
Int J Curr Pharm Res, Vol 18, Issue 1, 97-101Original Article
AN OBSERVATIONAL STUDY TO EVALUATE KNOWLEDGE, ATTITUDE, AND PRACTICE OF PHARMACOVIGILANCE AMONG THE HEALTHCARE PROFESSIONALS AND UNDERGRADUATE MEDICAL STUDENTS
JAYDEEP PATEL*, VIPUL CHAUDHARI, VIDHI THAKER
Department of Pharmacology, GCS Medical College, Hospital and Research Centre, Ahmedabad, Gujarat, India
*Corresponding author: Jaydeep Patel; *Email: jpjaydeeppatel5@gmail.com
Received: 06 Oct 2025, Revised and Accepted: 26 Nov 2025
ABSTRACT
Objective: The importance of pharmacovigilance (Pv) is to ensure the safe and rational use of medicines. Targeting healthcare professionals (HCPs) and undergraduate (UG) medical students (visiting hospitals) for sensitization towards pharmacovigilance is the key to implementing the practice of adverse drug reaction (ADR) reporting in clinical practice. Education and training are the most recognized means of improving ADR reporting. The objective was to evaluate the knowledge, attitude, and practices (KAP) of pharmacovigilance among HCPs and UG medical students.
Methods: This was a cross-sectional, observational, questionnaire-based study carried out for a period of four months (September to December 2023). The case record form includes demographic characteristics and a KAP questionnaire that focuses on the reporting of adverse drug reactions (ADRs). A KAP survey was analyzed question-wise, and statistical analysis was performed using Microsoft Excel Office 365.
Results: The results indicated that HCPs demonstrated a higher level of knowledge (78.14%) compared to UG medical students (70.2%). Both groups demonstrated an equally positive attitude towards pharmacovigilance, with a mean score of 93%. However, a significant difference was observed in the practice domain, where HCPs scored 70.9%, which was markedly higher than the 41.68% recorded among UG medical students.
Conclusion: The study reveals that more sensitization programs on pharmacovigilance practice are required in tertiary care hospitals to ensure ADR reporting to the pharmacovigilance centre, which will strengthen the health care setup.
Keywords: Attitude, Healthcare professional, Knowledge, Pharmacovigilance, Practice
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i1.8043 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
According to the World Health Organization (WHO), Pharmacovigilance is, “the science and activities relating to the detection, assessment, understanding and prevention of adverse effects or any other drug-related problem” [1]. The Ministry of Health and Family Welfare (MoHFW), Government of India, launched the Pharmacovigilance Programme of India (PvPI) in 2010 to enhance medicine safety as an integral part of clinical practice. The program aims to build confidence and trust among patients and healthcare professionals regarding drug safety. Since April 2011, the Indian Pharmacopoeia Commission (IPC), under the MoHFW, has served as the National Coordination Centre (NCC) for PvPI, overseeing pharmacovigilance activities nationwide [2]. Spontaneous reporting by healthcare professionals is akey component of this program.
One of the major cause of morbidity and mortality all over the world is adverse drug reactions (ADRs). Hence, proper monitoring of ADRs is a necessity [3]. It is important for healthcare professionals to know how to report and where to report an ADR. Active participation of healthcare professionals in the pharmacovigilance program can improve the ADR reporting [4]. However, the rate of ADR reporting by India is below1% [5]. Hence, it is evident that current status of pharmacovigilance in India is not satisfactory. Education and training are the most recognized means of improving ADR reporting [6]. The best time to inculcate knowledge, attitude, and practice of doctors toward pharmacovigilance is during their undergraduate training, so that medical students can play an important role in ADR reporting activities in the present time as well as in their future.
A knowledge, attitude, and practices (KAP) survey are a quantitative method (predefined questions formatted in standardized questionnaires) that provides access to quantitative information [7]. Hence, this study was planned with an aim to evaluate the knowledge, attitude, and practices of Pharmacovigilance among the healthcare professionals and undergraduate medical students.
MATERIALS AND METHODS
This was a cross-sectional, observational and validated questionnaire based study conducted among undergraduate medical students (pre-final and final year M. B. B. S) and healthcare professionals (doctors, nurses, and pharmacists) working in GCS medical college, hospital and Research Centre (GCSMCH and RC).
Permission was obtained from institutional ethics committee to conduct the study as per the protocol and the study was carried out for a period of 4 mo, i. e., September 2023 and December 2023.
It was a questionnaire-based study(done in google form), which was adopted from previous KAP study on healthcare professionals and undergraduate medical students about ADR reporting [8-14]. Few changes were made according to requirement of present study. The form includes demographic characteristics and a KAP questionnaire that focuses on the reporting of adverse drug reactions (ADRs). This questionnaire consisted of 17 questions in all, seven questions that evaluated Pv and ADR reporting knowledge (Q. No. 1 to 7), five questions to assess participants’ attitudes towards ADR reporting and Pv. (Q. No. 8 to 12) and five questions that identifying the practices of respondents about reporting. (Q. No. 13 to 17) all questions were close-ended and had been designed as multiple-choice questions.
Participants who voluntarily agreed and completed the questionnaire were included in the study. They were asked to fill out the form without any assistance. Those who were not willing to participate or did not submit the questionnaire within stipulated time were excluded. The KAP survey questionnaire was analysed question-wise and their percentage value was calculated with the help of Microsoft excel Office 365 version 2019.
RESULTS
The demographic characteristics of the participants are summarized in fig. 1 and 2. The study included152 doctors, 217 nurses, 19 pharmacists, 63 pre-final year MBBS students and 49 final-year MBBS students. The majority of the participants were nurses (43%) and doctors (30%) (fig. 1). Of the total 500 respondents, 63% were females and 37% were males (fig. 2).
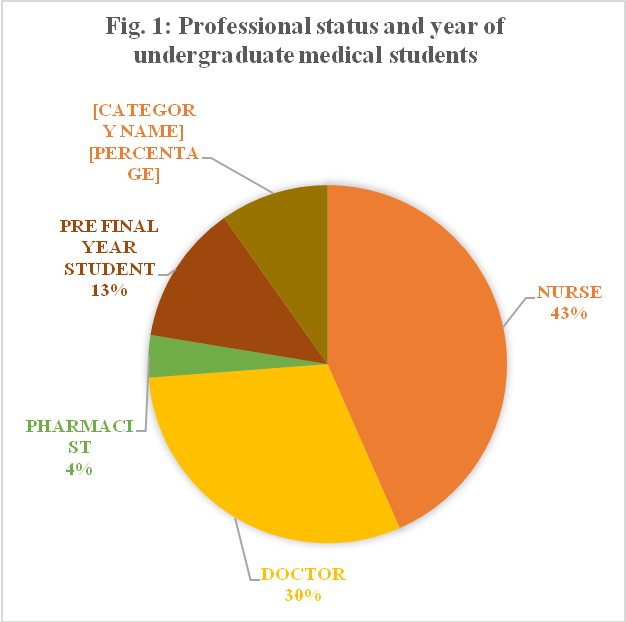
Fig. 1: Professional status and year of undergraduate medical students
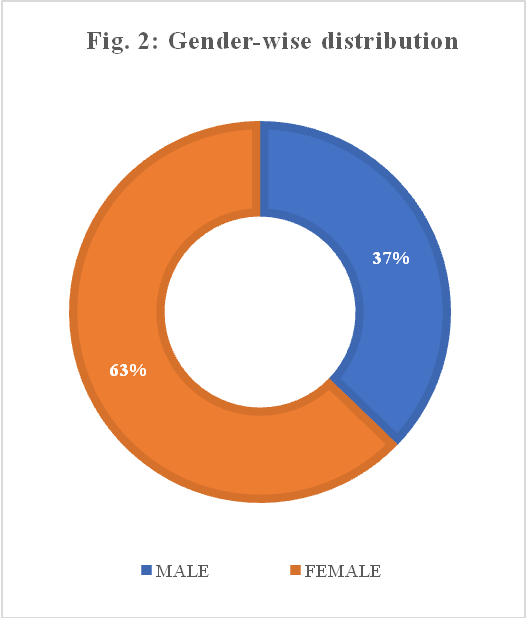
Fig. 2: Gender-wise distribution
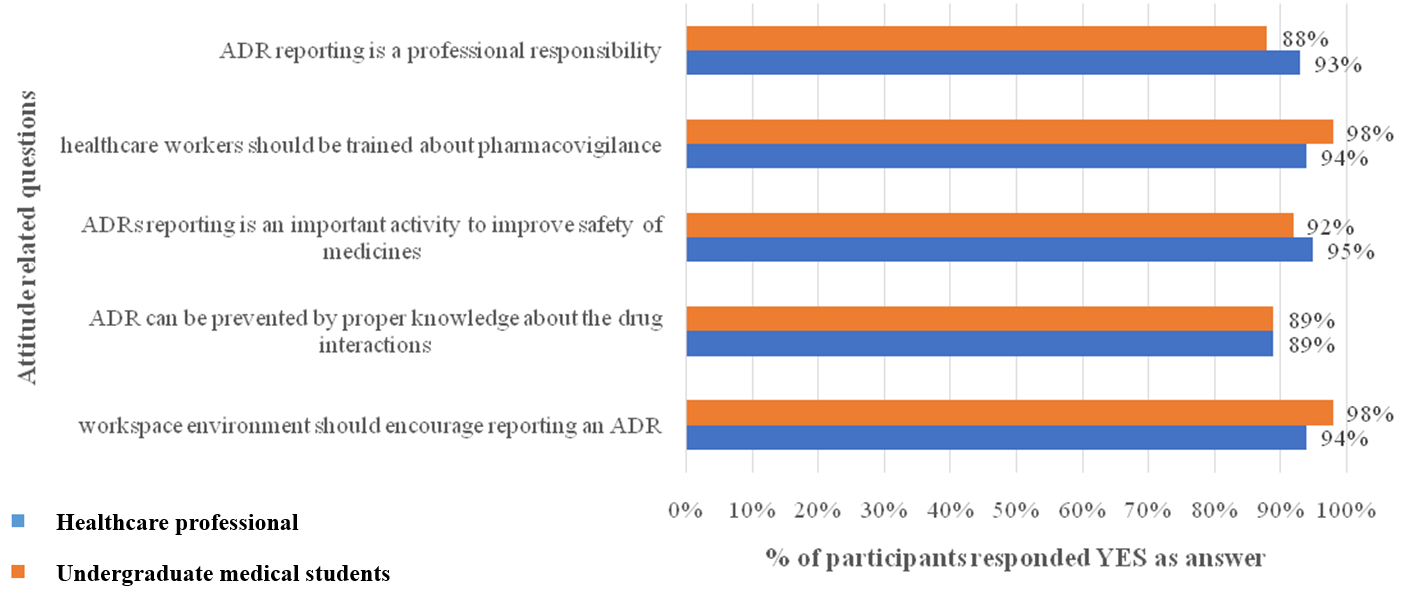
Fig. 3: Response of knowledge based questions among healthcare professionals and undergraduate medical students
Knowledge on pharmacovigilance
Healthcare professionals showed higher knowledge than students regarding the purpose of pharmacovigilance, ADR types, awareness of reporting centres and institutional ADR centres. In contrast, students demonstrated better understanding of serious ADRs (fig. 3).
Attitude on pharmacovigilance
Healthcare professionals and undergraduate medical students demonstrated predominantly positive attitudes toward pharmacovigilance and ADR reporting. The strongest agreement was observed for the necessity of training in pharmacovigilance and the need for a supportive workspace environment for reporting. Both groups widely acknowledged ADR reporting as a professional responsibility and an important activity for improving medicine safety (fig. 4).
Practice on pharmacovigilance
Healthcare professionals reported significantly higher engagement in pharmacovigilance practices, including conducting medication reviews, reporting ADRs to pharmacovigilance centres, reading articles on ADR prevention, and receiving training on ADR reporting. In contrast, a greater proportion of students had seen the ADR reporting form, highlighting a transition from high theoretical exposure among students to high clinical application among professionals (fig. 5).
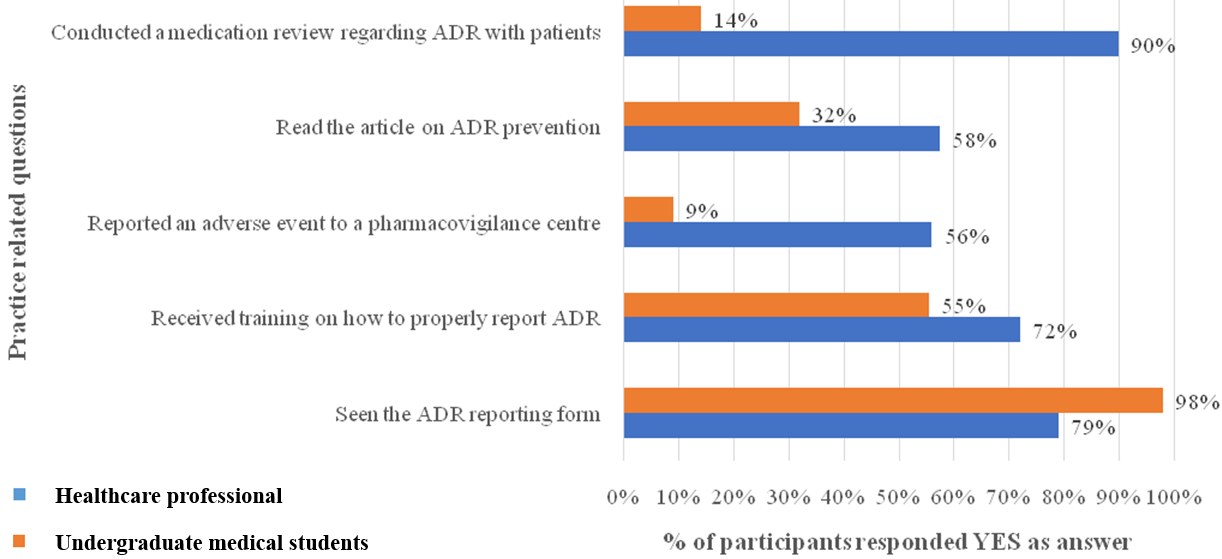
Fig. 4: Response of attitude-based questions among healthcare professional and undergraduate medical students
Table 1: Average score (%) across the questions
| Type of question | HCPs (388) | UG medical students (112) |
| Knowledge-related questions | 78.14 | 70.2 |
| Attitude-related questions | 93 | 93 |
| Practice-related questions | 70.9 | 41.68 |
Healthcare professionals achieved a higher mean score in the knowledge domain (78.1%) compared to undergraduate students (70.2%). Attitude scores were uniformly high in both groups (93%). A significant difference was noted in the practice domain, where healthcare professionals scored 70.9%, compared to 41.7% among students (table 1).
DISCUSSION
In the current era of modern medicine, patient care greatly depends on the rational and evidence-based use of medicines. Pharmacovigilance plays a pivotal role in ensuring drug safety by identifying, assessing, and preventing ADRs.
A high level of awareness regarding the main purpose of pharmacovigilance was observed among HCPs (92%) and UG students (76%). These findings show a improvement compared to those from earlier studies [12, 15–17], which may be attributed to ongoing educational initiatives and institutional efforts to strengthen pharmacovigilance programs. Similarly, awareness regarding the types of ADRs that should be reported was high, with 81% of HCPs and 79% of UG students demonstrating correct knowledge, a finding comparable to previous findings [18, 19]. The greater awareness among HCPs likely due to their direct involvement in patient care and medication monitoring in real-world clinical settings.
A critical component of pharmacovigilance understands of serious adverse drug reactions. Significantly, knowledge about serious ADRs was demonstrated by 97% of UG students, a much higher rate compared to the 84% reported by Bhatt and Kumar [12]. This indicates a strong theoretical understanding of ADR severity among the students. However, a significant gap between theoretical knowledge and practical application becomes evident, as only 61.6% of these students knew where to report ADRs, in contrast to 97% observed by Mahajan et al. [11]. This highlights the importance of case-based and hands-on training sessions to strengthen ADR reporting practices.
A large proportion of participants correctly identified the healthcare personnel responsible for ADR reporting, with 82% of HCPs and 77.6% of UG students demonstrating correct knowledge, findings which were comparable to earlier studies [12, 15, 16, 19]. Awareness of the existence of an ADR Monitoring Centre (AMC) within the institution was observed among 85% of HCPs, which was slightly higher than the proportions reported in previous studies [17, 19, 20]. However, only 60.7% of UG students were aware of such centres, which was markedly lower than the 95% reported by Mahajan et al. [11]. This difference is likely due to the limited exposure of students to institutional pharmacovigilance systems, as their curriculum primarily emphasizes theoretical aspects.
A positive attitude toward ADR reporting was observed, with 93% of HCPs and 88% of UG students considering it a professional responsibility. These proportions are higher than those reported in previous studies [11, 17–19]. This reflects a growing recognition of the importance of pharmacovigilance in clinical practice. Moreover, 94% of HCPs and 98% of UG students believed that all healthcare personnel should receive formal pharmacovigilance training, indicating a favourable attitude toward continuous education. This finding is higher than that reported in earlier studies [17, 21, 22] and suggests that the emphasis on training programs may not be consistent across all other institutions.
Almost all participants, 95% of HCPs and 92% of UG students acknowledged that ADR reporting enhances medication safety, which is consistent with previous findings [12, 20, 23]. This demonstrates strong professional commitment to patient safety. A pronounced difference was observed in ADR-related discussions with patients, 90% of HCPs had discussed ADRs during medication reviews compared to only 14% of UG students. This gap may be due to students limited clinical exposure and hands-on experience during training. Students primarily focus on theoretical learning and may not yet have the opportunity to engage in detailed discussions about adverse drug reactions during clinical rounds. Unlike HCPs, who are actively involved in patient care and medication management.
Despite this satisfactory awareness and positive attitude, a significant gap between knowledge and practice was evident in actual reporting rates. Only 56% of HCPs had ever reported an ADR, which aligns with Tripathi et al. [16] and B and K [20]. Among students, the reporting rate was as low as 9%, much lower than that observed by Bhatt and Kumar [12] (96%) and comparable to R et al. [18] and Tiwari et al. [15]. This indicates a persistent problem of underreporting and suggests that even though pharmacovigilance is taught in the MBBS curriculum, the practical importance of ADR reporting is underestimated by UG students. This gap emphasizes the need for structured, practical exposure through mandatory ADR reporting exercises, role-plays, and the integration of reporting activities within clinical postings.
A majority of HCPs (72%) and UG students (55.4%) had received training on ADR reporting, which is higher than the reported by other studies [15, 17, 19, 21]. This demonstrates improvement in training initiatives, but also emphasizes the need to make such training more accessible and consistent. Exposure to ADR reporting tools was high, with 79% of HCPs and 98% of students having seen a reporting form, which closely aligns with other studies [10–12, 17]. This indicates good exposure to reporting tools among UG students, likely due to the curriculum. However, actual reporting rates remain low, suggesting that practical engagement in ADR reporting remains an area for improvement.
Overall, KAP scores were higher among HCPs than UG students (table 1). This difference can likely be due to the regular sensitization sessions, continuing education, and direct patient care by HCPs. In contrast, lower scores among students are likely due to their limited real-time clinical experience and a curriculum that focuses more on theoretical aspects than hands-on training. Similar trend have been observed in studies by Bhatt and Kumar [12] and Tiwari et al. [15], where adequate knowledge did not translate into optimal practice.
CONCLUSION
This study demonstrates encouraging awareness and positive attitudes toward pharmacovigilance among HCPs and UG students. However, the persistently low rate of ADR reporting and limited participation in training highlight a clear gap between knowledge and practice. The relatively small sample size and self-reporting nature of the study may limit the generalizability of findings.
Institutions should promote a culture of pharmacovigilance through regular workshops, hands-on training, simplified reporting procedures, and feedback mechanisms. Integrating ADR reporting exercises into medical curricula and encouraging student participation during clinical postings can bridge the theory–practice gap. By adopting these strategies, institutions can enhance ADR reporting rates and foster sustained engagement in pharmacovigilance, ultimately improving patient safety. Future studies should explore barriers to ADR reporting and evaluate the effectiveness of targeted interventions.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
All authors have contributed equally.
CONFLICT OF INTERESTS
Declared none
REFEREPNCES
Malam P, Sondarva D, Chavda D. Knowledge, attitude and practice of pharmacovigilance among intern doctors of peripheral medical college in Gujarat. Natl J Physiol Pharm Pharmacol. 2022;12(8):1. doi: 10.5455/njppp.2022.12.11409202130122021.
Kalaiselvan V, Thota P, Singh GN. Pharmacovigilance programme of India: recent developments and future perspectives. Indian J Pharmacol. 2016;48(6):624-8. doi: 10.4103/0253-7613.194855, PMID 28066097.
Upadhyaya P, Seth V, Moghe VV, Sharma M, Ahmed M. Knowledge of adverse drug reaction reporting in first year postgraduate doctors in a medical college. Ther Clin Risk Manag. 2012;8:307-12. doi: 10.2147/TCRM.S31482, PMID 22767994.
Remesh A. Identifying the reasons for under-reporting of ADR: a cross-sectional survey. Res J Pharm Biol Chem Sci. 2012;3(4):86.
Prakash S. Pharmacovigilance in India. Indian J Pharmacol. 2007;39(3):123. doi: 10.4103/0253-7613.33430.
Vallano A, Castaneda PF, Quijada Manuitt MA, Simon PC, Pedros C, Quintana B, Esterlich E, Arnau JM. Hospital doctors views and concerns about pharmacovigilance. J Pharmacovigil. 2015;3(2):1-5. doi: 10.4172/2329-6887.1000160.
Acharya T, Trivedi M, Joshi K, Chhaiya S, Mehta D. An observational study to evaluate knowledge, attitude and practice of pharmacovigilance among undergraduate medical students of a Tertiary Care Teaching Hospital. Natl J Physiol Pharm Pharmacol. 2022;12(9):1361-5. doi: 10.5455/njppp.2022.12.01001202213012022.
Gupta SK, Nayak RP, Shivaranjani R, Vidyarthi SK. A questionnaire study on the knowledge attitude and the practice of pharmacovigilance among the healthcare professionals in a teaching hospital in South India. Perspect Clin Res. 2015;6(1):45-52. doi: 10.4103/2229-3485.148816, PMID 25657902.
Sneha K, Pingale A, Vaishnavi D. To evaluate the attitude, knowledge and practice regarding pharmacovigilance in undergraduate students and postgraduate residents at a tertiary healthcare centre a questionnaire-based study. Int J Pharm Clin Res. 2022;14(4):436-41.
Srinivasan V, Sheela D, Mridula D. Knowledge, attitude and practice of pharmacovigilance among the healthcare professionals in a Tertiary Care Hospital a questionnaire study. Biomed Pharmacol J. 2017 Sep 28;10(3):1441-7. doi: 10.13005/bpj/1251.
Mahajan S, Sharma A, Gupta S, Qazi HK. Knowledge attitude and practice (KAP) of pharmacovigilance among MBBS undergraduates. J Cardiovasc Dis Res. 2022;13(4):793‑8.
Bhatt S, Kumar H. Knowledge attitude and practice of pharmacovigilance among medical students. Int J Basic Clin Pharmacol. 2024 Feb 23;13(2):250-4. doi: 10.18203/2319-2003.ijbcp20240381.
Kharkar M, Bowalekar S. Knowledge, attitude and perception/practices (KAP) of medical practitioners in India towards adverse drug reaction (ADR) reporting. Perspect Clin Res. 2012 Jul;3(3):90-4. doi: 10.4103/2229-3485.100651, PMID 23125959.
Abdulsalim S, Farooqui M, Alshammari MS, Alotaibi M, Alhazmi A, Alqasomi A. Evaluation of knowledge, attitudes and practices about pharmacovigilance among community pharmacists in Qassim, Saudi Arabia. Int J Environ Res Public Health. 2023 Feb 17;20(4):3548. doi: 10.3390/ijerph20043548, PMID 36834240.
Tiwari A, More S, Mishra P, Sharma A, Hetawal P, Patel S. Assessment of knowledge, attitude and practices toward pharmacovigilance among undergraduate medical students of a Tertiary Care Teaching Hospital of Central India. Natl J Physiol Pharm Pharmacol. 2023;13(9):1806‑10. doi: 10.5455/njppp.2023.13.01052202304022023.
Tripathi R, Sachdeva M, Mehta G. Exploration of knowledge attitude and practice of pharmacovigilance among healthcare professionals: a cross-sectional study. Ars Pharm. 2024 Dec 20;66(1):16-24. doi: 10.30827/ars.v66i1.31316.
Maheshwari R, Manjunatha CH, Jesudoss Prabhakaran ACJ. Knowledge attitude and practice of pharmacovigilance among health‑care professionals of a Tertiary Care Hospital in Puducherry a questionnaire‑based study. Natl J Physiol Pharm Pharmacol. 2021;11(3):351‑5. doi: 10.5455/njppp.2021.11.01023202129012021.
Sravani MR, Vijetha MS, Avula N, Balla KS, Samatham B, Ahmed MZ, Deepa Latha C. A study on knowledge attitude and practice of pharmacovigilance and adverse drug reactions awareness among health‑care students in a Tertiary Care Teaching Hospital Telangana India. Natl J Physiol Pharm Pharmacol. 2024;14(3):509‑14. doi: 10.5455/njppp.2024.14.12590202329012024.
Kumari A, Haque I, Bhyan SJ, Sreelakshmi M, Goel N, Jain A. Knowledge attitude and practice of adverse drug reaction monitoring and pharmacovigilance among various healthcare professionals in India. Preprints.org; 2020. Available from: https://www.preprints.org/manuscript/202008.0067/v1. [Last accessed on 27 Jan 2025].
Jyothi DB, Swetha K. Knowledge awareness and practice of pharmacovigilance among health‑care providers at a Tertiary Care Hospital a questionnaire‑based study. Natl J Physiol Pharm Pharmacol. 2020;10(4):270‑5. doi: 10.5455/njppp.2020.10.02042202013022020.
Era N, Mukherjee S, Bordoloi S. Assessment of knowledge attitude and practice of pharmacovigilance among undergraduate medical students in a Tertiary Care Teaching Hospital of Eastern India: a questionnaire-based study. Natl J Physiol Pharm Pharmacol. 2020;10(6):460‑3. doi: 10.5455/njppp.2020.10.03062202025032020.
Bepari A, Niazi SK, Rahman I, Dervesh AM. The comparative evaluation of knowledge attitude and practice of different health-care professionals about the pharmacovigilance system of India. J Adv Pharm Technol Res. 2019;10(2):68-74. doi: 10.4103/japtr.JAPTR_4_19, PMID 31041185.
Agarwal M, Ahmed J, Roy V. Knowledge awareness and practice about pharmacovigilance among healthcare providers of a Tertiary Care Teaching Hospital in New Delhi (India). MAMC J Med Sci. 2017;3(3):146‑51. doi: 10.4103/mamcjms.mamcjms_31_17.