
Int J Curr Pharm Res, Vol 18, Issue 1, 58-61Original Article
ROLE OF SPHENOPALATINE GANGLION BLOCK IN REDUCING PERIOPERATIVE OPIOID CONSUMPTION IN TONGUE CANCER SURGERY: A CLINICAL CASE SERIES
ARCHANA GAUTAM1*, VANDNA BHARTI2
1Department of Anaesthesiology, Kalyan Singh Super Speciality Cancer Institute, Lucknow, India. 2Department of Anaesthesiology and Critical Care, King George's Medical University, Lucknow India
*Corresponding author: Archana Gautam; *Email: archana.dolly76@gmail.com
Received: 06 Oct 2025, Revised and Accepted: 26 Nov 2025
ABSTRACT
Objective: To determine the time interval for first rescue analgesia that is intravenous diclofenac in the post-anaesthesia care unit and assess its impact on perioperative fentanyl consumption.
Methods: This case series evaluated sphenopalatine ganglion block (SPGB) as an adjunct to general anaesthesia in seven patients with carcinoma of the tongue who underwent elective surgery. The ultrasound-guided SPGB via suprazygomatic approach using 4 ml of 0.5% bupivacaine was performed after administering general anaesthesia to the patient.
Results: SPGB was successfully performed within 5–6 min and was associated with 200–260ug perioperative fentanyl and 2.0–3.1µg/kg/min nitro-glycerine use. Hemodynamic remained stable, blood loss was 140–180 ml, and the surgical field improved. Postoperatively, pain scores (VAS<3) were maintained for 22–24 h, with delayed need for rescue analgesia, effectively managed with intravenous diclofenac. No major complications were observed.
Conclusion: These findings suggest that SPGB is a simple, safe, and effective component of enhanced recovery after surgery (ERAS) in oral cancer surgery, reducing opioid consumption and improving surgical and postoperative outcomes.
Keywords: Cancer of head and neck, Enhanced recovery after surgery, Regional anaesthesia, Pterygopalatine ganglion block, Ultrasonography
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i1.8024 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Inadequate management of postoperative pain remains a prevalent issue among surgical patients. Research suggests that nearly 40% of individuals undergoing surgery experience moderate to severe pain within the first 24 h following their procedure in a general surgical setting [1]. Cancer patients frequently present with chronic pain and established opioid requirements, which pose significant challenges to perioperative analgesic management. Early consultation with the acute pain service is therefore considered essential in optimizing outcomes for this population [2]. In cases of oral squamous cell carcinoma, pain is particularly prevalent when the neoplastic lesion involves the anterior portion of the tongue, reflecting the high density of sensory innervation in this region [3]. Within the framework of enhanced recovery after surgery (ERAS) protocols, multimodal and opioid-sparing analgesia is strongly advocated to minimize opioid-related side effects, optimize recovery, and improve patient outcomes [4-6]. Specifically, ERAS pathways in head and neck cancer surgery have demonstrated reductions in opioid use, length of hospital stay, and perioperative complications, underscoring their relevance in this patient population [7–10]. Incorporating regional techniques such as the sphenopalatine ganglion block aligns with these principles by providing effective perioperative pain control while reducing opioid consumption.
Sphenopalatine ganglion (SPG), also referred to as pterygopalatine ganglion, is located in the pterygopalatine fossa (PPF), posterior to the middle nasal turbinate, anterior to the pterygoid canal, and lateral to the sphenopalatine foramen [11].
The sphenopalatine ganglion block (SPGB) has demonstrated its efficacy in the management of trigeminal neuralgia, migraines, cluster headache, and post-dural puncture headache. It also reduces the analgesic requirements following endoscopic sinus surgery and maxillofacial cancer surgery, and provides significant analgesia to patients with advanced head and neck cancer [12].
SPGB can be performed through transnasal (topical and injection), transoral, or transcutaneous approaches (infra-zygomatic and suprazygomatic) [13].
This case series seeks to evaluate the role of SPGB in optimizing perioperative pain management among cancer patients undergoing surgery. The objectives include determining the time interval to first rescue analgesia in the post-anaesthesia care unit (PACU) and assessing its impact on perioperative fentanyl consumption.
MATERIALS AND METHODS
Patients diagnosed with carcinoma of the tongue (involving the anterior or lateral aspect) were enrolled in the study after obtaining written informed consent. A combination of general anaesthesia and SPGB was planned for the procedure.
Each patient underwent a thorough history and clinical examination, with specific attention to the anatomical site designated for local anaesthetic administration. Standard preoperative investigations were performed, including complete blood count, prothrombin time, activated partial thromboplastin time, international normalized ratio, and random blood glucose levels. Preoperative counselling was provided the day before surgery, during which fasting instructions were explained. Patients were also familiarized with the Visual analogue score for pain assessment, a validated 11-point scale ranging from 0 (no pain) to 10 (worst pain).
Upon arrival in the operating room, the heart rate, non-invasive blood pressure, oxygen saturation, respiratory rate, and temperature were monitored. An 18-gauge intravenous line was secured. General anaesthesia was induced using intravenous fentanyl (2 μg/kg) and incremental doses of propofol (2–3 mg/kg). Nasotracheal intubation was facilitated with intravenous vecuronium bromide (0.1 mg/kg). Anaesthesia was maintained with sevoflurane delivered at a low flow (1 l/min oxygen) with a mixture of 50% oxygen and 50% air, adjusting the minimum alveolar concentration (MAC) between 2% and 3%. Neuromuscular blockade was maintained with vecuronium bromide (0.05 mg/kg) administered every 30 min. Patients were mechanically ventilated to maintain an end-tidal CO₂ (EtCO₂) of 35–40 mmHg.
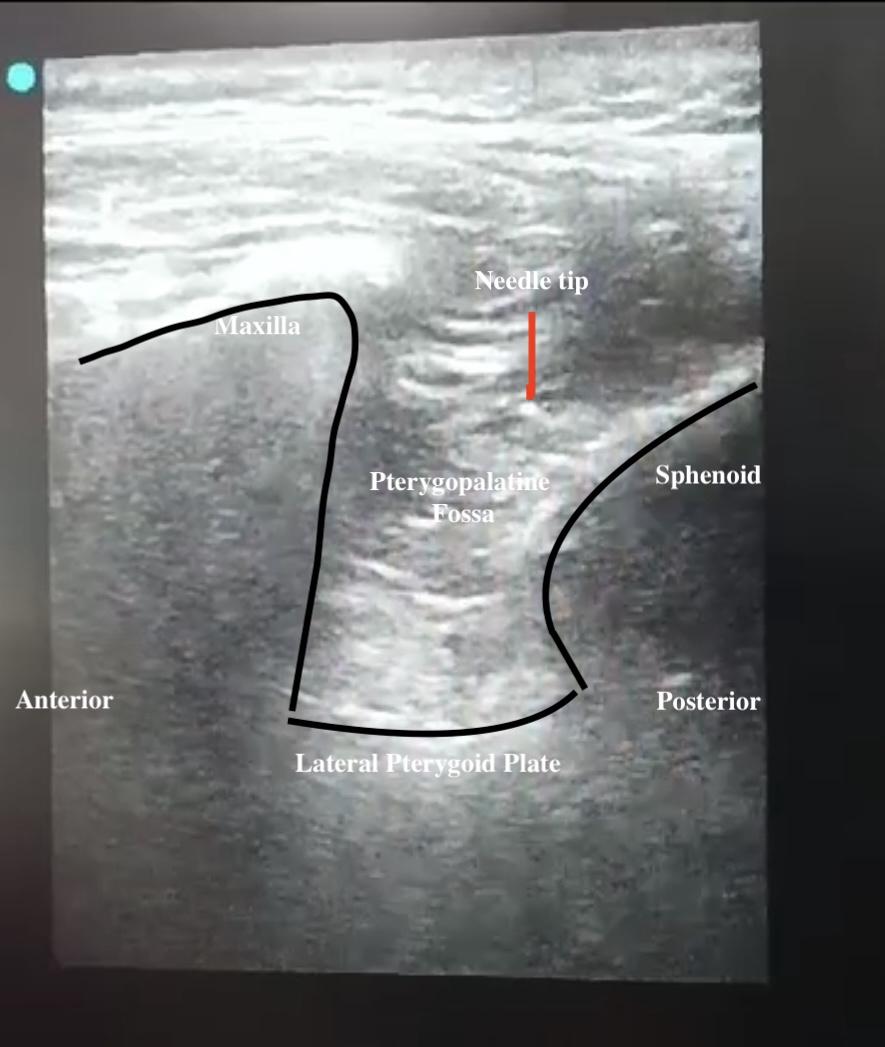
Fig. 1: Sonoanatomy of supra-zygomatic approach sphenopalatine ganglion block
The SPGB was administered under sterile conditions using an ultrasound-guided transcutaneous suprazygomatic approach on the affected side. A total of 4 ml of 0.5% bupivacaine was injected. The procedure was performed with the patient in the supine position, employing a linear ultrasound probe (5–12 MHz). The transducer was positioned in the infrazygomatic region on the tumour side, over the maxilla, and angled 45° cephalad. In this angle, PPF was visualized anteriorly between the maxilla and posteriorly between the pterygoid process [fig. 1]. A 25-gauge spinal needle was inserted above the zygomatic arch and lateral to the posterior orbital rim. It was then advanced 45° caudally and 10° anteriorly, directed toward the PPF. Afterward, 4 ml of 0.5% bupivacaine was injected over 15-20 sec, while the spread of the local anaesthetic was observed under USG. Time required to perform the technique is defined as the time from the beginning of scanning until the study drug injection and will be recorded. Intraoperative analgesia was supplemented with bolus doses of fentanyl (1μg/kg) if mean arterial pressure (MAP) and/or heart rate (HR) increased by more than 20% from baseline. Target intraoperative MAP of 60–65 mmHg was maintained by the adjustment of the nitroglycerine infusion from 0.5 to 10ug/kg/min according to the patient’s response. The total intraoperative fentanyl requirements were recorded. Hemodynamic parameters, including HR, MAP, and SpO₂, were recorded every 30 min. Bradycardia (HR<45 beats/min) was managed with intravenous atropine (300 μg).
Following extubating, all patients will be transferred to the post-anaesthesia care unit (PACU), where they will be monitored for 48 h. Post-operative analgesia will be provided by nursing staff with IV paracetamol 1 g every 8 h. Rescue analgesia will be provided by nursing staff when the patient complains of pain and has a VAS score>4. IV diclofenac 75 mg will be used as a rescue analgesic. The adverse effects, including nausea, vomiting, persistent paraesthesia, and respiratory depression, were noted and treated in accordance with customary protocols.
RESULTS
Table 1 summarizes the patient characteristics, including age, gender, body mass index (BMI), comorbidities, diagnosis, surgical procedure, and duration of surgery. Seven patients were included in the study, comprising five males and two females, with a mean age of approximately 52 y (range 38–71 y) and a mean BMI of 21.8 kg/m². One patient had diabetes mellitus (15 y, on oral hypoglycaemics), and one had hypertension controlled on telmisartan-hydrochlorothiazide; the remaining patients had no comorbidities. Four patients had squamous cell carcinoma of the left lateral tongue, and three of the right lateral tongue. Surgical procedures included two hemi-glossectomies with supraomohyoid neck dissection (SOHND), two wide local excisions with Modified neck dissection (MND), and three wide local excisions with SOHND. The average duration of surgery was 2.5 h in all patients.
Table 1: Patient characteristics and surgical details
| Case No. | Age/Gender | BMI kg/m2 | Co-morbidity | Diagnosis | Surgery done | Duration of the surgery done |
| 59/M | 21.3 | Type II Diabetes Mellitus | SCC left lateral border of the tongue | WLE+MND | 2 h | |
| 64/F | 22.1 | none | SCC left lateral border of the tongue | WLE+MND | 2.3 h | |
| 44/M | 23.4 | none | SCC right lateral border of the tongue (post-NACT) | Rt hemiglossectomy +MND | 3 h | |
| 38/M | 19.1 | none | SCC left lateral border of the tongue | WLE+SOHND | 2.3 h | |
| 71/F | 21.5 | none | SCC left lateral border of the tongue | Left hemiglossectomy +SOHND | 3 h | |
| 40/M | 24.3 | none | SCC right lateral border of the tongue | WLE+SOHND | 2 h | |
| 47/M | 20.9 | hypertension | SCC right lateral border of the tongue | WLE+SOHND | 2.2 h |
Abbreviations: M= Male; F= Female, BMI = Body mass index; SCC = Squamous cell carcinoma; WLE = Wide local excision; MND = Modified neck dissection; SOHND = Supraomohyoid neck dissection.
Table 2: Perioperative events
| Case no. | Time required to perform the SPGB (min) | Use of nitro-glycerine (µg/kg/min) | Intraoperative blood loss (ml) | Postoperative time of first rescue analgesia (h) | Peri-operative total fentanyl consumption (µg) | Complication in PACU |
| 5.0 | 2.5 | 150 | 20 h | 225 | none | |
| 5.3 | 3 | 180 | 24 h | 200 | none | |
| 5.5 | 2 | 140 | 24 h | 250 | hypo-aesthesia in the cheek | |
| 5.2 | 3.1 | 150 | 24 h | 225 | none | |
| 5.0 | 2 | 150 | 24 h | 260 | nausea and vomiting | |
| 5.4 | 2.2 | 140 | 24 h | 200 | none | |
| 5.5 | 3 | 180 | 22 h | 200 | nausea and vomiting |
PACU= Postoperative intensive care unit
Table 2 summarizes the perioperative events. These include the time required to perform the SPGB (in minutes), the dose of nitroglycerine (in µg/kg/min) administered to maintain MAP, intraoperative blood loss (in ml), the time required to administer first rescue analgesic in the PACU (in hours), total perioperative fentanyl consumption (in µg), and any SPGB-related complications observed in the PACU.
The mean time required to perform the SPGB via the suprazygomatic approach under general anaesthesia was 5.27 min (range 5.0–5.5). The average nitroglycerine dose administered to maintain MAP was 2.54 µg/kg/min (range 2.0–3.1), and the mean intraoperative blood loss was approximately 155.7 ml (range 140–180). The first rescue analgesic in the PACU was required after an average of 23 h (range 20–24). The mean perioperative fentanyl consumption was 222 µg (range 200–260). Among the seven patients studied, two (28.2%) experienced nausea and vomiting, while one (14.3%) patient reported ipsilateral cheek hypoesthesia; no serious adverse events were observed.
DISCUSSION
Managing pain in individuals with oral cancer poses a significant clinical challenge due to the multifactorial nature of pain in this population, which may arise from tumour infiltration, surgical intervention, or adjuvant treatments such as chemotherapy and radiotherapy. The complexity is further compounded by functional impairments that affect speech, swallowing, and nutrition, all of which contribute to a diminished quality of life. These factors necessitate a comprehensive, multimodal, and individualized approach to perioperative pain control, emphasizing the importance of integrating pharmacological, regional, and supportive strategies to achieve optimal outcomes. Regional anaesthesia techniques are increasingly recognized in head and neck oncological surgery as part of multimodal, opioid-sparing analgesia that align well with the principles of ERAS protocols. ERAS emphasizes multimodal, opioid-sparing analgesia, reduction of perioperative stress response, early mobilization, and improvement in overall postoperative recovery [8, 9, 1, 4].
Cata et al. reported that 15% of patients undergoing surgery for oral tongue tumours continued opioid use even one year postoperatively, underscoring the concern that poorly managed acute pain may contribute to persistent pain syndromes and chronic opioid dependence [15]. This risk is further influenced by factors such as preoperative opioid exposure and baseline pain severity [16].
In this case series, the SPGB provided effective perioperative analgesia, reduced fentanyl consumption, and prolonged the time to first rescue analgesia, with postoperative pain relief lasting up to 24 h. By optimizing perioperative pain control, SPGB may not only reduce the immediate pain burden but also help minimize long-term opioid dependence. Importantly, these benefits align with the principles of ERAS.
Patients in our case series required rescue analgesia only after 22–24 h in the PACU, with early VAS scores remaining below 3. Consistent with the patients receiving bilateral PPF block experienced lower VAS scores for pain control lasting up to 18 h postoperatively [17]. Rezaeian et al. reported significantly lower VAS scores within 24 h after endoscopic sinus surgery with SPGB [18]. The analgesic effect is mediated via the blockade of sensory fibres from the maxillary division of the trigeminal nerve and autonomic fibres within the ganglion, thereby attenuating nociceptive transmission, modulating autonomic responses, and providing a significant opioid-sparing benefit in oral cancer surgery. In all cases, the patient had complained of pain either at the neck suture line or at the drain site and demanded the rescue analgesia.
Intravenous diclofenac 75 mg was used as rescue analgesia, consistent with the important role of NSAIDs in multimodal pain management. Supporting this, Puttaswamy et al. compared aceclofenac and diclofenac in patients undergoing composite resection for oral cancer, reporting that both agents significantly reduced VAS scores at 72 h, with diclofenac providing slightly superior analgesic efficacy [19].
SPGB effectively attenuated intense perioperative nociceptive stimuli while maintaining stable intraoperative hemodynamic, reflected by target MAP achievement with markedly reduced fentanyl and nitroglycerin requirements (mean nitroglycerin 2.54 µg/kg/min, consistent with Abdelghafaretals 2 ± 1 µg/kg/min) [17]. Head and neck cancer surgeries typically demand high opioid doses, with Patino et al. reporting a median intraoperative fentanyl equivalent of 1,081 µg.([20]) In our series, patients required only 200–260 µg fentanyl, likely due to the analgesic effect of SPGB and the omission of reconstructive procedures, highlighting its potential to reduce perioperative opioid consumption substantially.
Additionally, the technique improved surgical field visualization, as evidenced by a reduction in intraoperative blood loss of approximately 25-50%, which is consistent with the numerous studies [17, 21, 23]. This effect may be attributed to SPG blockade, which promotes unopposed sympathetic activity, resulting in improved haemostasis and a clear surgical field.
We observed that the technique required only a short administration time, approximately 5.0–5.5 min. Similarly, Nader et al. [22] reported successful ultrasound-guided pterygopalatine fossa (PPF) blocks within 5 min of needle insertion, while Abdelghafar et al. [17] achieved comparable results within 6 min.
In our case series, 28.2% of patients experienced postoperative nausea and vomiting within the first 24 h. Similarly, Abdelghafar et al. reported an incidence of 33.3%, whereas Misk et al. observed this complication in only 4% of patients [17, 24]. Ipsilateral cheek hypoesthesia was noted in 14.3% of our patients, compared with 8% reported by Misk et al. [24]. While no serious adverse events were encountered in our series, further randomized controlled trials are warranted to define the incidence and spectrum of complications associated with the SPGB block in oral cancer surgeries. The technique is relatively simple to perform under ultrasound guidance, with minimal complications noted in our series, further supporting its feasibility as part of ERAS pathways.
CONCLUSION
Individualized multimodal strategies are essential for effective postoperative pain control in tongue cancer surgery. When combined with general anaesthesia, the SPGB enhances hemodynamic stability, reduces intraoperative blood loss, and improves postoperative analgesia while lowering opioid requirements. As a simple and safe technique, SPGB aligns with ERAS principles by supporting opioid-sparing multimodal analgesia and facilitating recovery.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
CONFLICT OF INTERESTS
Declared none
REFERENCES
Beauregard L, Pomp A, Choiniere M. Severity and impact of pain after day-surgery. Can J Anaesth. 1998 Apr;45(4):304-11. doi: 10.1007/BF03012019, PMID 9597202.
McCarthy J, Ratnasabapathy U. Anaesthesia for maxillofacial surgery. Anaesth Intensive Care Med. 2020;21(9):457-62. doi: 10.1016/j.mpaic.2020.06.002.
Hechler B, Carlson ER, Heidel RE, Fahmy MD, McCoy JM. Are oral pain and otalgia predictive of perineural invasion in squamous cell carcinoma of the oral tongue? J Oral Maxillofac Surg. 2020;78(8):1418-26. doi: 10.1016/j.joms.2020.03.029, PMID 32360237.
Echeverria Villalobos M, Stoicea N, Todeschini AB, Fiorda Diaz J, Uribe AA, Weaver T. Enhanced recovery after surgery (ERAS): a perspective review of postoperative pain management under ERAS pathways and its role on opioid crisis in the United States. Clin J Pain. 2020;36(3):219-26. doi: 10.1097/AJP.0000000000000792, PMID 31868759.
American College of Obstetricians and Gynecologists. ACOG committee opinion No. 750: perioperative pathways: enhanced recovery after surgery. Obstet Gynecol. 2018;132(3):e120-30. doi: 10.1097/AOG.0000000000002818, PMID 30134426.
Loria CM, Zborek K, Millward JB, Anderson MP, Richardson CM, Namburi N. Enhanced recovery after cardiac surgery protocol reduces perioperative opioid use. JTCVS Open. 2022 Sep 6;12:280-96. doi: 10.1016/j.xjon.2022.08.008, PMID 36590721.
Coyle MJ, Main B, Hughes C, Craven R, Alexander R, Porter G. Enhanced recovery after surgery (ERAS) for head and neck oncology patients. Clin Otolaryngol. 2016;41(2):118-26. doi: 10.1111/coa.12482, PMID 26083896.
Kiong KL, Vu CN, Yao CM, Kruse B, Zheng G, Yu P. Enhanced recovery after surgery (ERAS) in head and neck oncologic surgery: a case-matched analysis of perioperative and pain outcomes. Ann Surg Oncol. 2021;28(2):867-76. doi: 10.1245/s10434-020-09174-2, PMID 32964371.
Jandali DB, Vaughan D, Eggerstedt M, Ganti A, Scheltens H, Ramirez EA. Enhanced recovery after surgery in head and neck surgery: reduced opioid use and length of stay. Laryngoscope. 2020;130(5):1227-32. doi: 10.1002/lary.28191, PMID 31314144.
Nieminen T, Tapiovaara L, Back L, Lindford A, Lassus P, Lehtonen L. Enhanced recovery after surgery (ERAS) protocol improves patient outcomes in free flap surgery for head and neck cancer. Eur Arch Otorhinolaryngol. 2024 Feb;281(2):907-14. doi: 10.1007/s00405-023-08292-3, PMID 37938375.
Windsor RE, Jahnke S. Sphenopalatine ganglion blockade: a review and proposed modification of the transnasal technique. Pain Physician. 2004;7(2):283-6. PMID 16868606.
Ho KW, Przkora R, Kumar S. Sphenopalatine ganglion: block, radiofrequency ablation and neurostimulation a systematic review. J Headache Pain. 2017;18(1):118. doi: 10.1186/s10194-017-0826-y, PMID 29285576.
Alexander CE, Dua A, Block SG. StatPearls. Treasure Island, FL: StatPearls Publishing; 2022 Jan. p. 2025.
Patel J, Jones C. Enhanced recovery after surgery for major head & neck surgery a narrative review. J Oral Maxillofac Anesth. 2023;2:24. doi: 10.21037/joma-23-15.
Cata JP, Patino M, Gorur A, Du KN, Uhelski ML, Myers J. Persistent and chronic postoperative opioid use in a cohort of patients with oral tongue squamous cell carcinoma. Pain Med. 2020 May 1;21(5):1061-7. doi: 10.1093/pm/pnz242, PMID 31609416.
Inchingolo AM, Dipalma G, Inchingolo AD, Palumbo I, Guglielmo M, Morolla R. Advancing postoperative pain management in oral cancer patients: a systematic review. Pharmaceuticals (Basel). 2024 Apr 22;17(4):542. doi: 10.3390/ph17040542, PMID 38675500.
Abdelghafar EM, Abbas DN, Othman A, Zayed SB, Shawki AH. A prospective randomized clinical trial to evaluate analgesic efficacy of bilateral pterygopalatine fossa injection in patients undergoing maxillofacial cancer surgeries under general anesthesia. Egypt J Anaesth. 2021;37(1):159-66. doi: 10.1080/11101849.2021.1903667.
Rezaeian A, Hashemi SM, Dokhanchi ZS. Effect of sphenopalatine ganglion block with bupivacaine on postoperative pain in patients undergoing endoscopic sinus surgery. Allergy Rhinol (Providence). 2019 Jan 23;10:2152656718821282. doi: 10.1177/2152656718821282, PMID 30719401.
Puttaswamy G, Narayana S, Mohiyuddin SA. The efficacy and safety of intramuscular aceclofenac and diclofenac for managing postoperative pain in patients undergoing composite resection for oral cancer. J Cancer Metastasis Treat. 2023;9:7. doi: 10.20517/2394-4722.2022.84.
Patino MA, Ramirez RE, Perez CA, Feng L, Kataria P, Myers J. The impact of intraoperative opioid use on survival after oral cancer surgery. Oral Oncol. 2017 Nov;74:1-7. doi: 10.1016/j.oraloncology.2017.09.006, PMID 29103736.
Ali AR, Sakr SA, Ahmed Shawky MA, Rahman. Bilateral sphenopalatine ganglion block as adjuvant to general anesthesia during endoscopic trans-nasal resection of pituitary adenoma. Egypt J Anaesth. 2010;26(4):273-80. doi: 10.1016/j.egja.2010.05.002.
Nader A, Kendall MC, De Oliveria GS, Chen JQ, Vanderby B, Rosenow JM. Ultrasound-guided trigeminal nerve block via the pterygopalatine fossa: an effective treatment for trigeminal neuralgia and atypical facial pain. Pain Physician. 2013 Sep-Oct;16(5):E537-45. PMID 24077204.
Ghosh S, Desai S, Dhakad V, Joseph B, Saldanha E, Patel D. Intraoperative blood loss in head and neck cancer operations: a retrospective observational study in a tertiary care surgical oncology center in central India. Natl J Med Res. 2020;10(3):153-6.
Misk M, Alshawadfy A, Lamei M, Khames F, Abd Elgawad M, Hendawy HA. Evaluating the perioperative analgesic effect of ultrasound-guided trigeminal nerve block in adult patients undergoing maxillofacial surgery under general anesthesia: a randomized controlled study. Egypt J Anaesth. 2023;39(1):598-604. doi: 10.1080/11101849.2023.2238529.