
Int J Curr Pharm Res, Vol 18, Issue 1, 88-92Original Article
AN ETIOLOGICAL, CLINICAL PROFILE AND OUTCOME OF PATIENT WITH ACUTE FEBRILE ILLNESS IN TERTIARY CARE CENTRE
MOHIT KUMAR CHANDRAKAR*, RAKESH SINGH JAGAT, NIKHIL GUPTA, SIMMI DUBE
Department of General Medicine, Gandhi Medical College, Bhopal, India
*Corresponding author: Mohit Kumar Chandrakar; *Email: mohit17chandrakar@gmail.com
Received: 06 Oct 2025, Revised and Accepted: 26 Nov 2025
ABSTRACT
Objective: Acute Febrile Illness (AFI) represents a diagnostic and therapeutic challenge, especially in tropical regions where diverse infectious agents coexist. Prompt identification of etiology is essential to mitigate complications such as encephalopathy, shock, and multiorgan failure. We aimed to identify clinical features and classify the etiologies of AFI among adult patients admitted to a tertiary care hospital and assess their outcomes.
Methods: This hospital-based cross-sectional study was conducted from October 2023 to April 2025 at Gandhi Medical College and Hamidia Hospital, Bhopal. Adults (18–60 years) presenting with fever<2 w duration were enrolled. Patients underwent comprehensive clinical evaluation and diagnostic investigations, including serological, microbiological, and radiological tests. Outcomes were recorded as recovery with/without diagnosis or death.
Results: Among 319 patients, most were aged 21–30 years (31.9%) and male (60.5%). Common symptoms included myalgia, headache, malaise, and gastrointestinal complaints. The most frequent etiologies were dengue (26.6%), pneumonia (20.1%), and viral hepatitis A (10.3%). Neurological involvement was observed in meningitis cases (9.4%), with viral meningoencephalitis being the most prevalent subtype. Overall recovery was 90.3%, with 5.6% mortality-predominantly in pneumonia and undiagnosed viral fever cases. Etiology was significantly associated with patient outcomes (p=0.0001).
Conclusion: Dengue and pneumonia were leading causes of AFI. Rapid diagnosis and management are critical, especially for CNS and respiratory involvement. Strengthening diagnostic infrastructure and early intervention strategies may reduce AFI-related mortality in resource-limited settings.
Keywords: Febrile illness, Dengue, Malaria, Outcome, Mortality
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i1.8041 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Acute febrile illness (AFI) encompasses a spectrum of clinical syndromes marked by an elevated body temperature of short duration-typically less than two weeks-and represents a leading cause of hospitalization among both children and adults [1, 2]. Despite continual progress in diagnostic technology, AFI persists as a major public health problem worldwide, particularly in tropical and subtropical regions where a wide array of infectious agents coexist [3]. Patients generally present with nonspecific complaints such as fever, headache, and malaise, but may rapidly progress to severe conditions including encephalopathy, shock, multi-organ dysfunction, or profound thrombocytopenia [2, 4].
Children with acute fever are especially susceptible to severe disease and unfavorable outcomes, largely due to their immature immune systems and the potential for swift clinical deterioration [5, 7]. Pediatric patients frequently exhibit high-grade fever accompanied by seizures or neurological signs, including encephalopathy and meningoencephalitis [7, 8]. Early differentiation between central nervous system (CNS) infections-such as viral encephalitis, bacterial meningitis, or cerebral malaria-and self-limiting febrile illnesses is essential to prevent long-term neurological complications and reduce mortality [8].
Adults with AFI also present a complex diagnostic challenge due to their varied comorbidities, immune status, and broad spectrum of possible etiologies [2, 9]. While classic tropical infections such as malaria and dengue are common, other diseases-including leptospirosis, scrub typhus, and emerging zoonotic infections-can present with similar nonspecific febrile symptoms [2]. In some adults, AFI may manifest as acute febrile encephalopathy, pancreatitis, or acute-on-chronic liver failure secondary to infectious triggers [10].
Neurological manifestations grouped under the term “acute febrile encephalopathy” are of particular concern due to their association with significant morbidity and mortality [8, 11]. Common etiologies include viral encephalitis (notably Japanese encephalitis and herpes simplex encephalitis), cerebral malaria, acute bacterial meningitis, and rickettsial infections, which often present with altered sensorium, seizures, focal deficits, or coma [8, 11]. Delayed recognition and treatment frequently result in irreversible neurological damage. Multiple hospital-based studies from India have highlighted the importance of incorporating rapid polymerase chain reaction (PCR) assays for herpes simplex virus and region-specific serological tests for Japanese encephalitis, dengue, and scrub typhus into diagnostic protocols [8]. However, significant gaps persist in determining optimal empirical treatment strategies in regions with high rates of antimicrobial resistance.
The present study aims to identify and categorize the etiological agents responsible for AFI among patients admitted to a tertiary care hospital and to characterize the clinical and laboratory profiles associated with each category. Additionally, we sought to evaluate the relationship between etiological diagnosis, clinical outcomes, and mortality.
MATERIALS AND METHODS
This hospital-based cross-sectional study was conducted in the Department of Medicine, Gandhi Medical College and associated Hamidia Hospital, Bhopal, over an 18 mo period from October 2023 to April 2025.
Study population
All patients aged 18 to 60 y admitted with a history of fever (≥38 °C) of less than two weeks’ duration and who provided written informed consent were included in the study. Patients were excluded if they had received antibiotic therapy for more than three days prior to admission, had known chronic comorbid conditions (such as chronic kidney disease or chronic liver disease), or presented with non-infectious causes of fever, including autoimmune disorders, malignancies, or ongoing immunosuppressive therapy.
Ethical considerations
Ethical approval was obtained from the Institutional Ethics Committee prior to the commencement of the study. Written informed consent was taken from all participants before enrolment.
Data collection and clinical evaluation
Each eligible participant underwent a detailed history and thorough physical examination using a pretested structured proforma. Baseline and disease-specific investigations were performed as per clinical judgment.
Investigations
Routine hematological and biochemical tests included complete blood count (CBC), random blood sugar, serum electrolytes, liver function tests (LFT), renal function tests (RFT), erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), lactate dehydrogenase (LDH) or ferritin, and coagulation profile (PT/INR/APTT). Microbiological investigations comprised blood culture, urine routine examination and culture, peripheral smear and rapid antigen tests for malaria, Widal test, dengue NS1 antigen and IgM/IgG antibodies, chikungunya IgM, HIV ELISA, and other relevant viral markers as indicated. Imaging modalities included chest radiography, abdominal ultrasonography, and neuroimaging (non-contrast CT or MRI of the brain) when clinically warranted. Specialized tests such as cerebrospinal fluid (CSF) analysis in suspected meningoencephalitis, D-dimer, arterial blood gas (ABG) analysis, total creatine phosphokinase (CPK), and CK-MB levels were performed when indicated. All findings were systematically documented.
Follow-up and outcome assessment
Patients were monitored throughout their hospital stay until discharge or death. Outcomes were categorized as:
Diagnosed and recovered
Undiagnosed and recovered
Died
Complications such as acute respiratory distress syndrome (ARDS), shock, or organ failure were recorded prospectively. Based on final diagnostic confirmation, patients were grouped into etiological categories, including malaria, typhoid, dengue, chikungunya, acute viral hepatitis, Japanese encephalitis, HIV infection, meningitis, and other confirmed causes.
Statistical analysis
Data were compiled using Microsoft Excel and analyzed with IBM SPSS Statistics version 25.0. Categorical variables were expressed as frequencies and percentages. Associations between categorical variables were tested using the Chi-square test or Fisher’s exact test, as appropriate. A p-value of <0.05 was considered statistically significant.
RESULTS
This study was conducted on a total of 319 cases presenting with acute febrile illness at our study area.
Table 1: Distribution of cases according to baseline variables
| Baseline variables | Number of cases (n=319) | Percentage |
| Age (years) | ≤20 y | 46 |
| 21-30 y | 102 | |
| 31-40 y | 57 | |
| 41-50 y | 63 | |
| 51-60 y | 46 | |
| >60 y | 5 | |
| Gender | Female | 126 |
| Male | 193 |
Of the 319 cases analysed, nearly one-third (31.97 %) were aged 21– 30 y, making this the most represented age band. Also, we observed a male predominance in our study (60.50%) (table 1).
Table 2: Distribution of cases according to clinical symptoms
| Clinical symptoms | Number of cases (n=319) | Percentage |
| Headache, loss of appetite, malaise, myalgia | 85 | 26.6 |
| Cough with fever | 1 | .3 |
| Myalgia | 17 | 5.3 |
| Myalgia, Headache, fever | 13 | 4.1 |
| Rash, joint pain, jaundice, abdominal pain | 1 | .3 |
| Myalgia, fever, sore throat, nausea, vomiting | 64 | 20.1 |
| Fever, headache, altered sensorium | 30 | 9.4 |
| Diarrhea, Myalgia, jaundice, nausea, vomiting | 33 | 10.3 |
| Rash, Jaundice, nausea, vomiting | 30 | 9.4 |
| Myalgia, abdominal pain, increased urine frequency | 19 | 6.0 |
| Respiratory Distress, Rash | 16 | 5.0 |
| Diarrhea, abdominal pain, nausea, vomiting, jaundice | 8 | 2.5 |
| Jaundice, Respiratory Distress, Myalgia, black Escher on left shoulder | 2 | 0.6 |
The most frequently reported cluster of symptoms was headache, loss of appetite, malaise, and myalgia (26.6%), followed by myalgia, fever, sore throat, nausea, and vomiting (20.1%) (table 3).
Table 3: Distribution of cases according to etiology
| Etiology | Number of cases (n=319) | Percentage |
| Dengue fever with NS1 positive | 85 | 26.6 |
| Acute pancreatitis | 1 | 0.3 |
| Viral Fever | 17 | 5.3 |
| Cerebral venous sinus thrombosis | 13 | 4.1 |
| Dengue fever IgM+with HAV acute viral hepatitis | 1 | 0.3 |
| Pneumonia | 64 | 20.1 |
| Meningitis | 30 | 9.4 |
| Acute Viral Hepatitis: HAV IgM positive | 33 | 10.3 |
| Enteric Fever (Typhoid) | 30 | 9.4 |
| Urinary tract infection E. coli | 19 | 6.0 |
| Malaria (P. falciparum) | 16 | 5.0 |
| Leptospirosis | 8 | 2.5 |
| Scrub Typhus | 2 | 0.6 |
The most common etiology of acute febrile illness was dengue fever (26.6%), followed by pneumonia (20.1%). About 30 cases had meningitis, of them, 14 cases had viral meningoencephalitis, 10 cases had acute bacterial meningitis and 6 cases had tubercular meningitis (table 3).
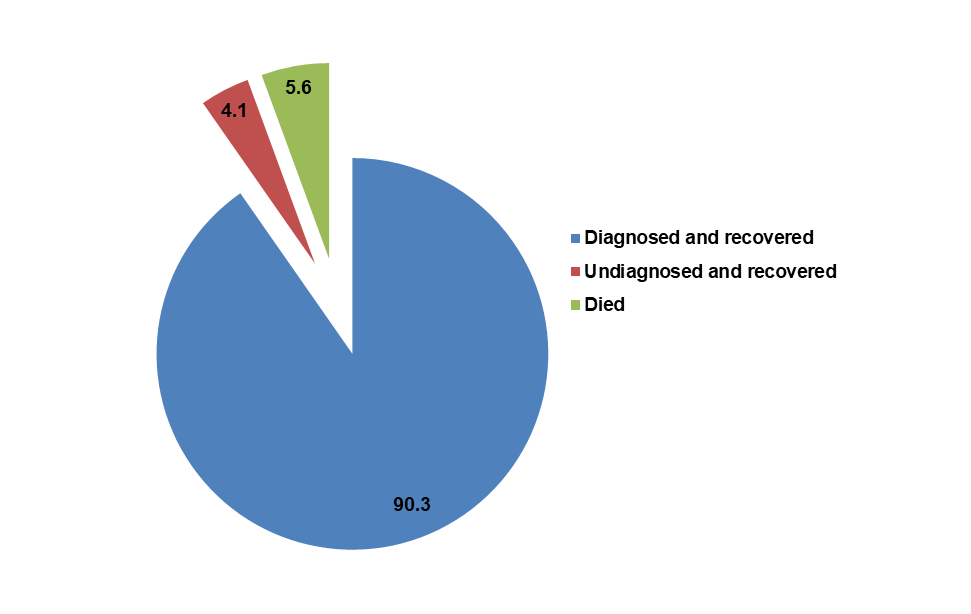
Fig. 1: Distribution of cases according to clinical outcome, the majority (288, 90.3%) were diagnosed and recovered from their illness, whereas 13 individuals (4.1%) remained undiagnosed yet still recovered. Eighteen patients (5.6%) died in our study (fig. 1)
Table 4: Association of etiology with outcome
| Etiology | Outcome | Total | ||
| Diagnosed and recovered | Undiagnosed and recovered | Died | ||
| Dengue fever with NS1 positive | 82 (96.5%) | 0 (0) | 3 (3.5%) | 85 |
| Acute pancreatitis | 1 (100%) | 0 (0) | 0 (0) | 1 |
| Viral Fever | 6 (35.3%) | 9 (52.9%) | 2 (11.8%) | 17 |
| Cerebral venous sinus thrombosis | 12 (92.3%) | 0 (0) | 1 (7.7%) | 13 |
| Dengue fever IgM+with HAV acute viral hepatitis | 1 (100%) | 0 (0) | 0 (0) | 1 |
| Pneumonia | 52 (81.3%) | 4 (6.3%) | 8 (12.5%) | 64 |
| Meningitis | 29 (96.7%) | 0 (0) | 1 (3.3%) | 30 |
| Acute Viral Hepatitis: HAV IgM positive | 33 (100%) | 0 (0) | 0 (0) | 33 |
| Enteric Fever (Typhoid) | 28 (93.3%) | 0 (0) | 2 (6.7%) | 30 |
| Urinary tract infection E. coli | 19 (100%) | 0 (0) | 0 (0) | 19 |
| Malaria (P. falciparum) | 15 (93.8%) | 0 (0) | 1 (6.3%) | 16 |
| Leptospirosis | 8 (100%) | 0 (0) | 0 (0) | 8 |
| Scrub Typhus | 2 (100%) | 0 (0) | 0 (0) | 2 |
| P value | 0.0001 | |||
As observed from table 4, significantly higher proportions of patients with dengue fever, with or without anti-HAV, acute viral hepatitis, acute pancreatitis, UTI, leptospirosis, scrub typhus, cerebral venous sinus thrombosis, meningitis, typhoid fever and malaria were diagnosed and recovered, whereas 52.9% cases with viral fever remained undiagnosed but recovered. On the other hand, we documented mortality in 12.5% cases with pneumonia, and 11.8% cases with viral fever. The observed association of etiology with outcome was statistically significant (p<0.05).
DISCUSSION
Acute febrile illness (AFI) continues to present a diagnostic and therapeutic challenge in tropical regions, where multiple pathogens can produce similar clinical pictures and timely intervention is critical for optimal outcomes. In our adult cohort, nearly two-thirds of patients were under 40 y of age, with the largest subgroup belonging to the 21–30-year range, and males predominated. This demographic distribution suggests that occupational exposure, lifestyle factors, and gender-based disparities in healthcare access contribute to disease risk and patterns of hospital presentation.
The most frequently reported symptom cluster comprised headache, loss of appetite, malaise, and myalgia (26.6%), followed by myalgia, sore throat, nausea, and vomiting (20.1%). Gastrointestinal manifestations were also prominent, indicating that AFI can mimic a wide spectrum of clinical presentations. These findings are consistent with those of Gundikeri et al. [12] while Patil et al. [13] similarly highlighted the diversity of symptom profiles, especially in leptospirosis and other zoonotic infections. The marked clinical overlap across infections underscores the diagnostic difficulty inherent in managing undifferentiated fevers. Possible reasons include shared environmental exposures (e. g., contaminated water or vectors), delayed healthcare-seeking that blurs early disease-specific features, and co-infections in endemic regions. Regional ecological variations may also explain why certain hallmark features-such as rash or eschar-were less common in our cohort.
Dengue fever (26.6%) emerged as the most frequently confirmed aetiology, followed by pneumonia (20.1%) and acute viral hepatitis A (10.3%). A smaller proportion of cases was attributed to enteric fever, urinary tract infection, malaria, leptospirosis, and other causes. These results align with the findings of Prasad et al. [14] and Veligandla et al. [15] who reported dengue as a leading cause of febrile admissions in tropical hospitals, reflecting its endemicity. Geographic and seasonal factors likely account for inter-study variations, as monsoon rainfall promotes mosquito breeding and dengue transmission, while overcrowded housing may facilitate pneumonia outbreaks. In contrast, diseases such as scrub typhus or leptospirosis are closely linked to agricultural practices and local ecology. Moreover, diagnostic capacity-such as availability of rapid antigen or serologic testing-directly influences the recognition and reporting of specific infections. Thus, ongoing surveillance and refinement of diagnostic algorithms remain essential for accurately mapping the aetiological spectrum of AFI.
Among patients with meningitis or meningoencephalitis (n = 30), viral meningoencephalitis (46.7%) was the most common, followed by acute bacterial meningitis (33.3%) and tubercular meningitis (20%). Chrispal et al. [2] emphasised the importance of differentiating tubercular from other forms of meningitis, as the clinical course and management differ substantially. Similarly, Singhi et al. [16] highlighted viral meningoencephalitis as a major contributor to neurological involvement in AFI, particularly in tropical areas where arboviral infections are prevalent. Our findings reinforce the diagnostic complexity of CNS infections, which often present with overlapping features such as fever with rigors, altered sensorium, or seizures. The lack of advanced diagnostic tools in resource-limited settings further complicates aetiological confirmation. Variations in meningitis aetiology may stem from local immunisation practices, HIV prevalence, or ecological factors promoting vector-borne viral spread. Strengthening diagnostic capacity through polymerase chain reaction (PCR) assays and improved culture techniques could enable more precise identification. Early recognition of CNS involvement remains vital to prompt therapy and the prevention of complications, morbidity, and mortality.
In our study, 90.3% of patients were successfully diagnosed and recovered, while 4.1% recovered without a definitive diagnosis and 5.6% succumbed. This recovery rate mirrors the multicentre data of Singhi et al. [16] and Ray et al. [9] likewise observed that most patients recovered, with mortality concentrated among those with multiorgan dysfunction or delayed hospital presentation. The 5.6% mortality in our cohort reflects the severity of certain infections-particularly pneumonia, severe dengue, and sepsis-and highlights the impact of delayed referral, comorbidities, and suboptimal early management. Regional variations in case-fatality rates likely reflect disparities in healthcare access and infrastructure, as well as differences in pathogen virulence. The subset of undiagnosed yet recovered patients underscores the diagnostic limitations of resource-limited settings, where specialised tests are not universally available. Enhancing laboratory infrastructure and adopting standardised work-up protocols could help reduce the burden of undiagnosed febrile illnesses.
Disease-specific outcomes further illuminate this pattern. Dengue fever, acute viral hepatitis, and urinary tract infections were associated with high recovery rates following confirmed diagnosis and appropriate management, whereas pneumonia and certain unclassified viral fevers contributed disproportionately to mortality. Similar observations were made by Prasad et al. [15] and Kigozi et al. [17] who identified pneumonia and viral fevers as leading causes of death in AFI cases with severe systemic involvement. In our cohort, pneumonia accounted for the largest proportion of deaths (12.5% among pneumonia cases), primarily due to complications such as acute respiratory distress syndrome (ARDS) and sepsis. Conversely, the high rate of undiagnosed recoveries, especially in mild viral syndromes, may reflect self-limiting infections or the effectiveness of supportive care.
Nevertheless, the findings should be interpreted in light of certain limitations. Being a single-centre, hospital-based study, referral bias is inevitable, as milder community cases or those unable to access tertiary care were likely under-represented. The diagnostic repertoire was restricted to tests routinely available in our laboratory, so true-positive cases of dengue, leptospirosis, rickettsial infection, or viral encephalitis may have been missed if patients presented outside the optimal diagnostic window.
CONCLUSION
This hospital-based analysis delineates the epidemiological profile of AFI in our region-dominated by dengue fever, community-acquired pneumonia, and a range of other tropical infections that share overlapping clinical features but distinct risk determinants. Over 90% of patients recovered following accurate diagnosis and timely evidence-based therapy; however, mortality clustered among older individuals and those with prolonged fever or rapid physiological deterioration (e. g., dengue-associated encephalitis, pneumonia with ARDS). These results underscore the dual need for robust community-level vector control and public-health interventions to prevent infection, alongside hospital-based investments in rapid diagnostics, early warning systems, and critical-care readiness to improve outcomes in acute febrile illnesses.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
All authors have contributed equally
CONFLICT OF INTERESTS
Declared none
REFERENCES
Mondal R, Misra S, Datta S, Gupta D, Das MK. Clinical and etiological profile of acute febrile illness in children admitted to a Tertiary Care Hospital in Eastern India. SVU-International Journal of Medical Sciences. 2024;7(2):1008-15. doi: 10.21608/svuijm.2023.177415.1458.
Chrispal A, Boorugu H, Gopinath KG, Chandy S, Prakash JA, Thomas EM. Acute undifferentiated febrile illness in adult hospitalized patients: the disease spectrum and diagnostic predictors an experience from a Tertiary Care Hospital in South India. Trop Doct. 2010 Oct;40(4):230-4. doi: 10.1258/td.2010.100132, PMID 20870680.
Gundikeri SK, Dhananjaya M. Acute undifferentiated febrile illness-clinical spectrum and outcome from a Tertiary Care Teaching Hospital of North Karnataka. Int J Biol Med Res. 2013;4(2):3399-402.
Shukla AK, Kishore K, Goel J, Sengupta S. Clinical profile of febrile encephalopathy patients at a tertiary care hospital in India: a retrospective study. J Acute Dis. 2023;12(4):145-50. doi: 10.4103/2221-6189.385681.
Dasari R, Reddy KS, Nimmagadda A, Kuruvada SS, Sneha N, Srikrishna S. Clinico-etiological profile of acute undifferentiated fever in children 6 mo-15 y. IP International Journal of Medical Paediatrics and Oncology. 2021;7(1):1-5. doi: 10.18231/j.ijmpo.2021.001.
Biswas R, Basu K, Tripathi I, Roy SK. A study on etiology, clinical profile and outcome of acute febrile encephalopathy in children: a prospective study at a Tertiary Care Center of Eastern India. Asian J Med Sci. 2021 Apr 1;12(4):86-91. doi: 10.3126/ajms.v12i4.35153.
Rauf A, Singhi S, Nallasamy K, Walia M, Ray P. Non-respiratory and non-diarrheal causes of acute febrile illnesses in children requiring hospitalization in a Tertiary Care Hospital in North India: a prospective study. Am J Trop Med Hyg. 2018 Jul 9;99(3):783-8. doi: 10.4269/ajtmh.18-0056, PMID 29988003.
Ary KA, Singh H, Suri V, Sharma K, Biswal M, Singh MP. Changing clinical profile and predictors of mortality in patients of acute febrile encephalopathy from North India. J Glob Infect Dis. 2023 Jul 1;15(3):101-7. doi: 10.4103/jgid.jgid_18_23, PMID 37800084.
Ray A, Mohta S, Soneja M, Jadon R, Wig N, Sood R. Clinical spectrum and outcome of critically ill hospitalized patients with acute febrile illness and new-onset organ dysfunction presenting during monsoon season. Drug Discov Ther. 2019 Apr 30;13(2):101-7. doi: 10.5582/ddt.2019.01023, PMID 31080200.
Ramu R, Paul V, Devipriya S, Philip NC. Etiology, clinical profile and outcome of acute pancreatitis in a Tertiary Care Teaching Hospital in Rural South India: a ten-year retrospective study. Int Surg J. 2019 Sep 26;6(10):3794-9. doi: 10.18203/2349-2902.isj20194443.
Rawat CS, Dubey TN, Saxena RS. Study of clinico-etiological profile of acute febrile encephalopathy. Int J Med Res. 2019;4(2):123-6.
Gundikeri SK, Dhananjaya M. Acute undifferentiated febrile illness-clinical spectrum and outcome from a Tertiary Care Teaching Hospital of North Karnataka. Int J Biol Med Res. 2013;4(2):3399-402.
Patil H, Agrawal V, Patil V. Clinical profile and outcome of leptospirosis at Tertiary Care Centre in Western Maharashtra. J Acad Med Sci. 2012;2(1):30. doi: 10.4103/2249-4855.104013.
Prasad N, Murdoch DR, Reyburn H, Crump JA. Etiology of severe febrile illness in low and middle-income countries: a systematic review. PLOS One. 2015 Jun 30;10(6):e0127962. doi: 10.1371/journal.pone.0127962, PMID 26126200.
Veligandla G, Vanan E, Padmavathi E, Bhaskar M. Etiological spectrum and prevalence of acute undifferentiated febrile illness (AUFI) in fever cases attending our tertiary care centre. Int J Curr Microbiol App Sci. 2017;6(5):954-62. doi: 10.20546/ijcmas.2017.605.105.
Singhi S, Rungta N, Nallasamy K, Bhalla A, Peter JV, Chaudhary D. Tropical fevers in Indian intensive care units: a prospective multicenter study. Indian J Crit Care Med. 2017;21(12):811-8. doi: 10.4103/ijccm.IJCCM_324_17, PMID 29307960.
Kigozi BK, Kharod GA, Bukenya H, Shadomy SV, Haberling DL, Stoddard RA. Investigating the etiology of acute febrile illness: a prospective clinic-based study in Uganda. BMC Infect Dis. 2023 Jun 16;23(1):411. doi: 10.1186/s12879-023-08335-4, PMID 37328808.