 ml, consisting of 4 ml of 1% 2-Chloroprocaine (40 mg) combined with 0.2 ml of Buprenorphine (0.06 mg).
Anesthetic procedure
All patients underwent a standardized pre-anesthetic evaluation. Prior to surgery, the premedication included an oral tablet of pantoprazole 40 mg, an ondansetron tablet of 4 mg, and a 0.5 mg alprazolam tablet given the night before. Patients were instructed to remain nil per oral (NPO) from midnight. Upon arrival in the operating room, standard non-invasive monitoring was established, including electrocardiography (ECG), non-invasive blood pressure (NIBP), and peripheral oxygen saturation (SpO2). An intravenous line was secured with an 18-gauge or 20-gauge cannula, and an infusion of Ringer's lactate solution was initiated as a preload.
The subarachnoid block was performed with the patient in the sitting position under strict aseptic precautions. The lumbar region was prepared with an antiseptic solution and draped. After local infiltration of the skin with lidocaine, a 25-gauge Quincke spinal needle was introduced into the L3-L4 or L4-L5 intervertebral space using a midline approach. The positioning of the needle tip within the subarachnoid space was validated by the free flow of cerebrospinal fluid (CSF), then the pre-prepared investigational solution was administered intrathecally. Immediately after the completion of the injection, the patient was gently assisted into the supine position.
Outcome measures and data collection
A standardized protocol was used for the collection of all outcome data. The primary objectives were to evaluate the onset and duration of the sensory and motor blocks. The secondary objectives were to assess the duration of postoperative analgesia, recovery milestones, hemodynamic stability, and the incidence of adverse effects.
Sensory block assessment
The onset of sensory block was defined as the time from the completion of the intrathecal injection to the loss of sensation to a pinprick at the L1 dermatome, assessed every 30 seconds. The time to reach the peak sensory block level and the level itself were recorded, with assessments performed bilaterally every 2 min. The duration of sensory block was evaluated by recording the time for two-segment regression from the peak level and the total time for regression to the S2 dermatome, with assessments at 10 min intervals.
Motor block assessment
The degree of motor blockade was assessed using the Modified Bromage Scale (0 = no motor block, 3 = complete motor block). The onset of motor block was defined as the time to reach a Bromage Score of 1, assessed every 2 min. The time to reach peak motor block (Bromage Score 3) and the total duration of motor block (time until return to Bromage Score 0) were recorded.
Postoperative recovery assessment
Postoperative pain was assessed using a 10-point Visual Analogue Scale (VAS), where 0 represents no pain and 10 represents the worst imaginable pain. The duration of effective analgesia was defined as the time from the spinal injection until the patient first reported a VAS score of 4 or greater, at which point rescue analgesia was administered. Other key recovery milestones recorded were the time to first spontaneous void and the time to achieve unassisted ambulation.
Hemodynamic monitoring and safety
Heart rate (HR), systolic blood pressure (SBP), diastolic blood pressure (DBP), mean arterial pressure (MAP), and SpO2 were recorded at baseline, every 2 min for the first 15 min, every 5 min up to 30 min, and then every 15 min thereafter. Hypotension was defined as a decrease in SBP of more than 20% from baseline and was treated with intravenous boluses of me phentermine 6 mg. Bradycardia was defined as a heart rate below 60 beats per minute and was treated with intravenous atropine 0.6 mg. The incidence of any other adverse events, such as nausea, vomiting, shivering, or pruritus, was also recorded.
Statistical analysis
The sample size was calculated a priori based on previously published data, determining that a minimum of 34 subjects per group was required to detect a clinically significant difference with a power of 80% and a significance level (α) of 0.05. The data were systematically input into a spreadsheet and subsequently analyzed employing SPSS software (version 16) alongside Microsoft Excel. Continuous variables were articulated as mean±standard deviation (SD). The means of continuous variables between the two study groups were compared using a one-way analysis of variance (ANOVA). A p-value of less than 0.05 was considered to be statistically significant for all comparisons.
RESULTS
Patient flow and baseline characteristics
All 68 patients who were enrolled and randomized for the study successfully completed the protocol. No participants were removed or excluded from the final analysis. The process of randomization resulted in two study groups that were well-matched and statistically comparable with respect to all demographic and baseline clinical variables. As can be observed from the comprehensive data presented within table 1, it is evident that there were no significant differences between the Buprenorphine group, (Group B), and the Nalbuphine group, (Group N), when it comes to various demographic and physical characteristics such as the mean age of the participants, the distribution of gender among the participants, mean weight recorded, mean height measured, and mean Body Mass Index (BMI) of the individuals involved in these groups. Furthermore, the mean duration of the surgical procedures was also similar between the two groups, ensuring that this variable did not act as a confounder in the assessment of anesthetic duration or recovery. This baseline homogeneity confirms the internal validity of the study, allowing for the attribution of any observed differences in outcomes to the specific intrathecal adjuvant used.
Primary outcomes: neuraxial block characteristics
The comparative analysis of the primary outcome measures revealed statistically significant differences in the onset and duration characteristics of the neuraxial blockade between the two groups, as summarized in table 2.
Table 1: Demographic and baseline clinical characteristics
| Parameter | Group B (Buprenorphine) (n=34) | Group N (Nalbuphine) (n=34) | p-value |
| Age (years) | 41.26±11.08 | 42.88±10.48 | 0.397 |
| Sex (Male/Female) | 15/19 | 20/14 | - |
| Weight (kg) | 65.59±9.50 | 66.06±8.44 | 0.415 |
| Height (cm) | 165.00±11.56 | 164.18±8.85 | 0.371 |
| BMI (kg/m2) | 24.52±5.34 | 24.78±4.55 | 0.412 |
| Duration of Surgery (min) | 24.78±4.55 | 24.52±5.34 | 0.097 |
Data are presented as mean±SD or number of patients. A p-value<0.05 was considered significant.
Table 2: Comparison of intraoperative sensory and motor block characteristics
| Parameter | Group B (Buprenorphine) (n=34) | Group N (Nalbuphine) (n=34) | p-value |
| Onset of Sensory Block (min) | 2.6±1.28 | 3.71±1.00 | 0.000311 |
| Time to Peak Sensory Block (min) | 14.38±3.11 | 13.79±2.47 | 0.391401 |
| Time for Two-Segment Regression (min) | 42.09±2.77 | 35.38±7.36 | <0.00001 |
| Time for Regression to S2 (min) | 66.82±9.27 | 71.29±8.66 | <0.00001 |
| Onset of Motor Block (min) | 2.85±1.23 | 3.71±1.00 | 0.001302 |
| Time to Peak Motor Block (min) | 6.21±1.77 | 6.18±1.69 | 0.471354 |
| Duration of Motor Block (min) | 59.65±9.74 | 64.41±7.86 | 0.014895 |
Data are presented as mean±SD. Statistically significant p-values (<0.05) are in bold.
Sensory block
Patients who received Buprenorphine experienced a significantly faster onset of sensory blockade. The mean time to loss of pinprick sensation at the L1 dermatome in Group B was 2.6±1.28 min, compared to 3.71±1.00 min in Group N (p=0.000311). However, the time required to reach the peak level of sensory block was comparable between the two groups, with no statistically significant difference observed (p=0.391401). The duration of the sensory block showed a mixed profile. The Buprenorphine group experienced a significantly longer time for the block to regress by two dermatomal segments (42.09±2.77 min) compared to the Nalbuphine group (35.38±7.36 min), with a p-value of less than 0.00001. Conversely, the total duration of sensory block, measured as the time for regression to the S2 dermatome, was significantly longer in the Nalbuphine group (71.29±8.66 min) compared to the Buprenorphine group (66.82±9.27 min; p<0.00001).
Motor block
The pattern observed with the onset of motor blockade mirrored that of the sensory block. The Buprenorphine group demonstrated a significantly faster onset of motor weakness, with a mean time to reach a Modified Bromage Score of 1 of 2.85±1.23 min, compared to 3.71±1.00 min in the Nalbuphine group (p=0.001302). Similar to the sensory block, the time taken to achieve peak motor blockade (Bromage Score 3) was not significantly different between the two groups (p=0.471354). The total duration of motor blockade, however, was found to be significantly longer in the Nalbuphine group. The mean time for complete motor function to return (Bromage Score 0) was 64.41±7.86 min in Group N, compared to 59.65±9.74 min in Group B (p=0.014895).
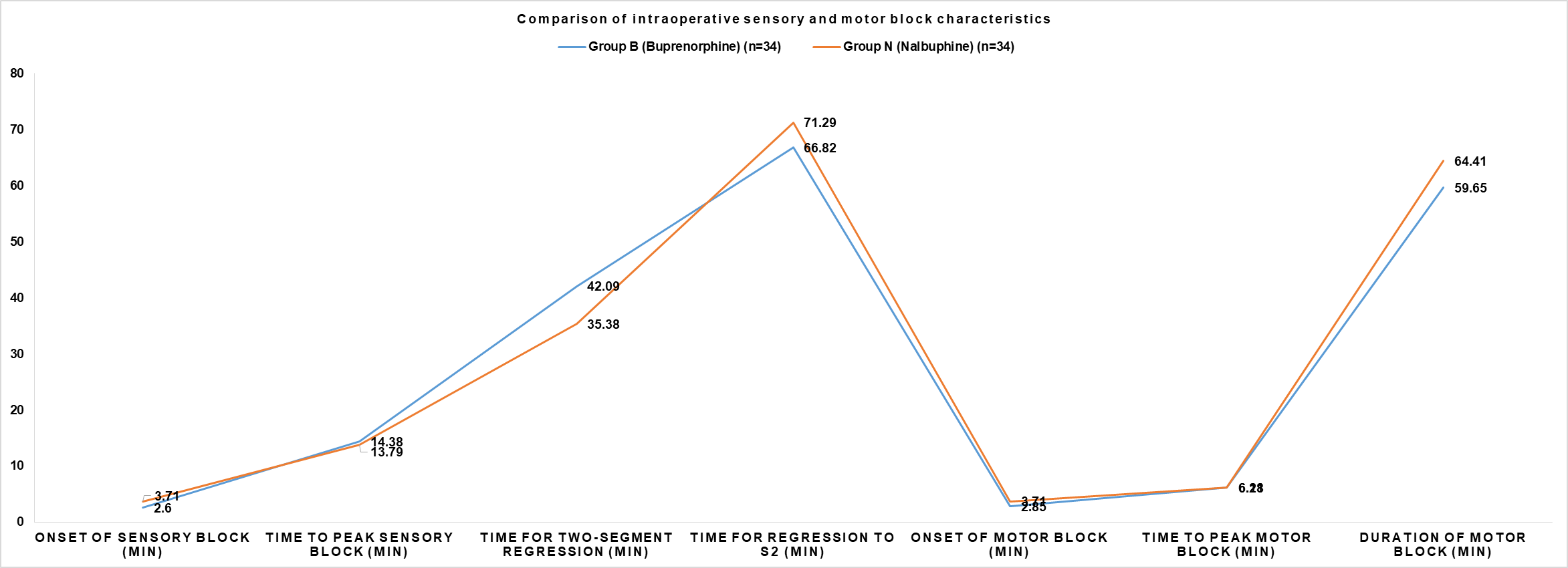
Fig. 1: Comparison of intraoperative sensory and motor block characteristics between buprenorphine and nalbuphine groups
Secondary outcomes: postoperative recovery and analgesia
The analysis of secondary outcomes related to postoperative recovery and analgesia revealed clinically important differences between the two adjuvants, particularly concerning autonomic function. These findings are presented in table 3.
Table 3: Comparison of postoperative recovery and analgesia parameters
| Parameter | Group B (Buprenorphine) (n=34) | Group N (Nalbuphine) (n=34) | p-value |
| Time for First Rescue Analgesia (min) | 206.00±24.48 | 213.00±26.33 | 0.24254 |
| Time for First Void (min) | 378.76±46.56 | 320.76±46.56 | <0.00001 |
| Time for Unassisted Ambulation (min) | 299.00±30.89 | 312.7±35.57 | 0.09454 |
Data are presented as mean±SD. Statistically significant p-values (<0.05) are in bold.
The duration of effective postoperative analgesia, defined as the time to the first request for rescue medication (VAS score ≥ 4), was comparable between the two groups. Patients in the Nalbuphine group had a mean analgesic duration of 213.00±26.33 min, while those in the Buprenorphine group had a mean duration of 206.00±24.48 min. This difference was not statistically significant (p=0.24254).
The most striking difference in recovery profiles was observed in the time to first spontaneous micturition. Patients in the Buprenorphine group experienced a significantly prolonged delay in the return of normal bladder function. The mean time to first void in Group B was 378.76±46.56 min. In contrast, patients in the Nalbuphine group were able to void significantly earlier, with a mean time of 320.76±46.56 min. This difference of approximately 58 min was highly statistically significant (p<0.00001). The time to achieve unassisted ambulation was also slightly longer in the Nalbuphine group (312.7±35.57 min) compared to the Buprenorphine group (299.00±30.89 min), but this difference did not reach statistical significance (p=0.09454).
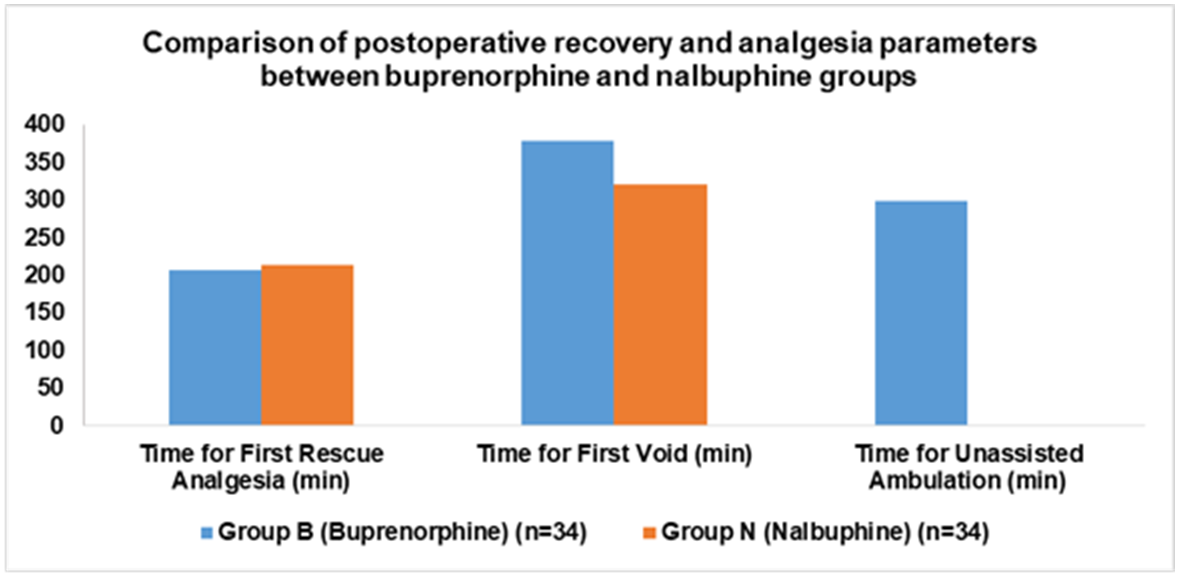
Fig. 2: Comparison of postoperative recovery and analgesia parameters between buprenorphine and nalbuphine groups
Hemodynamic stability and safety profile
Both intrathecal combinations were exceptionally well-tolerated and were associated with excellent hemodynamic stability throughout the intraoperative and early postoperative periods. There were no statistically significant differences in the mean values of heart rate, systolic blood pressure, diastolic blood pressure, or mean arterial pressure between the Buprenorphine and Nalbuphine groups at any recorded time point from baseline to 120 min post-injection (all p>0.5). Both groups exhibited a slight, gradual, and clinically insignificant decrease in these parameters following the induction of spinal anesthesia, which is a typical physiological response.
The overall incidence of perioperative adverse events was low and comparable between the two groups, as shown in table 4. A total of six adverse events were recorded in Group B, and seven were recorded in Group N. Bradycardia occurred in two patients in each group. Bradycardia accompanied by hypotension was more frequent in the Buprenorphine group (4 cases) compared to the Nalbuphine group (1 case). Conversely, isolated hypotension (3 cases) and postoperative nausea and vomiting (1 case) were observed only in the Nalbuphine group. All adverse events were mild, transient, and easily managed with standard supportive measures. Critically, no serious adverse events, such as post-dural puncture headache (PDPH), a transient neurological symptoms (TNS), or significant respiratory depression, were observed in any patient in either group. This confirms the favorable safety profile of both Nalbuphine and Buprenorphine as adjuvants to 2-Chloroprocaine in this clinical setting.
Table 4: Incidence of perioperative adverse events
| Adverse event | Group B (Buprenorphine) (n=34) | Group N (Nalbuphine) (n=34) |
| Bradycardia | 2 (5.9%) | 2 (5.9%) |
| Hypotension | 0 (0%) | 3 (8.8%) |
| Bradycardiawith Hypotension | 4 (11.8%) | 1 (2.9%) |
| Postoperative Nausea and Vomiting (PONV) | 0 (0%) | 1 (2.9%) |
| Total Patients with any Adverse Event | 6 (17.6%) | 7 (20.6%) |
Data are presented as number of patients (%).
DISCUSSION
The selection of an optimal anesthetic technique is crucial for short-duration surgeries, such as elective perianal procedures, where rapid onset, effective blockade, and swift recovery are paramount. Subarachnoid block (SAB) is a preferred method, and the use of adjuvants with short-acting local anesthetics like 2-chloroprocaine aims to prolong postoperative analgesia without delaying recovery. This study sought to compare the efficacy and safety of two opioid adjuvants, nalbuphine and buprenorphine, when added to intrathecal 1% 2-chloroprocaine. Our findings indicate that both agents are effective, but they present distinct clinical profiles that may guide their use in specific scenarios.
A key strength of this study is the effective randomization, as evidenced by the comparable demographic profiles of the two groups. The mean age in Group N (42.88 y) and Group B (41.26 y) was statistically similar. These fig. are consistent with the age ranges reported in previous studies, such as those by Jayaprakash [17] (45.64 y), Annamalai [7] (40.08 y), and Sherikar [18] (42.9 y). Similarly, the mean body weight in our nalbuphine (66.06 kg) and buprenorphine (65.59 kg) groups falls squarely within the range of 61 to 69 kg observed in studies by Annamalai [7], Jayaprakash [17], and Anshul Vishnoi [19]. This homogeneity in patient characteristics minimizes the risk of these variables acting as confounders, thereby strengthening the internal validity of our findings.
In our analysis of sensory block characteristics, buprenorphine demonstrated a more rapid onset (2.6±1.28 min) compared to nalbuphine (3.71±1 min). This finding aligns with the literature; Jayaprakash [17] and Annamalai [7] reported similarly rapid onsets for buprenorphine at 2.93±0.94 min and 1.89±0.32 min, respectively. This property reinforces buprenorphine's utility in clinical settings where a quick start to the surgical procedure is desirable. Regarding the duration of sensory block, our finding that buprenorphine provides a longer time for two-segment regression (42.09 min vs. 35.38 min for nalbuphine) is supported by Jayaprakash [17] (40.78 min) and Annamalai [7] (44.80 min). However, the time for regression to the S₂ segment showed greater variability. Our study found a slightly longer duration with nalbuphine (71.29 min) compared to buprenorphine (66.82 min), whereas Annamalai [7] reported a longer duration with buprenorphine (89.30 min). Conversely, studies by Sherikar [18] and Anshul Vishnoi [19]. Found much longer S₂ regression times with nalbuphine (96.5 min and 202.40 min, respectively), suggesting that while variable, nalbuphine can sustain sensory effects comparably or even longer in some settings. The motor block characteristics also revealed important differences. Consistent with its effect on sensory block, buprenorphine provided a significantly faster onset of motor blockade (2.85 min) compared to nalbuphine (4.78 min). This is in line with earlier reports from Jayaprakash [17] (2.73 min) and Annamalai [7] (2.71 min) and confirms buprenorphine's advantage in reducing surgical preparation time. The time to reach peak motor block, however, was nearly identical for both agents in our study, indicating equivalent efficacy in achieving a dense blockade once established. In our study, the duration of motor block was slightly longer with nalbuphine (64.41 min), a finding supported by the work of Sherikar [18] (90 min) and Anshul Vishnoi [19] (78.86 min). This contrasts with Annamalai [7], who reported a longer duration for buprenorphine (82.80 min), suggesting that nalbuphine may be advantageous in procedures that, while short, might extend slightly longer than anticipated.
Postoperative recovery and analgesia are critical endpoints in ambulatory surgery. A significant finding of our study was the wide disparity in analgesic duration compared to previous literature. The time to first rescue analgesia was comparable between our two groups (nalbuphine: 213 min; buprenorphine: 206 min). Our nalbuphine result is nearly identical to that of Sherikar [18] (210.38 min). However, the duration of analgesia for buprenorphine in our study was markedly shorter than the 855.82 min reported by Jayaprakash [17] and 715.88 min by Annamalai [7]. This suggests that while both agents provided adequate analgesia for the short-duration surgeries in our trial, nalbuphine may offer a more consistent and predictable analgesic profile across different studies. Regarding recovery, nalbuphine demonstrated an advantage in the earlier return of bladder function (time to first void: 320.76 min vs. 378.76 min for buprenorphine). This supports nalbuphine's suitability for ambulatory settings where facilitating timely micturition is beneficial.
The safety profiles of both adjuvants were favorable. The overall incidence of side effects was low and comparable between the groups. We observed two cases of bradycardia in each group, which is consistent with the 2.2% incidence reported by Jayaprakash [17] in their buprenorphine group. Hypotension occurred in three nalbuphine and four buprenorphine patients in our study, a rate notably lower than the 26.7% reported by Jayaprakash [17]. Importantly, there were no cases of pruritus, post-dural puncture headache (PDPH), or transient neurological symptoms (TNS), reinforcing the overall tolerability of intrathecal nalbuphine and buprenorphine.
Nalbuphine and buprenorphine are both effective adjuvants to 1% 2-chloroprocaine for perianal surgeries, each with unique characteristics for clinical application. Buprenorphine offers a rapid onset of sensory and motor block, enhancing operating room efficiency. In contrast, nalbuphine has a slower onset but provides predictable analgesia duration, potentially longer motor block, and quicker return of autonomic function, making it suitable for ambulatory procedures focused on fast recovery and discharge.
Limitations and future directions
This study, while robust in its design, has several limitations that should be acknowledged. Firstly, it was conducted at a single institution, which may limit the generalizability of the findings to other patient populations or practice settings. A multi-center trial would be beneficial to confirm these results. Secondly, the study utilized a fixed-dose design for the adjuvants. It is possible that different doses of either Nalbuphine or Buprenorphine could yield a more optimal balance of efficacy and side effects. Future dose-ranging studies are needed to determine the minimum effective dose of each adjuvant and improve their clinical use. Finally, while this study focused on opioid adjuvants, it would be beneficial for future research to compare these findings with non-opioid alternatives, such as dexmedetomidine, to explore other pathways for optimizing ambulatory spinal anesthesia.
CONCLUSION
In conclusion, this randomized, double-blind clinical trial demonstrates that both intrathecal Buprenorphine and Nalbuphine are effective and safe adjuvants to 1% 2-Chloroprocaine for spinal anesthesia in patients undergoing ambulatory perianal surgery. The two agents, however, offer distinct and clinically important profiles that present a clear trade-off for the clinician. Buprenorphine provides a statistically significant, albeit modest, advantage in terms of a more rapid onset of neuraxial blockade. In contrast, Nalbuphine offers a more balanced profile, combining a clinically adequate duration of postoperative analgesia with a significantly faster recovery of autonomic bladder function. In the specific context of ambulatory surgery, where facilitating a safe, predictable, and timely discharge is a primary objective, the ability to expedite a critical recovery milestone like micturition is of substantial value. Therefore, Nalbuphine may represent the more advantageous choice, optimizing not only the recovery of the individual patient but also the operational efficiency of the day-care surgical environment.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
All authors have contributed equally
CONFLICT OF INTERESTS
Declared none
REFERENCES
Divizia A, Giuseppe S Sica. Ambulatory surgery for perianal disease. In: Sarwar Zaman G, editor. Ultimate guide to outpatient care. IntechOpen; 2022. doi: 10.5772/intechopen.97119.
Dwivedi G, Sood A, Patnaik U, Kumari A. Assessment of level of patient satisfaction with day-care surgery: an observational study. Indian J Otolaryngol Head Neck Surg. 2022;74(3):5252-7. doi: 10.1007/s12070-020-01972-0, PMID 36742571.
Bhaskara B, Shruthi S, Ramachandraiah R. A study to evaluate intrathecal 1% chloroprocaine and 0.5% levobupivacaine in perianal surgeries: a prospective randomized study. Anesth Essays Res. 2020 Jul-Sep;14(3):406-11. doi: 10.4103/aer.AER_2_21, PMID 34092850.
Jain S. The effect of different doses of chloroprocaine 1% for low dose saddle anaesthesia in outpatient perianal surgery: a prospective randomized study. Indian J Clin Anaesth. 2021;8(2):270-6. doi: 10.18231/j.ijca.2021.051.
Fanelli A, Ghisi D, Allegri M. Is spinal anaesthesia a suitable technique for ultra-short outpatient procedures? Acta Biomed. 2013 Jun 1;84(1):76-80. PMID 24189768.
Koppal R, Varaprasad BV, Anuradha GR, Kurahatti S. A comparative study of bupivacaine heavy and 2-chloroprocaine for saddle block in perianal day care surgeries: a prospective randomized double blind clinical trial. Indian J Clin Anaesth. 2021;8(3):377-80. doi: 10.18231/j.ijca.2021.071.
Ghisi D, Bonarelli S. Ambulatory surgery with chloroprocaine spinal anesthesia: a review. Ambul Anesth. 2015;2015:111. doi: 10.2147/AA.S64884.
Goldblum E, Atchabahian A. The use of 2-chloroprocaine for spinal anaesthesia. Acta Anaesthesiol Scand. 2013 May;57(5):545-52. doi: 10.1111/aas.12071, PMID 23320599.
Hejtmanek MR, Pollock JE. Chloroprocaine for spinal anesthesia: a retrospective analysis. Acta Anaesthesiol Scand. 2011 Mar;55(3):267-72. doi: 10.1111/j.1399-6576.2010.02371.x, PMID 21288208.
Sell A, Tein T, Pitkanen M. Spinal 2-chloroprocaine: effective dose for ambulatory surgery. Acta Anaesthesiol Scand. 2008;52(5):695-9. doi: 10.1111/j.1399-6576.2008.01639.x, PMID 18419724.
Koyyalamudi V, Sen S, Patil S, Creel JB, Cornett EM, Fox CJ. Adjuvant agents in regional anesthesia in the ambulatory setting. Curr Pain Headache Rep. 2017 Jan;21(1):6. doi: 10.1007/s11916-017-0604-1, PMID 28210917.
Annamalai AMR, Gnanasekaran AS, Sathappan K, Chandran P. A prospective comparative study of fentanyl and buprenorphine as an adjuvant to intrathecal chloroprocaine for perianal surgeries. Bali J Anesthesiol. 2022 Jan-Mar;6(1):38-42. doi: 10.4103/bjoa.bjoa_180_21.
Gupta P, Suthar A, Deganwa M, Goyal V, Devgan S. A Comparative study of dexmedetomidine versus nalbuphine used as an adjuvant to chloroprocaine for daycare surgeries performed under subarachnoid block. Anesth Essays Res. 2022 Jul-Sep;16(3):336-9. doi: 10.4103/aer.aer_85_22, PMID 36620115.
Arunkumar R, GN, Sandhya K. A comparative study on the effect of intrathecal nalbuphine and buprenorphine as an adjuvant to 0.5% hyperbaric bupivacaine in elective infraumbilical surgeries. Indian J Clin Anaesth. 2024;11(2):159-66. doi: 10.18231/j.ijca.2024.033.
Mishra M, Marulappa SK, Madhusudhana R. Study of 2-chlorprocaine 1% with adjuvants fentanyl and Buprinorphine in comparison with plain 2-chlorprocaine 1% for subarachnoid blocks in perianal surgeries. Indian J Anaesth. 2018;5(10):1662-7. doi: 10.21088/ijaa.2349.8471.51018.12.
Mukherjee A, Pal A, Agrawal J, Mehrotra A, Dawar N. Intrathecal nalbuphine as an adjuvant to subarachnoid block: what is the most effective dose? Anesth Essays Res. 2011 Jul-Dec;5(2):171-5. doi: 10.4103/0259-1162.94759, PMID 25885383, PMCID PMC4173402.
Siddaiah J, Pujari VS, Madalu AS, Bevinaguddaiah Y, Parate LH. A comparative study on the effect of addition of intrathecal buprenorphine to 2-chloroprocaine spinal anesthesia in short duration surgeries. J Anaesthesiol Clin Pharmacol. 2019 Oct-Dec;35(4):533-9. doi: 10.4103/joacp.JOACP_65_19, PMID 31920240, PMCID PMC6939557.
Sherikar P, Osheen MJ, Mitragotri M, Gangadhar A. Comparison among intrathecal nalbuphine and fentanyl in combination with chloroprocaine 1% for short surgical lower limb procedures a prospective randomized control trial. Indian J Pain. 2023 May-Aug;37(2):115-9. doi: 10.4103/ijpn.ijpn_97_22.
Vishnoi A. Use of chloroprocaine in spinal anaesthesia-a comparative study of combination of chloroprocaine with fentanyl vs. chloroprocaine with nalbuphine for day care surgeries. Int J Med Pharm Res. 2025;6(1):331-8. doi: 10.5281/zenodo.12781166.