
Int J Curr Pharm Res, Vol 18, Issue 1, 69-71Original Article
COMPARATIVE EVALUATION OF CNS DEPRESSIVE ACTIVITY OF CHLORPHENIRAMINE MALEATE AND EBASTINE IN WISTAR RATS USING ROTARAD AND PHOTOACTOMETER
LENIN R., UMAMAHESHWARI C., RAJARAM G.*
Department of Pharmacology, Govt Villupuram Medical College, Villupuram, Tamilnadu, India
*Corresponding author: Rajaram G.; *Email: gvmcpharm@gmail.com
Received: 06 Oct 2025, Revised and Accepted: 26 Nov 2025
ABSTRACT
Objective: Allergic conditions are commonly treated with Antihistamines. Though the first-generation H1 antihistamines like Chlorpheniramine Maleate (CPM) are effective, they can cause sedation because of its blood-brain barrier crossing capacity leading to central nervous system (CNS) depression. But Ebastine, a 2nd-generation H1 antihistamine, is considered to be a non-sedating drug because of its limited CNS penetration. Hence, evaluating the CNS effects of these drugs is crucial for guiding safe and rational clinical practice. Aim of this study was to compare the CNS depressant effects of Chlorpheniramine Maleate and Ebastine in Wistar rats using Rotarod and Photoactometer behavioral models.
Methods: This was a prospective cross-sectional study conducted at Department of Pharmacology, Government Villupuram Medical College and Hospital, Villupuram, Tamilnadu, India between January to June 2025. Eighteen adult Wistar rats were randomly assigned into three groups, with 6 rats in each group viz, Group 1: Control rats receiving Normal saline, Group 2: rats receiving CPM and Group 3: rats receiving Ebastine. CNS activity was assessed 60 min after drug administration. Motor coordination was measured using the Rotarod test and locomotor activity was assessed using a digital photoactometer. Data were analyzed using one-way ANOVA followed by Tukey’s post hoc test.
Results: Chlorpheniramine Maleate significantly reduced the fall-off time on the Rotarod (71.6±7.8 sec) and decreased locomotor activity (124.2±14.8 counts), indicating CNS depression (p<0.01). Ebastine-treated rats showed no significant difference in either parameter compared to the control group (fall-off time: 139.7±12.6 sec; locomotor activity: 284.4±19.2 counts).
Conclusion: Chlorpheniramine Maleate produces significant CNS depressant activity while Ebastine does not impair motor coordination or spontaneous activity in rats. These findings support the evidence that Ebastine as a non-sedating antihistamine and favor its use in clinical practice where sedation is undesirable.
Keywords: Chlorpheniramine maleate, CNS depression, Ebastine, Photoactometer, Rotarod, Wistar rats
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i1.8030 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Histamine plays an important role in mediating allergic responses and it primarily acts through four receptor subtypes (H1 to H4). Among these, the H1 receptor is mainly responsible for allergic manifestations like rhinitis, urticaria and conjunctivitis. H1 receptor antagonists, commonly referred to as antihistamines, play a crucial role in the management of such conditions [1].
These antihistamines are broadly classified into first-generation and second-generation drugs primarily based on their pharmacological profiles, particularly their ability to cross the blood-brain barrier (BBB) and induce central nervous system (CNS) effects. First-generation antihistamines, such as Chlorpheniramine Maleate (CPM), are highly lipophilic which enables them to penetrate the CNS, leading to sedation, drowsiness, impaired psychomotor performance, and cognitive dulling [2]. These adverse effects limit their use in individuals involved in activities requiring mental alertness, such as driving or operating machinery [3].
In contrast, second-generation antihistamines like Ebastine have been developed mainly to minimize these CNS-related side effects. Due to high H1 receptor selectivity, low lipophilicity and poor BBB penetration it acts as non-sedating antihistamine. It is metabolized in the liver to an active metabolite, carebastine, which retains peripheral H1 antagonistic activity while lacking significant central depressant effects [4].
Evaluating the CNS effects of antihistamines is essential, especially in preclinical studies involving animals. Behavioral models such as the Rotarod test assess motor coordination and balance, which are often affected by CNS depressants, while the Photoactometer measures spontaneous locomotor activity, an indirect index of CNS excitability or depression [5]. Both these methods are simple, reliable and widely accepted methods for preliminary screening of neuropharmacological effects of drugs.
Although both CPM and Ebastine are effective in treating allergic conditions, their CNS profiles differ significantly. Hence, this study was undertaken to comparatively evaluate the CNS depressant activity of chlorpheniramine maleate and ebastine in adult wistar rats using rotarod and photoactometer models. The results aim to clarify a better understanding of their CNS safety profiles and support rational clinical decision-making, especially when sedation is a concern.
MATERIALS AND METHODS
This was a randomized controlled experimental study conducted at the Department of Pharmacology, Government Villupuram Medical College and Hospital, Villupuram, Tamilnadu, India between January and June 2025. The study evaluated and compared the CNS depressant activity of CPM and Ebastine in adult Wistar rats using behavioral models. The study followed the guidelines outlined by the Committee for the Control and Supervision of Experiments on Animals (CCSEA).
A total of 18 healthy adult wistar albino rats of either sex, weighing 150–200 g, were procured and housed in the central animal house under standard laboratory conditions viz, Temperature 22±2 °C, Humidity 50–60%, Light/dark cycle 12 h/12 h and a standard pellet diet and water provided ad libitum. The animals were acclimatized to laboratory conditions for 7 days before the experiment. The study protocol was reviewed and approved by the Institutional Animal Ethics Committee (IAEC) (Approval No: GVMC/2 IAEC/2024/01)
The drugs (CPM and Ebastine) were suspended in normal saline while Normal saline (0.9% NaCl) was used a vehicle control. All drugs were administered orally at a dose of 10 mg/kg body weight, based on literature-reported effective sedative/non-sedative doses in rats. Rats were divided into three groups of six animals (n=6) each with Group 1 (Control) receiving Normal saline, Group 2 receiving CPM (10 ml/kg, oral) and Group 3 receiving Ebastine (10 ml/kg, oral). All animals were tested 60 min after drug administration for CNS activity using Rotarod and Photoactometer. The sample size (n=6 per group) was chosen based on prior similar pharmacological studies and guidelines for behavioral experiments involving rodents [6].
Behavioral assessment
Rotarod test (Motor coordination)
The Rotarod apparatus consists of a horizontal rod (diameter 7 cm) rotating at 20 revolutions per minute (rpm). Rats were pre-trained to remain on the rod for at least 180 seconds during three consecutive training trials. On the day of the experiment, after drug administration and a 60-minute waiting period, each rat was placed individually on the rotating rod, and latency to fall (in seconds) was recorded as the fall-off time. Three trials were conducted per rat with a 5-minute interval between trials, and the average time was taken for analysis. A decrease in fall-off time indicates motor incoordination or CNS depression [7].
Photoactometer test (Locomotor activity)
Spontaneous locomotor activity was measured using a digital photoactometer which consists of an enclosed square arena with photocell beams across each wall. Interruptions of these light beams by the animal's movement were digitally recorded as counts. Each rat was placed individually in the arena for 10 min, and the total activity counts were recorded. The apparatus was cleaned with 70% alcohol between animals to remove any odour cues. A reduction in activity count is indicative of sedative or CNS depressant activity [8].
Statistical analysis
All experimental data were compiled in Microsoft Excel and expressed as mean±standard error of the mean (SEM) for each group. The data obtained from behavioral tests (Rotarod and Photoactometer) were subjected to one-way analysis of variance (ANOVA) to determine whether there were statistically significant differences among the treatment groups. If ANOVA showed significance (p<0.05), it was followed by Tukey’s Honestly Significant Difference (HSD) post hoc test to perform pairwise comparisons between individual groups. This test was chosen because it controls for Type I error across multiple comparisons and is appropriate when sample sizes are equal, as in this study (n = 6 per group) [9].
All statistical analyses were conducted using GraphPad Prism version 10.0 (GraphPad Software Inc., USA). A p-value of less than 0.05 was considered statistically significant, while values<0.01 were considered highly significant.
RESULTS
In the present study, the CNS activity of Chlorpheniramine Maleate (CPM) and Ebastine was assessed using two behavioral models—Rotarod test for motor coordination and Photoactometer test for spontaneous locomotor activity-in wistar rats.
Rotarod test (Motor coordination)
The mean fall-off time on the Rotarod for the control group was 145.8±9.4 seconds, indicating normal motor coordination. Rats treated with CPM (10 mg/kg, oral) showed a significant reduction in fall-off time to 71.6±7.8 sec (p<0.01 vs control), suggesting impaired motor coordination. In contrast, rats treated with Ebastine (10 mg/kg, oral) showed a mean fall-off time of 139.7±12.6 sec, which was not significantly different from the control group (p>0.05), indicating that Ebastine does not impair motor coordination.
Photoactometer test (Locomotor activity)
The spontaneous locomotor activity in the control group was 298.5± 17.4 counts/10 min. Rats administered CPM showed a highly significant decrease in activity to 124.2±14.8 counts (p<0.01 vs control), indicating decreased locomotor activity. Rats treated with Ebastine exhibited 284.4±19.2 counts, which was not significantly different from the control group (p>0.05), confirming that Ebastine does not significantly affect spontaneous motor activity.
One-way ANOVA showed significant differences among the groups for both fall-off time and locomotor activity (p<0.001). Tukey’s post hoc test revealed that CPM differed significantly from control in both tests, while Ebastine did not, confirming that only CPM induced measurable CNS depression under the experimental conditions (table 1). Statistical analysis was done using One-way ANOVA followed by Tukey’s post hoc test.
Table 1: Comparison of effect of chlorpheniramine maleate and ebastine on rotarod performance and locomotor activity in wistar rats
| Group | Rotarod fall-off time (in seconds, Mean±SEM) | Photoactometer activity (in counts/10 min, Mean±SEM) | p-value (vs control) |
| Control (Normal saline) | 145.8±9.4 | 298.5± 17.4 | |
| CPM (10 mg/kg, oral) ** | 71.6±7.8 | 124.2±14.8 | p<0.01 |
| Ebastine (10 mg/kg, oral) | 139.7±12.6 | 284.4±19.2 | P>0.05 |
(Value are expressed as mean±SEM; n = 6 rats per group)
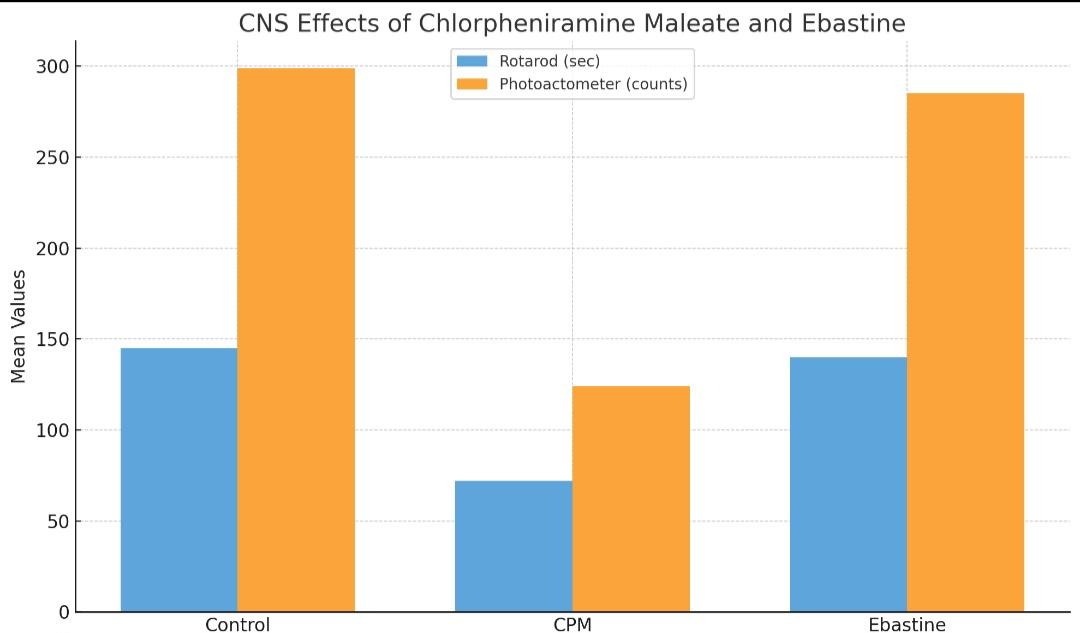
Fig. 1: Comparison of CNS effects of CPM and ebastine in wistar rats using rotarod and photoactometer tests
DISCUSSION
The present study was undertaken to evaluate and compare the central nervous system (CNS) depressant effects of two antihistamines-Chlorpheniramine Maleate (CPM), a first-generation antihistamine and Ebastine, a second-generation antihistamine, using validated behavioral models in Wistar rats. The results demonstrated that CPM treated rats showed significantly impaired motor coordination and reduced spontaneous locomotor activity, indicating CNS depression. In contrast, Ebastine treated rats did not produce such effects, indicating its lack of sedative action.
The Rotarod test is a widely accepted method for assessing motor coordination and balance, which are typically compromised in the presence of CNS-depressant agents [10]. In this study, rats treated with CPM exhibited a marked reduction in fall-off time, suggesting sedation and impaired neuromuscular coordination. This finding is consistent with the well-known pharmacological profile of first-generation antihistamines, which are highly lipophilic, allowing them to readily cross the blood-brain barrier (BBB) and interact with central H1 receptors, leading to sedation, drowsiness, and impaired psychomotor performance [2].
In contrast, Ebastinetreated animals, maintained motor coordination comparable to the control group, as reflected by the non-significant change in fall-off time. This observation aligns with the known non-sedating nature of Ebastine, attributed to its low lipid solubility, high selectivity for peripheral H1 receptors, and extensive first-pass metabolism to its active metabolite, carebastine, which does not significantly penetrate the CNS [11].
The Photoactometer test assesses spontaneous locomotor activity, a reliable indirect marker of CNS stimulation or depression. CPM produced a statistically significant decrease in activity counts, confirming its CNS depressant effects. This is consistent with previous studies where CPM and similar agents like diphenhydramine significantly reduced locomotor activity in rodent models [7].
Ebastine, however, did not significantly affect locomotor activity, indicating an absence of CNS depression, which is desirable in clinical practice. Its lack of sedative effect is particularly beneficial in populations such as drivers, students and machine operators, where mental alertness is critical. Multiple clinical trials and post-marketing surveillance data have further confirmed Ebastine's favorable CNS safety profile [12].
The findings in the present study were similar to those observed in the studies by Tagawa M et al. and Shamil KS et al. [13, 14]. These findings support the rational clinical use of second-generation antihistamines like Ebastine over first-generation agents when sedation is to be avoided. However, CPM may still be useful in conditions where night-time sedation is beneficial, such as allergic rhinitis with sleep disturbances.
Limitations of the study was that it was conducted in animal models; caution is needed when extrapolating to humans. Only a single dose of each drug was tested; dose-response relationships were not evaluated. Long-term CNS effects were not assessed.
CONCLUSION
The study concludes that Chlorpheniramine Maleate significantly impairs motor coordination and reduces locomotor activity, indicating CNS depressant effects. In contrast, Ebastine does not exhibits such changes, confirming its non-sedating profile. These results support the clinical preference for second generation antihistamines like Ebastine in situations where sedation is undesirable.
ACKNOWLEDGMENT
The authors gratefully acknowledge the support of the Government Villupuram Medical College, Villupuram particularly the Department of Pharmacology, for providing the necessary facilities to conduct this study. We also thank the technical staff and animal house personnel for their assistance in the care and handling of experimental animals.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
All authors have contributed equally
CONFLICTS OF INTERESTS
Declared none
REFERENCES
Simons FE. Advances in H1-antihistamines. N Engl J Med. 2004;351(21):2203-17. doi: 10.1056/NEJMra033121, PMID 15548781.
Church MK, Maurer M, Simons FE, Bindslev Jensen C, Van Cauwenberge P, Bousquet J. Risk of first-generation H(1)-antihistamines: a GA(2)LEN position paper. Allergy. 2010;65(4):459-66. doi: 10.1111/j.1398-9995.2009.02325.x, PMID 20146728.
Kawauchi H, Yanai K, Wang DY, Itahashi K, Okubo K. Antihistamines for allergic rhinitis treatment from the viewpoint of nonsedative properties. Int J Mol Sci. 2019 Jan;20(1):213. doi: 10.3390/ijms20010213, PMID 30626077.
Noveck RJ, Preston RA, Swan SK. Pharmacokinetics and safety of ebastine in healthy subjects and patients with renal impairment. Clin Pharmacokinet. 2007;46(6):525-34. doi: 10.2165/00003088-200746060-00006, PMID 17518511.
Simons FE. Comparative pharmacology of H1 antihistamines: clinical relevance. Am J Med. 2002;113(Suppl 9A):38S-46S. doi: 10.1016/S0002-9343(02)01436-5, PMID 12517581.
Festing MF, Altman DG. Guidelines for the design and statistical analysis of experiments using laboratory animals. ILAR J. 2002;43(4):244-58. doi: 10.1093/ilar.43.4.244, PMID 12391400.
Vogel HG. Drug discovery and evaluation: pharmacological assays. 3rd ed. Springer; 2008.
Sharma HL, Sharma KK. Principles of pharmacology. 2nd ed. Paras Medical Publisher; 2013.
Zar JH. Biostatistical Analysis. 5th ed. Upper Saddle River (NJ): Prentice-Hall/Pearson Education; 2010.
Kulkarni SK. Handbook of experimental pharmacology. 4th ed. New Delhi: Vallabh Prakashan; 2012.
Yanai K, Yoshikawa T, Yanai A, Nakamura T, Iida T, Leurs R. The clinical pharmacology of non-sedating antihistamines. Pharmacol Ther. 2017 Oct;178:148-56. doi: 10.1016/j.pharmthera.2017.04.004, PMID 28457804.
Sastre J. Ebastine in the treatment of allergic rhinitis and urticaria: 30 y of clinical studies and real-world experience. J Investig Allergol Clin Immunol. 2020;30(3):156-68. doi: 10.18176/jiaci.0401, PMID 30977465.
Tagawa M, Kano M, Okamura N, Higuchi M, Matsuda M, Mizuki Y. Differential cognitive effects of ebastine and (+)-chlorpheniramine in healthy subjects: correlation between cognitive impairment and plasma drug concentration. Br J Clin Pharmacol. 2002;53(3):296-304. doi: 10.1046/j.0306-5251.2001.01183.x, PMID 11874393.
Shamil KS, Prakruti PP, Anuradha MG, Bela JS, Chetna KD. Old versus new antihistamines: effects on cognition and psychomotor functions. J Fam Med Prim Care. 2022 Oct;11(10):5909-17. doi: 10.4103/jfmpc.jfmpc_77_22, PMID 36618177.