
Int J Curr Pharm Res, Vol 18, Issue 1, 53-57Original Article
PRESCRIPTION AUDIT OF GENERAL OPD AT A TERTIARY CARE HOSPITAL IN WEST BENGAL: CROSS-SECTIONAL STUDY
ARUNAVA BISWAS1*, SUHRITA PAUL2
1Department of Pharmacology, Barasat Government Medical College and Hospital, Barasat, West Bengal, India. 2Barasat Government Medical College and Hospital, Barasat, West Bengal, India
*Corresponding author: Arunava Biswas *Email: drabiswas@gmail.com
Received: 04 Oct 2025, Revised and Accepted: 20 Nov 2025
ABSTRACT
Objective: Rational prescription practices are essential for effective healthcare delivery, ensuring optimal therapeutic outcomes and minimizing adverse effects and healthcare costs. Prescription audits are important tools to evaluate and improve prescribing behavior, especially in public health institutions in developing countries like India.
Methods: A cross-sectional observational study was conducted at the General Outpatient Department (OPD) of Barasat Government Medical College and Hospital (BGMCH), West Bengal, from December 2024 to February 2025. A total of 274 prescriptions were analyzed using the World Health Organization (WHO) core prescribing indicators. Parameters assessed included prescription completeness, drug details, use of generics, fixed-drug combinations (FDCs), antibiotics, injections, and essential drug list compliance.
Results: All prescriptions included patient and prescriber identifiers. The average number of drugs per prescription was 3.39. Generic drugs were prescribed in 44.53% of prescriptions, while branded drugs appeared in 347 instances. Antibiotics constituted 11.4% of drugs, and injections 1.3%. About 65% of prescribed drugs were from the National List of Essential Medicines (NLEM) 2022. Only 31.39% of prescriptions included patient instructions, and no prescriptions were written in the local language.
Conclusion: The audit revealed satisfactory practices in antibiotic use and essential medicine prescribing. However, improvements are needed in diagnosis documentation, generic prescribing, and patient communication. Regular audits, standardized templates, and prescriber training are recommended to ensure rational and patient-centered prescription practices.
Keywords: Prescription audit, Rational drug use, WHO prescribing indicators, Generic drugs, Fixed-drug combinations, Essential medicines, Outpatient department
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i1.8012 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Rational drug use is a critical element in the provision of effective, safe, and economical healthcare. The World Health Organization (WHO) defines rational drug use as the process in which "patients receive medications appropriate to their clinical needs, in doses that meet their individual requirements, for an adequate period, and at the lowest cost to them and their community" [1]. Despite this definition being widely recognized, irrational prescribing remains a significant challenge, especially in low-and middle-income countries, including India.
Inappropriate prescribing practices can manifest in various forms such as overprescribing, underprescribing, incorrect dosing, polypharmacy, and the unwarranted use of antibiotics or injections. These issues not only lead to therapeutic failure but also increase the risk of adverse drug reactions, contribute to antimicrobial resistance (AMR), and place an undue financial burden on patients and healthcare systems [2]. According to studies, up to 50% of all medicines are inappropriately prescribed, dispensed, or sold, and 50% of patients fail to take them correctly [3].
India, with its vast and diverse healthcare infrastructure, is particularly vulnerable to these challenges. Public hospitals often deal with large patient volumes, time constraints, and limited resources, which can adversely affect the quality of prescription practices. Despite the introduction of national programs such as the Jan Aushadhi scheme, the National List of Essential Medicines (NLEM) or state-level programs like Fair Price Shop, irrational prescribing practices continue to persist due to inadequate training, lack of audit systems, and poor regulatory enforcement [4].
Prescription audits offer a systematic and evidence-based approach to evaluate and improve prescribing behaviour. These audits help identify patterns, detect deviations from standard treatment protocols, and facilitate targeted interventions. Moreover, audits are essential tools in promoting the rational use of medicines, improving patient outcomes, and ensuring adherence to ethical and legal standards [5]. The WHO has developed a set of core prescribing indicators to evaluate prescription quality, including the average number of medicines per encounter, the percentage of drugs prescribed by generic name, and the percentage of encounters with antibiotics or injections [6].
Barasat Government Medical College and Hospital (BGMCH) is a newly established tertiary care institution in West Bengal, India, that serves a large and diverse population. With the increasing burden of outpatient care, especially in general OPD settings, it becomes essential to monitor prescription practices to ensure the delivery of quality healthcare. Although prescription audits are a recognized tool for quality improvement, limited literature exists on their implementation and outcomes in newly functional medical colleges.
This study presents the first prescription audit conducted at the General OPD of BGMCH. The primary aim was to assess the quality of prescriptions using WHO core indicators, identify common prescribing patterns, and evaluate adherence to rational drug use principles. The findings are expected to guide future interventions, facilitate prescriber education, and promote safer and more effective patient care.
MATERIALS AND METHODS
This study employed a cross-sectional observational design to assess prescription practices in the General Outpatient Department (OPD) of Barasat Government Medical College and Hospital (BGMCH), a tertiary care facility in West Bengal. The study was conducted by the faculties from the Department of Pharmacology over a three-month period from December 2024 to February 2025.
Study setting and population
The General OPD at BGMCH caters to a diverse patient population from both rural and urban areas. The department functions six days a week and handles an average footfall of 500–700 patients per day. Prescriptions issued during routine consultation hours formed the data pool for this study.
Sample size and sampling technique
A total of 280 prescriptions were randomly collected. Prescriptions were selected through a systematic sampling approach by choosing every 10th prescription from the registration list. Of these, 274 prescriptions were deemed eligible for analysis, while six were excluded due to incomplete or missing data.
Inclusion and exclusion criteria
Inclusion criteria: All outpatient prescriptions with complete information on patient identifiers, drug details, and prescriber details.
Exclusion criteria: Prescriptions lacking critical information such as patient identification, prescriber signature, or illegible handwriting.
Data collection tool
A standardized data abstraction form based on WHO core drug use indicators was used to extract information from each prescription [6]. The form included parameters such as:
Patient and prescriber identifiers
Date and time of prescription
Drug name (generic or brand), dose, route, frequency, and duration
Presence of fixed-drug combinations (FDCs)
Use of antibiotics and injections
Use of drugs from the National List of Essential Medicines (NLEM 2022)
Availability of medicines in the hospital pharmacy
Documentation of clinical findings and diagnosis
Presence of patient instructions or advice
Use of abbreviations and local language
Ethical considerations
The study involved a retrospective review of anonymised prescriptions and did not involve any patient contact. With the permission obtained from the authority of the medical college, the study was approved by the Institutional Ethics Committee of BGMCH, and ethical clearance was waived off. All data were handled with strict confidentiality and used solely for academic and institutional improvement purposes.
Statistical analysis
Data were compiled in Microsoft Excel and analysed using SPSS version 22. Descriptive statistics such as frequencies, means, and percentages were calculated to describe the data. The results were presented using tables and fig. for ease of interpretation. This methodological framework ensured systematic, reproducible, and ethically sound data collection, which forms the foundation for deriving meaningful insights from the prescription audit [7, 8].
RESULTS
A total of 274 prescriptions were analysed, comprising 930 prescribed drugs. The findings are categorized into prescription characteristics, prescribing indicators, and patient instruction parameters. Table 1 depicts the general prescription profile of the patients who attend the general OPD and (table 2) indicates the result of legibility and diagnostic indicators.
Table 1: General prescription profile
| Parameter | Number (%) |
| Total prescriptions analyzed | 274 |
| Total drugs prescribed | 930 |
| Average drugs per prescription | 3.39 |
| Prescriptions with complete patient/prescriber ID | 274 (100%) |
| Prescriptions with date/time | 274 (100%) |
| Prescriptions with signature | 219 (80%) |
Table 2: Legibility and diagnostic indicators
| Parameter | Frequency (%) |
| Legible prescriptions | 242 (88.32%) |
| Clinical findings/symptoms mentioned | 138 (50.36%) |
| Diagnosis mentioned (provisional/final) | 96 (35.04%) |
The pattern of drug found to be prescribed as shown in (table 3) indicates most of them were generic and antibiotic/injections were advised in minimum quantity. Most of the drugs in the prescription was as per NLEM and were dispensed from the hospital pharmacy (table 4).
Table 3: Drug prescribing patterns
| Parameter | Value |
| Generic drugs | 414 (44.53%) |
| Branded drugs | 347 (37.3%) |
| FDCs used | 84 prescriptions (30.66%) |
| Antibiotics prescribed | 106 (11.4%) of total drugs |
| Injections prescribed | 12 (1.29%) |
Table 4: Essential drug use and dispensation
| Parameter | Value |
| Drugs from NLEM 2022 | 605 (65%) |
| Drugs dispensed from hospital pharmacy | 587/930 (63.11%) |
The study also reveals that use of abbreviation in the prescription was rampant and genuine essential advice to the patients were lacking with no instruction provided to the patients in local language as shown in table 5 and the trend of using abbreviation among prescriber is depicted in fig. 1.
Table 5: Communication and language
| Parameter | Frequency (%) |
| Instructions/advice present | 86 (31.39%) |
| Prescriptions with abbreviations | 268 (97.81%) |
| Prescriptions in local language | 0 |
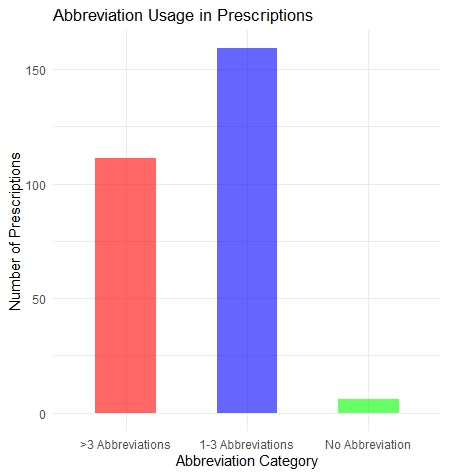
Fig. 1: Number of abbreviation used by the prescribers, the pie chart in (fig. 2) shows that based on the sample analysed, how many doctors used brand names and how many such medicines were there in the prescription
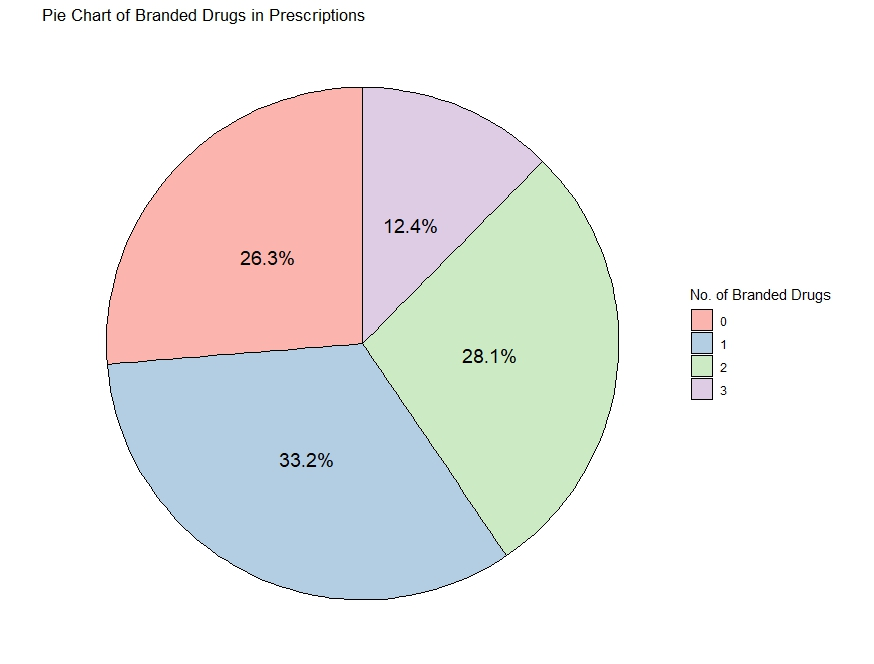
Fig. 2: Pie chart of branded drugs in prescriptions
All he above results of this study altogether indicates a reasonably rational use of antibiotics and injections, good coverage of essential medicines, but highlight areas for improvement in diagnosis documentation, generic prescribing, and patient instruction practices.
DISCUSSION
This prescription audit at Barasat Government Medical College and Hospital (BGMCH) offers a comprehensive overview of current prescribing practices in the General OPD. The findings are a mixture of positive trends and areas requiring intervention. One of the strengths of this study was the 100% documentation of patient and prescriber identification, as well as the consistent recording of the date and time. This level of documentation meets both administrative and medico-legal standards. In contrast, a study conducted at a tertiary care hospital in North India by Sharma et al. (2021) reported only 91% of prescriptions with complete patient details and 87% with prescriber identification [9]. Another study from Western Nepal showed even lower compliance at 78% [5]. Thus, BGMCH demonstrates commendable adherence in this domain.
While the legibility rate (88.32%) is above average compared to other studies, the absence of prescriber signatures in nearly 20% of prescriptions remains concerning. In a study by Gupta et al. (2016) in Uttarakhand, 25% of prescriptions were unsigned, raising similar legal and ethical concerns [10]. Signature verification is critical in holding prescribers accountable and ensuring authenticity.
Only 50.36% of the prescriptions included clinical findings, and a mere 35.04% mentioned a diagnosis. This under-documentation severely restricts the continuity of care and is a trend echoed across studies. Jha et al. (2005) in Nepal reported that just 42% of prescriptions included clinical diagnoses [11]. Similarly, a study in Andhra Pradesh found diagnosis documented in only 29% of prescriptions [12]. This highlights a systemic gap in clinical documentation across South Asian healthcare system.
The average number of drugs per prescription was 3.39, aligning with WHO recommendations of less than or equal to three. Comparable results were found in a study conducted in a government medical college in Maharashtra where the average was 3.1 [13]. Polypharmacy, although not excessive, must be closely monitored to avoid adverse drug reactions and drug interactions.
The percentage of generic drug prescribing (44.53%) was relatively low, reflecting trends seen in many Indian institutions. A study by Shankar et al. (2012) in South India showed only 35% of prescriptions using generic names [14]. Despite government mandates and availability of generic alternatives through schemes like Fair Price Shop located within the hospital campus, prescribers often prefer branded drugs due to habit or perceived efficacy.
Fixed drug combinations (FDCs) were used in 30.66% of the prescriptions. While some FDCs are clinically justified (e. g., amoxicillin+clavulanic acid), others may contribute to unnecessary exposure to multiple active ingredients. In a study by Desalegn (2013) in Ethiopia, 26% of prescriptions included FDCs, with many lacking clear therapeutic justification [15]. This highlights the need for prescriber education and guideline adherence.
The proportion of antibiotic prescriptions (11.4%) and injections (1.3%) in this study was low compared to global and national trends. The WHO recommends that less than 30% of encounters should involve antibiotic use [16]. A survey by Holloway and Henry (2014) analysing WHO database fig. found that antibiotic prescribing in developing countries often exceeds 40% [17]. Thus, BGMCH shows encouraging results, possibly reflecting increasing awareness of antimicrobial resistance.
The study found that 65% of drugs were from the National List of Essential Medicines (NLEM) 2022, aligning with public health goals. In comparison, a prescription audit in Karnataka reported NLEM compliance of 52% [18], and in Nepal, compliance ranged between 45–60% [5]. This suggests that institutional procurement practices and policy awareness at BGMCH may be relatively robust.
Only 31.39% of prescriptions included instructions for patients, and none were written in the local language. These fig. are consistent with similar studies across India, where patient instructions are frequently omitted. A South Indian study by Thiruthopu et al. (2014) found that only 20% of prescriptions contained usage instructions [19]. The absence of local language use further isolates patients, especially those with limited literacy. Nearly 98% of prescriptions used abbreviations. Although some abbreviations are widely accepted (e. g., TID, QID), others may be ambiguous and lead to misinterpretation. Studies from Malaysia and the Philippines report similar findings, linking abbreviation use with increased medication errors [20]. Training programs emphasizing clarity in prescription writing could address this. These trends underscore the need for systemic interventions, including electronic prescribing systems, regular CME (continuing medical education) sessions, prescription template standardization, and robust internal audits.
LIMITATIONS
Prescriptions from the General OPD were only audited, excluding inpatient and specialty departments. As the duration of the study was conducted over three months, the data may not reflect seasonal variations. The present study focused on prescription content without linking it to clinical outcomes. During this study patient and prescriber perspectives were not captured and risk of observational bias and transcription errors cannot be ruled out.
CONCLUSION
This prescription audit at BGMCH, Barasat provides a valuable snapshot of current outpatient prescribing practices and highlights both commendable efforts and areas in need of targeted improvement. Positive trends such as high documentation compliance, restrained antibiotic and injection usage, and adherence to essential medicines were observed. However, challenges, including under-documentation of diagnosis, limited generic prescribing, excessive use of abbreviations, and insufficient patient instructions, remain evident. These findings emphasize the necessity for regular audits, prescriber education, and implementation of standardized prescription formats. Promoting rational drug use through institutional policies, continuous medical education, and better patient communication is crucial to optimizing therapeutic outcomes and ensuring patient safety. Institutionalizing such audits will not only improve prescription practices but also contribute to broader public health goals by fostering a culture of accountability, transparency, and evidence-based clinical care in the public healthcare system.
ACKNOWLEDGEMENT
The authors would like to the thank the senior resident doctors from the Department of Pharmacology for assisting in data collection, statistician for analysis of data and the college authorities for providing all logistic support during conduct of the study.
AUTHORS CONTRIBUTIONS
AB: Concept, design of study and literature search, Data acquisition, data analysis, statistical analysis, manuscript preparation
SP: Manuscript preparation, manuscript editing and manuscript review
CONFLICT OF INTERESTS
Declared none
REFERENCES
World Health Organization. Guide to good prescribing: a practical manual. WHO; 1994.
Holloway K, van Dijk L. Rational use of medicines: key concepts. WHO; 2011.
World Health Organization. Medicines: rational use of medicines. WHO Fact Sheet No. 338; 2010.
Ministry of Health and Family Welfare; Govt. of India. National list of essential medicines (NLEM); 2022.
Tuladhar H. An overview of reproductive health of women in Bajhang district. Nepal Med Coll J. 2005;7(2):107-11. PMID 16519075.
WHO. How to investigate drug use in health facilities: selected drug use indicators. WHO/DAP/93.1. Geneva: WHO; 1993.
Hogerzeil HV. Promoting rational prescribing: an international perspective. Br J Clin Pharmacol. 1995;39(1):1-6. doi: 10.1111/j.1365-2125.1995.tb04402.x, PMID 7756093.
Laporte JR. Towards a healthy use of pharmaceuticals. Dev Dialogue. 1985;1(2):48-55. PMID 12341047.
Sharma S. Prescription audit in a tertiary care hospital in North India: a cross-sectional study. Int J Basic Clin Pharmacol. 2021;10(3):224-9.
Gupta SK, Nayak RP. Drug prescribing patterns in a tertiary care teaching hospital in Uttarakhand. J Clin Diagn Res. 2016;10(2):1-4.
Desalegn AA. Assessment of drug use pattern using WHO prescribing indicators at Hawassa University teaching and referral hospital, South Ethiopia: a cross-sectional study. BMC Health Serv Res. 2013;13(1):170. doi: 10.1186/1472-6963-13-170, PMID 23647871.
Reddy P. Evaluation of prescribing pattern in a tertiary care hospital of Andhra Pradesh. Int J Res Dev Pharm l Sci. 2014;3(1):749-53.
Chavda DA, Yadav P, Patel VJ. Drug utilization pattern in medicine department in a Tertiary Care Teaching Hospital. Int J Basic Clin Pharmacol. 2014;3(6):1015-19.
Shankar PR. Prescribing practices in the teaching hospital in South India: a WHO-based prescription audit. J Clin Diagn Res. 2012;6(1):145-9.
Desalegn AA. Rational drug use: drug prescribing indicators in public health facilities of Southern Ethiopia. J Pharm Sci Res. 2013;5(1):45-8.
WHO. The rational use of drugs: report of the conference of experts. Nairobi; 1985.
Holloway KA, Henry D. WHO essential medicines policies and use in developing and transitional countries: an analysis of reported policy implementation and medicines use surveys. PLOS Med. 2014;11(9):e1001724. doi: 10.1371/journal.pmed.1001724, PMID 25226527.
Kumar R. Evaluation of rational drug prescribing practices in Tertiary Care Hospital, Karnataka. Int J Med Sci Public Health. 2016;5(4):707-11.
KP, Kunikullaya UK, Goturu J. Glycosylated haemoglobin (HbA1c)-a marker of circulating lipids in type 2 diabetic patients. J Clin Diagn Res. 2014;8(2):20-3. doi: 10.7860/JCDR/2014/7742.3996, PMID 24701471.
Pham HT. Abbreviation use in prescriptions and its impact on patient safety: a study in Malaysian healthcare setting. Pharm Pract. 2012;10(3):104-9.