
Int J Curr Pharm Res, Vol 18, Issue 1, 72-75Original Article
COMBINED BCL-2–KI-67 SCORE AND ITS CORRELATION WITH TUMOUR INFILTRATING LYMPHOCYTES IN ENDOMETRIAL CARCINOMA – A NOVEL STUDY
K. T. ATHULYA KRISHNA KUMAR1*, KRISHNARAJ UPADHYAYA2
1Department of Pathology, Chamrajnagar Institute of Medical Sciences, Chamrajnagar, Karnataka, India
2Department of Pathology, Yenepoya Medical College, Mangaluru, Karnataka, India
*Corresponding author: K. T. Athulya Krishna Kumar; *Email: athunambiar156@gmail.com
Received: 06 Oct 2025, Revised and Accepted: 26 Nov 2025
ABSTRACT
Objective: This study aimed to evaluate a combined Bcl-2–Ki-67 score and assess its correlation with TILs in endometrioid endometrial carcinoma.
Methods: A retrospective analysis was performed on 40 cases of endometrioid endometrial carcinoma, from January 2017 to June 2018. Immunohistochemically staining for Bcl-2 and Ki-67 was performed, and a combined score was generated by inverting the Bcl-2 score and summing with the Ki-67 score. TILs were evaluated as low (<10%), moderate (20–40%), and high (>50%). Correlation between the combined score and TILs was analyzed using Fisher’s exact test, and interobserver agreement for TIL assessment was calculated using the Kappa coefficient.
Results: Combined Bcl-2–Ki-67 scores ranged from 0–6. A significant inverse correlation was observed between the combined score and TILs (p=0.00105, Fisher’s exact test). Interobserver agreement for TIL scoring was high (κ = 0.82).
Conclusion: The combined Bcl-2–Ki-67 score inversely correlates with TIL levels in endometrioid endometrial carcinoma, highlighting its potential use as a prognostic marker. Further studies with larger cohorts are warranted to validate these findings.
Keywords: Bcl-2, Ki-67, Tumor Infiltrating Lymphocytes, Endometrial Carcinoma, Immunohistochemistry.
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i1.8032 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Endometrial carcinoma is the second most common malignancy of the female genital tract in developing countries [1, 2]. Type I endometrioid carcinomas represent approximately 80% of cases and are usually hormone-dependent, low-grade tumors with a favorable prognosis, whereas Type II carcinomas are high-grade and aggressive in nature [3, 4].
Bcl-2 plays a pivotal role in regulating apoptosis [4]. Normal endometrium exhibits cyclic Bcl-2 expression, peaking during the proliferative phase and decreasing in the secretory phase [5, 6]. Dysregulation of Bcl-2 is implicated in tumorigenesis, with studies in endometrial carcinoma showing an inverse relationship with Bcl-2 expression and increased tumour grade [7-10].
Ki-67 is a nuclear protein expressed in proliferating cells and serves as a robust marker of cellular proliferation [11]. Elevated Ki-67 expression correlates with higher tumor grade and poorer prognosis in endometrial carcinoma [11-13].
Tumor-infiltrating lymphocytes (TILs) reflect host anti-tumor immune response and have prognostic significance in multiple cancers, including endometrial carcinoma [13]. However, studies correlating Bcl-2 and Ki-67 expression with TIL density remain scarce.
In this study, we aimed to develop a combined Bcl-2–Ki-67 score and analyze its relationship with TILs in endometrioid endometrial carcinoma.
MATERIALS AND METHODS
This retrospective study was conducted at a tertiary care center from January 2017 to June 2018 after obtaining institutional ethical clearance. Forty cases of endometrioid endometrial carcinoma diagnosed on hysterectomy specimens were included. Non-endometrioid and non-epithelial tumors were excluded.
Immunohistochemistry was performed using Bcl-2 (Clone 124, DAKO) and Ki-67 (Clone Mib-1, DAKO). Tonsil tissue was used as a positive control. Bcl-2 expression was scored by intensity of cytoplasmic expression (0: absent, 1: weak, 2: moderate, 3: strong) [14]. Ki-67 expression was calculated by using percentage positivity of cells (0:<5%, 1+: 5–25%, 2+: 25–50%, 3+: 50–100%) [15].
The combined score was calculated by inverting the Bcl-2 score (Score 3 → 0, Score 2 → 1, Score 1 → 2, Score 0 → 3) and summing it with the Ki-67 score, producing a range of 0–6. We further categorized these as Low (Combined score: 0–2), Moderate (Combined Score: 3–4), and High (Combined Score: 5–6).
TILs were independently assessed by two pathologists under ×200 objective. Scoring was: low (<10%), moderate (20–40%), and high (>50%) [13]. Due to few number of cases, for the purpose of analysis we formed two main groups of tumour infiltrating lymphocyte percentage, wherein low and moderate groups were taken as one, and high percentage as a separate group.
Percentage of tumour-infiltrating lymphocytes were calculated only within the tumour mass, and areas showing necrosis or endometrial hyperplasia were excluded [13]. Interobserver agreement was assessed using kappa coefficient. Fisher’s exact test evaluated the correlation between combined score and TIL levels. P<0.05 was considered statistically significant.
RESULTS
In the 40 cases of endometrioid endometrial carcinoma, the combined Bcl-2/Ki-67 score ranged from 0 to 6. Categorizing the scores, 16 cases (40%) fell in the low category (0–2), 8 cases (20%) in the intermediate category (3–4), and 16 cases (40%) in the high category (5–6). Tumour-infiltrating lymphocytes (TILs) were independently evaluated by two pathologists, showing substantial interobserver agreement (Cohen’s κ = 0.82).
The relationship between the combined score and TILs is summarized in table 1. Tumors with low combined scores (0–2) were predominantly associated with high TIL infiltration (16 of 16 cases), whereas high combined scores (5–6) were more frequently associated with low TIL density (8 of 16 cases) (fig. 1–4). Fisher’s exact test indicated a statistically significant association between combined score and TIL category (p = 0.00105), suggesting that tumors with higher proliferative/apoptotic imbalance tend to have lower immune infiltration.
The combined score also demonstrated a clear relationship with tumor grade. Low combined scores (0–2) were mostly observed in Grade 1 tumors (14 of 16 cases), whereas high scores (5–6) were predominantly found in Grade 2 and 3 tumors (10 of 16 cases). Intermediate scores (3–4) were evenly distributed between low-and high-grade tumors (6 cases each). Fisher’s exact test confirmed a statistically significant association between combined score and tumor grade (p = 0.00011), indicating that a higher combined immunoscore correlates with higher tumor grade in endometrioid endometrial carcinoma.
Table 1: Correlation of combined Bcl-2/Ki-67 score with tumor-infiltrating lymphocytes (TILs)
| Combined score category | No. of cases (n) | TIL-(Low and moderate) (n) | TIL–high (n) |
| 0–2 (low) | 16 | 02 | 16 |
| 3–4 (intermediate) | 08 | 06 | 06 |
| 5–6 (high) | 16 | 08 | 02 |
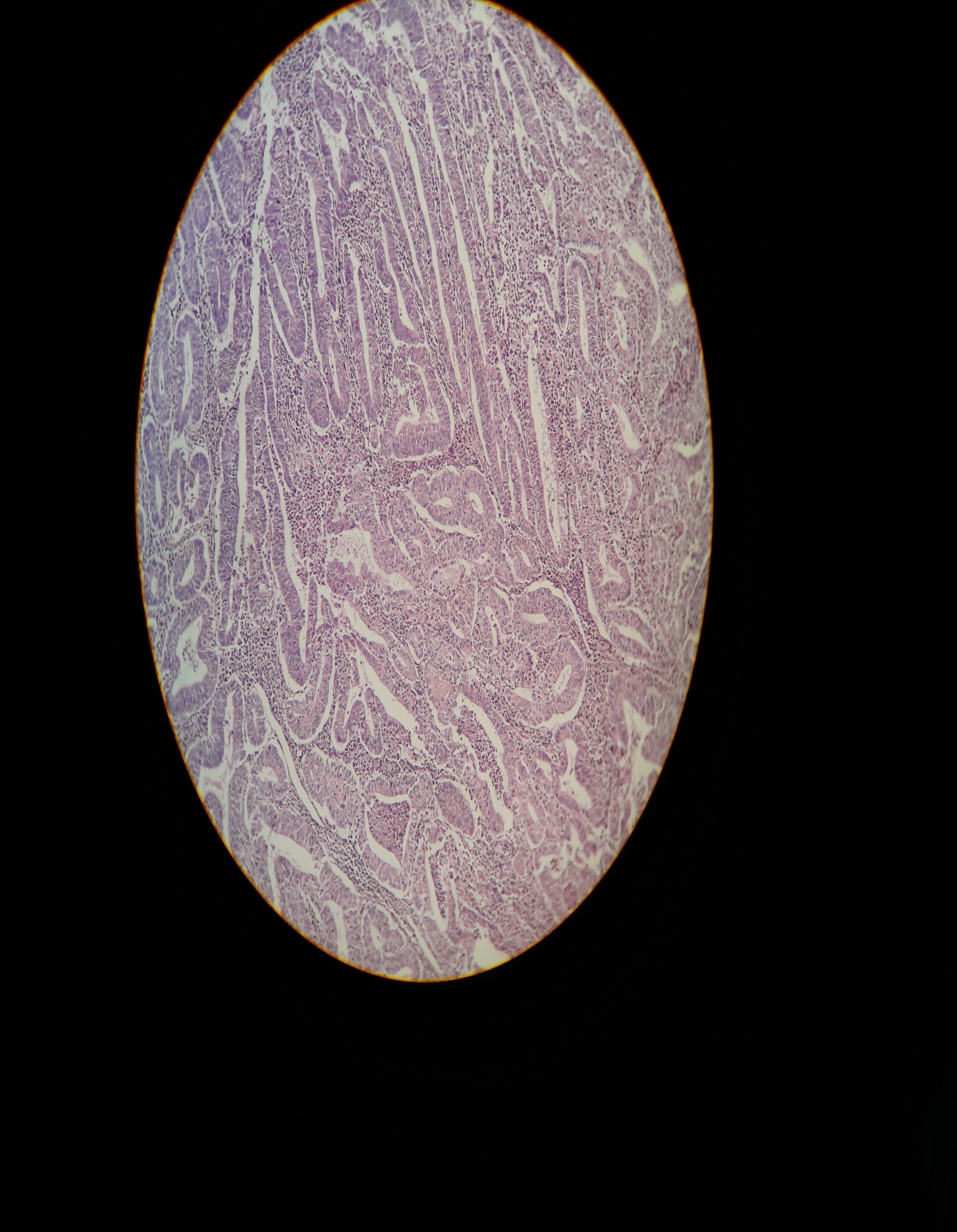
Fig. 1: Photomicrograph showing high TIL (>50%) in a case of Endometrioid endometrial carcinoma (HandE, 20x)
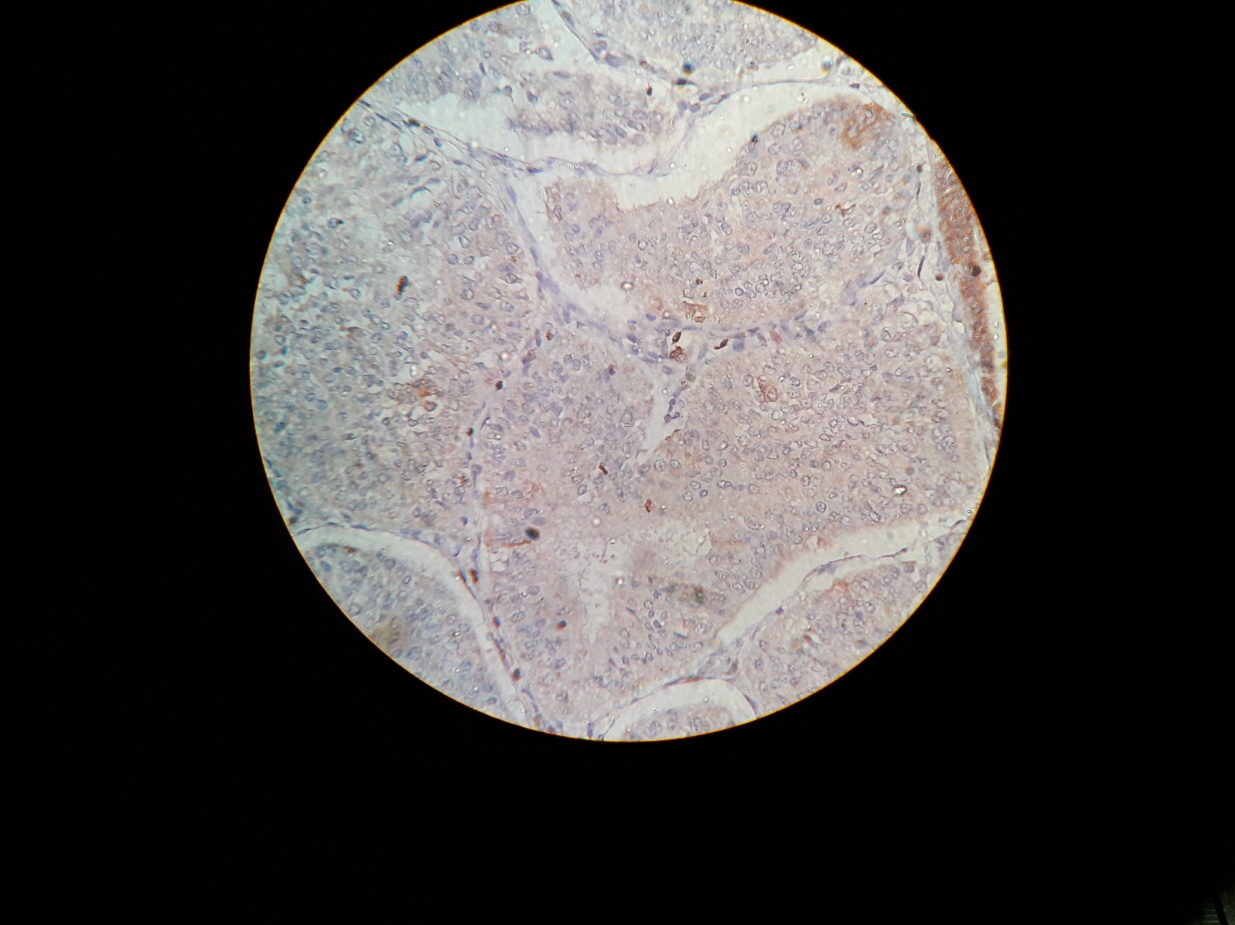
Fig. 2: Photomicrograph of Bcl-2 score 1 staining (weak intensity) in endometrioid endometrial carcinoma (Bcl-2, 40x)
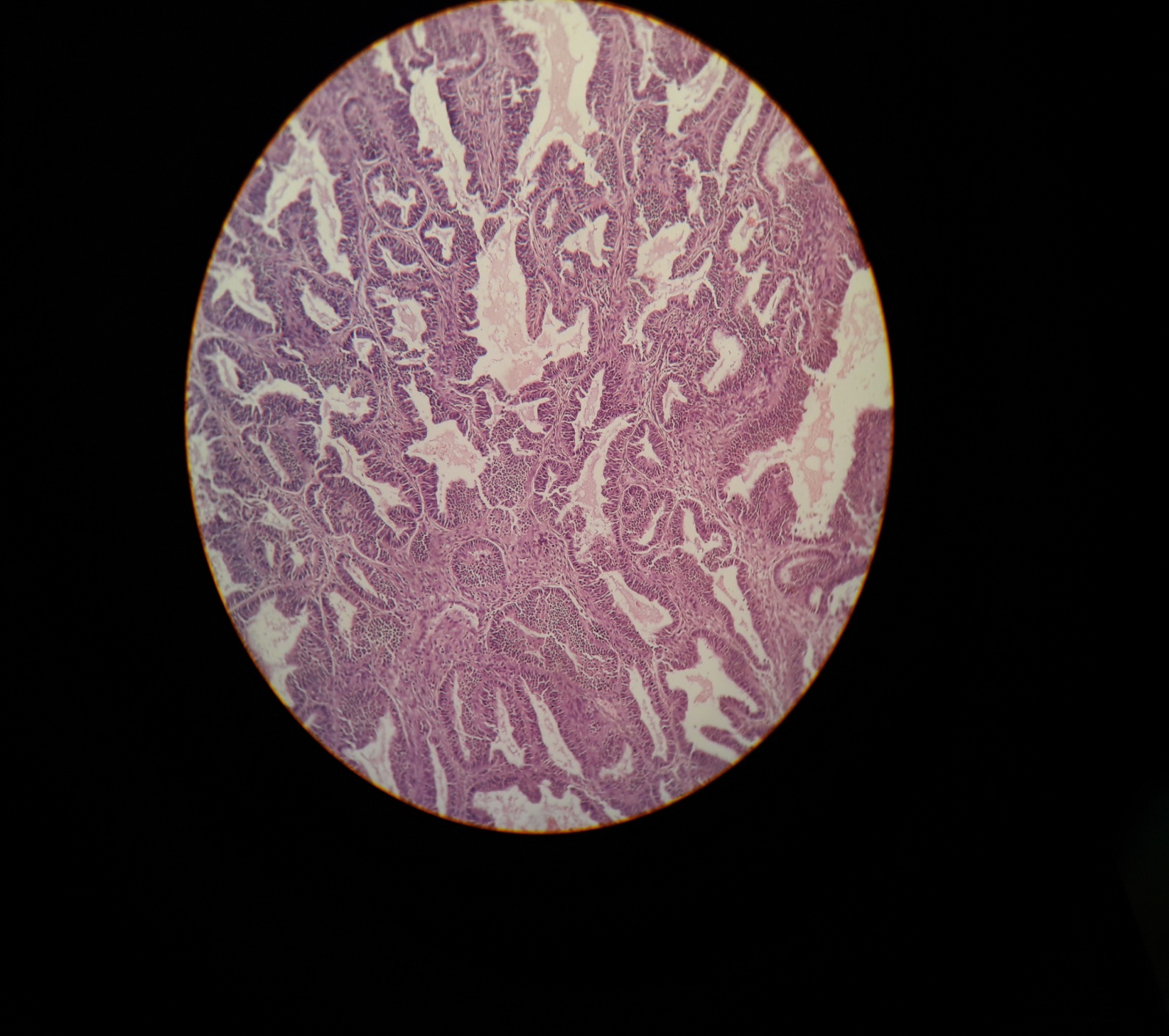
Fig. 3: Photomicrograph showing low TIL (<10%) in a case of Endometrioid endometrial carcinoma (HandE, 20x)
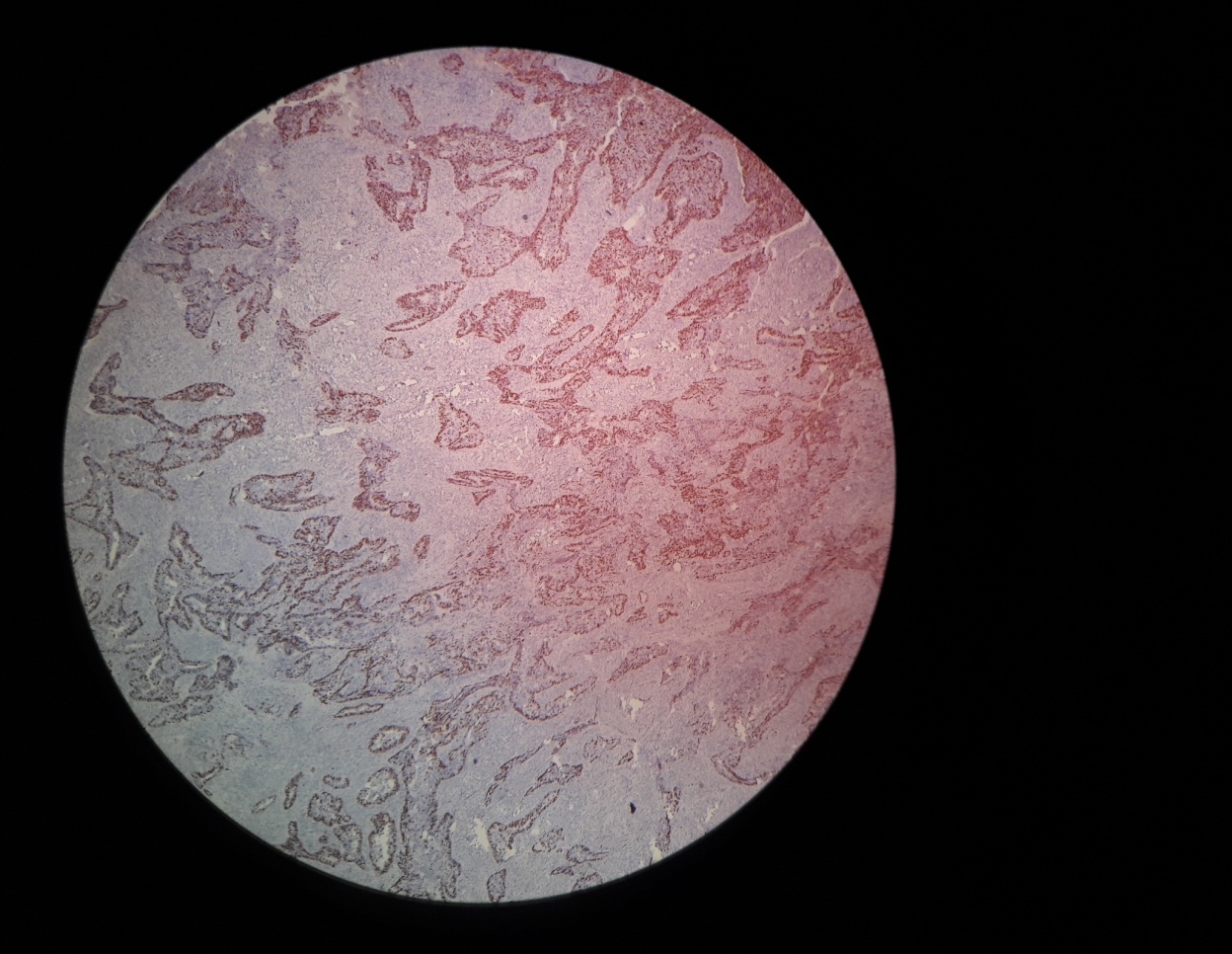
Fig. 4: Photomicrograph of Ki 67 (3+) staining in endometrioid endometrial carcinoma (Ki-67, 4x)
DISCUSSION
This study demonstrates a significant correlation between a combined Bcl-2–Ki-67 score and both tumor-infiltrating lymphocytes (TILs) and tumor grade in endometrioid endometrial carcinoma. Low combined scores were frequently associated with high TIL infiltration, suggesting a robust anti-tumor immune response, whereas high scores correlated with reduced TILs and higher tumor grade, indicating aggressive tumor biology.
Bcl-2 downregulation combined with high Ki-67 expression reflects diminished apoptotic control and increased proliferation, which aligns with previous reports on individual markers [12, 15]. High TIL density has been shown to correlate with favorable outcomes in various malignancies, including endometrial carcinoma [13]. The substantial interobserver agreement in TIL scoring (κ = 0.82) supports the reliability of this evaluation as a reproducible prognostic measure [13].
Previous studies have documented the prognostic significance of Bcl-2 in endometrial carcinoma. Studies have shown that Bcl-2 expression is variably associated with outcomes and may hold prognostic value [5, 6]. Similarly, Bcl-2 expression may play a role in early tumor development and progression [5].
Ki-67 has been established as a key proliferation marker with prognostic relevance. Analyses reported that higher Ki-67 is associated with worse cancer-specific survival and correlates with more aggressive features [12]. Increased Ki-67 expression may be driven by deregulation of cell cycle pathways, including PI3K/Akt and MAPK signaling [13].
Integrating Bcl-2 and Ki-67 into a combined score allows simultaneous assessment of anti-apoptotic activity and proliferative activity in a tumor, providing a more comprehensive evaluation of tumor aggressiveness. The observed inverse correlation between combined score and TIL density aligns with studies indicating that rapidly proliferating, apoptosis-resistant tumors often exhibit immune evasion [13].
The correlation of higher combined scores with higher tumor grade supports its potential as a prognostic indicator, as tumor grade is a key predictor of clinical outcome [13]. These findings suggest that a combined Bcl-2–Ki-67 immunoscore could help stratify patients for more aggressive therapy or closer follow-up.
While our study is limited by a small sample size, it highlights the relevance of combining apoptosis and proliferation markers in the context of the tumor immune microenvironment. Future studies with larger cohorts are needed to validate the utility of this combined score and its predictive value for therapy response.
CONCLUSION
The combined Bcl-2–Ki-67 score inversely correlates with TIL density in endometrioid endometrial carcinoma. This novel scoring system may serve as a prognostic biomarker and help identify tumors with low immune infiltration and aggressive behavior.
ACKNOWLEDGEMENT
None
FUNDING
Nil
AUTHORS CONTRIBUTIONS
All authors have contributed equally
CONFLICTS OF INTERESTS
Declared none
REFERENCES
Srikantia N, Rekha B, Rajeev AG, Kalyan SN. Endometrioid endometrial adenocarcinoma in a premenopausal woman with multiple organ metastases. Indian J Med Paediatr Oncol. 2009;30(2):80-3. doi: 10.4103/0971-5851.60053, PMID 20596308.
Njoku K, Barr CE, Crosbie EJ. Current and emerging prognostic biomarkers in endometrial cancer. Front Oncol. 2022;12:890908. doi: 10.3389/fonc.2022.890908, PMID 35530346.
Kurman RJ, Carcangiu ML, Herrington CS, Young RH. WHO classification of tumours of female reproductive organs. 4th ed. Lyon: IARC; 2014. doi: 10.1016/B978-92-832-2431-5.00001-6.
Coll-de la Rubia E, Martinez Garcia E, Dittmar G, Gil-Moreno A, Cabrera S, Colas E. Prognostic biomarkers in endometrial cancer: a systematic review and meta-analysis. J Clin Med. 2020;9(6):1900. doi: 10.3390/jcm9061900, PMID 32560580.
Krishna Kumar K, Upadhyaya K, Cn RT. Bcl-2 may contribute to evolution of endometrial hyperplasia, but it isn’t a factor in subsequent carcinogenesis. Arch Razi Inst. 2024;79(4):827-32. doi: 10.32592/ARI.2024.79.4.827, PMID 40256590.
Kandaswamy S, Palanisamy P. Immunohistochemical expression of BCL-2 in endometrial carcinoma and its comparison with hormone receptor status and epidermal growth factor. Cureus. 2023;15(1):e33346. doi: 10.7759/cureus.33346, PMID 36751174.
Laban M, Ibrahim EA, Agur W, Elddin Ahmed AM. Bcl-2 may play a role in the progression of endometrial hyperplasia and early carcinogenesis, but not linked to further tumorigenesis☆. J Microsc Ultrastruct. 2015;3(1):19-24. doi: 10.1016/j.jmau.2014.11.001, PMID 30023177.
Zannoni GF, Bragantini E, Castiglione F, Fassan M, Troncone G, Inzani F. Current prognostic and predictive biomarkers for endometrial cancer in clinical practice: recommendations/proposal from the Italian study group. Front Oncol. 2022 Apr 8;12:805613. doi: 10.3389/fonc.2022.805613, PMID 35463299.
Rashidi B, Rad JS, Rad LR. Immunohistochemical (Ki-67) study of endometrial maturation in mice after use of phosphodiesterase type 5 inhibitor. Adv Biomed Res. 2015;4:154. doi: 10.4103/2277-9175.161581, PMID 26380239.
Bohiltea RE, Sajin M, Furtunescu F, Bohiltea LC, Mihart A, Baros A. Clinical and pathological correlations in endometrial pathology. J Med Life. 2015 Oct-Dec;8(4):552-62. PMID 26664489.
Jia M, Pi J, Zou J, Feng M, Chen H, Lin C. The potential value of Ki-67 in prognostic classification in early low-risk endometrial cancer. Cancer Control. 2023;30:10732748231206929. doi: 10.1177/10732748231206929, PMID 37924202.
Kitson S, Sivalingam VN, Bolton J, McVey R, Nickkho Amiry M, Powell ME. Ki-67 in endometrial cancer: scoring optimization and prognostic relevance for window studies. Mod Pathol. 2017;30(3):459-68. doi: 10.1038/modpathol.2016.203, PMID 27910946.
Ozturk C, Askan G, Ozturk SD, Okcu O, Sen B, Bedir R. High tumor infiltrating lymphocytes are associated with overall survival and good prognostic parameters in endometrial endometrioid carcinoma patients. Turk Patoloji Derg. 2023;39(1):75-82. doi: 10.5146/tjpath.2022.01596, PMID 36441005.
Rogers PA, Lederman F, Plunkett D, Affandi B. Bcl-2, Fas and caspase 3 expression in endometrium from levonorgestrel implant users with and without breakthrough bleeding. Hum Reprod. 2000;15(Suppl 3):152-61. doi: 10.1093/humrep/15.suppl_3.152, PMID 11041231.
Sakuragi N, Ohkouchi T, Hareyama H, Ikeda K, Watari H, Fujimoto T. Bcl-2 expression and prognosis of patients with endometrial carcinoma. Int J Cancer. 1998 Apr 17;79(2):153-8. doi: 10.1002/(SICI)1097-0215(19980417)79:2<153::AID-IJC10>3.0.CO;2-B, PMID 9583730.