
Int J Curr Pharm Res, Vol 18, Issue 2, 96-105Original Article
FORMULATION AND EVALUATION OF A NANOEMULGEL CONTAINING INDOMETHACIN AND BROMELAIN FOR LOCALIZED ANTI-INFLAMMATORY ACTIVITY
SAMIDHA S. GOLATKAR*, MADAN D. POMAJE, APEKSHA C. RAHATE, PARNIKA P. KADAM
Department of Pharmaceutics, Govindrao Nikam College of Pharmacy, Sawarde, Maharashtra-415606, India
*Corresponding author: Samidha S. Golatkar; *Email: golatkarsamidha17@gmail.com
Received: 15 Nov 2025, Revised and Accepted: 03 Jan 2026
ABSTRACT
Objective: This study aimed to develop and evaluate a topical nanoemulgel containing indomethacin and bromelain for effective localized anti-inflammatory therapy.
Methods: Nanoemulsions were formulated using eucalyptus and nirgundi oils as the oil phase, Tween 20, and propylene glycol as the surfactant and co-surfactant. Pseudo-ternary phase diagrams identified the largest nanoemulsion region at Smix 1:1. The nanoemulsions were incorporated into Carbopol 934 to obtain nanoemulgel. Preformulation studies and FTIR and DSC analyses confirmed drug-excipient compatibility, while molecular docking showed strong binding of indomethacin and bromelain to inflammatory targets, including COX-2 and TNF-α. The physicochemical parameters of nanoemulgel, including globule size, PDI, zeta potential, pH, viscosity, spreadability, and extrudability, were evaluated. In vitro anti-inflammatory activity was assessed using protein denaturation and HRBC membrane stabilization assays, while permeation was studied using Franz diffusion cells.
Results: The formulations (F1-F5) demonstrated nanoscale droplets ranging from 119.6 to 189.4 nm, with zeta potential between −12.0 and −19.2 mV. The pH values were between 6.50 and 6.73, and the viscosity was approximately 21.355–21.392 mPa·s. The spreadability was measured between 33.01 and 39.32 g·cm/s, and extrudability ranged from 61.00% to 74.81%. The in vitro anti-inflammatory activity of formulations F3 and F4 was significant and comparable to that of a marketed indomethacin gel. Franz diffusion studies indicated an increase in cumulative permeation over time, with high flux and permeation coefficients.
Conclusion: The developed nanoemulgels, particularly F3, exhibited satisfactory rheological properties, enhanced drug permeation, and significant anti-inflammatory activity comparable to that of a marketed indomethacin gel formulation.
Keywords: Nanotechnology, Nanoemulgel, Rheumatoid arthritis, Topical drug delivery, Anti-inflammatory activity, Indomethacin, Bromelain
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i2.8073 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Rheumatoid arthritis (RA) is a chronic autoimmune disorder characterized by synovial inflammation, cartilage degradation, and bone erosion, which leads to pain, stiffness, and functional disability [1, 2]. Although systemic pharmacotherapy using NSAIDs, corticosteroids, and DMARDs remains central to RA management, long-term systemic administration often causes significant adverse effects such as gastrointestinal irritation, renal toxicity, hepatotoxicity, and immunosuppression [3, 4]. These limitations necessitate alternative drug delivery approaches capable of providing effective localized therapy while minimizing systemic exposure [1, 5].
Topical drug delivery systems have emerged as a promising strategy for managing localized inflammatory conditions, offering site-specific drug action, avoidance of first-pass metabolism, reduced systemic side effects, and improved patient compliance [6, 7]. However, the efficacy of conventional topical formulations is often limited by the stratum corneum barrier, which restricts drug penetration into deeper tissues, such as synovial joints. Thus, advanced carrier systems are required to enhance skin permeation and drug retention at target sites. Nanotechnology-based drug delivery systems have gained attention for topical applications because of their ability to improve drug solubility, stability, and skin permeation [8]. Among nanocarriers, nanoemulsions are particularly effective because of their small droplet size, high kinetic stability, and enhanced interfacial surface area, which facilitate improved drug diffusion across the skin barrier [8–12].
Indomethacin is a potent NSAID used to manage inflammatory disorders, including RA, through cyclooxygenase enzyme inhibition and prostaglandin synthesis suppression. Despite its efficacy, oral indomethacin causes severe gastrointestinal and renal adverse effects, limiting its long-term use [13]. Bromelain, a proteolytic enzyme derived from pineapple (Ananas comosus), exhibits anti-inflammatory, anti-edematous, and immunomodulatory properties, along with a favorable safety profile [14-16]. The combination of indomethacin and bromelain offers complementary therapeutic effects, potentially enhancing anti-inflammatory efficacy while reducing the dosage of synthetic NSAID. This study aimed to develop and evaluate a nanoemulgel-based topical delivery system for indomethacin and bromelain co-delivery for localized anti-inflammatory therapy.
MATERIALS AND METHODS
Materials
Indomethacin was procured from Yarrow Chemicals and bromelain from Vital Herbs. Eucalyptus oil was purchased from Loba Chemie Pvt. Ltd., and Nirgundi oil was sourced from R V Essentials. Sesame oil was obtained from the local market, and Karanja oil was supplied by Research Lab Fine Chem Industries, India.
Methods
Solubility of drugs in oils, surfactants, and Co-surfactants [18]
The solubility of indomethacin and bromelain in selected oils, surfactants, and co-surfactants was determined using the shake flask method. An excess amount of each drug was added to 2 ml of each excipient and vortexed for 10 min. The mixtures were then shaken at 3000 rpm for 48 h at room temperature and centrifuged at 15,000 rpm for 10 min. The supernatant was filtered through a 0.45 µm membrane filter, diluted with methanol, and analyzed using a UV spectrophotometer at 320 nm and 280 nm for indomethacin and bromelain, respectively. Solubility is expressed in mg/ml.
Screening of surfactant and Co-surfactant [18]
Surfactants were screened based on their emulsification efficiency and transparency. The oil phase (300 mg) was mixed with an equal amount of surfactant and heated to 40 °C. From this mixture, 50 mg was diluted to a final volume of 50 ml using distilled water. The ease of emulsification was evaluated by counting the number of flask inversions required to obtain a uniform emulsion. The percentage transmittance was measured at 440 nm using distilled water as a blank.
Co-surfactants such as PEG 400, PEG 600, propylene glycol, and isopropyl alcohol were screened similarly by preparing mixtures of oil, selected surfactant (Tween 20), and co-surfactant, followed by the evaluation of the emulsification efficiency and transparency.
Construction of pseudo-ternary phase diagrams [18, 19]
Pseudo-ternary phase diagrams were constructed using the water titration method to identify the nanoemulsion region and optimize the surfactant to co-surfactant ratio. Smix ratios (1:1, 2:1, and 1:2) were prepared, vortexed, and equilibrated at 40 °C. The data obtained from these experiments were used to construct pseudo ternary phase diagrams using Chemix School-Portable Chemistry Software 7.0. The phase boundaries were recorded, and the nanoemulsion regions were identified.
Drug–excipient compatibility studies
FTIR analysis
FTIR spectroscopy was performed to assess the compatibility between indomethacin, bromelain, and the selected excipients by comparing the characteristic functional group peaks.
Differential scanning calorimetry
DSC analysis was conducted to evaluate the thermal behaviour, crystallinity, and potential interactions between the drugs and excipients by analyzing the changes in the melting endotherms.
Molecular docking study [20]
Molecular docking studies were performed using AutoDock Vina to predict the binding affinity and interaction patterns of indomethacin and bromelain with inflammatory target proteins, including COX-1, COX-2, TNF-α, IL-6, phospholipase A2, and phospholipase D2. The binding energies (kcal/mol) were used to assess the interaction strength.
Formulation development
Preparation of drug loaded nanoemulsion
Blank nanoemulsions within the nanoemulsion region were identified based on pseudo-ternary phase diagrams constructed using eucalyptus oil, nirgundi oil, Tween 20, propylene glycol (Smix 1:1) and water. The formulation exhibiting visual clarity and stability without phase separation after 24 h was selected for drug loading.
Indomethacin was dissolved in the oil phase, and bromelain was dissolved in an aqueous phase. The oil phase (eucalyptus oil: nirgundi oil, 1:1) was mixed with Smix (Tween 20: propylene glycol, 1:1) under magnetic stirring. Distilled water was added dropwise to form a coarse emulsion, which was then homogenized at 8000 rpm for 15–20 min to obtain a nanoemulsion. Five nanoemulgel formulations (F1–F5) were prepared by varying the proportions of indomethacin and bromelain (table 1), while the oil phase composition and Smix ratio were maintained constant.
Table 1: % w/w of indomethacin and bromelain in each formulation
| Component | F1 | F2 | F3 | F4 | F5 |
| Indomethacin (% w/w) | 1.0 | 0.25 | 0.5 | 0.75 | – |
| Bromelain (% w/w) | – | 0.75 | 0.5 | 0.25 | 1.0 |
Preparation of nanoemulgel
A gel base was prepared by dispersing Carbopol 934 (1% w/v) in distilled water and allowing it to hydrate for 24 h. The prepared nanoemulsions (F1–F5) were incorporated into the gel base in a 1:1 (w/w) ratio under gentle stirring to obtain uniform nanoemulgels. Preservatives were added prior to incorporating the nanoemulsion.
Evaluation
Evaluation of nanoemulsion
Organoleptic properties of nanoemulsion
pH determination of nanoemulsion
Droplet size and polydispersity index
Zeta potential
The prepared nanoemulsion formulations were evaluated for their organoleptic properties, such as appearance, colour, odour, and clarity, which were visually examined and recorded.
The pH of the prepared nanoemulsion formulations was measured using a calibrated digital pH meter at room temperature
The droplet size and polydispersity index (PDI) of the nanoemulsion formulations were determined using dynamic light scattering (DLS).
The zeta potential of the nanoemulsion formulations was measured using a zeta sizer to determine the surface charge and electrostatic stability of the droplets.
Evaluation of nanoemulgel
Organoleptic properties of nanoemulgel
pH determination of nanoemulgel [21]
Viscosity of nanoemulgel [21]
Spreadability of nanoemulgel [21]
Extrudability of nanoemulgel [22]
Drug content
In vitro anti-inflammatory activity
In vitro permeation study [19]
The prepared nanoemulgel formulations were evaluated for their organoleptic properties, including appearance, color, odour, and texture, through visual inspection.
Approximately 1 g of nanoemulgel was dispersed in 10 ml of distilled water and allowed to stand for 2 h, after which the pH was measured at room temperature using a calibrated digital pH meter.
Viscosity measurements were performed using a Brookfield viscometer fitted with an appropriate spindle (spindle no. 6) at 32.6 °C and 10 rpm.
The spreadability was determined using the slip-and-drag method. Approximately 2 g of nanoemulgel was placed between two ground glass slides. A weight of 1 kg was applied for 5 min to ensure uniform thickness. The upper slide was pulled using a 70 g load, and the time required to move a distance of 7.5 cm was recorded for each sample. Spreadability was calculated using the following equation:
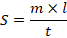
Extrudability was assessed by filling the nanoemulgel into collapsible aluminium tube. A 500 g weight was applied, and the amount of extruded gel was collected and weighed. Extrudability was expressed as the percentage of gel extrusion.
The drug content was determined using UV–Visible spectrophotometry. For single-drug formulations, the drug content was calculated directly from calibration curves. For combination formulations, simultaneous estimation was performed at 280 nm and 320 nm using absorptivity coefficients and Beer-Lambert’s law. One g of nanoemulgel was dissolved in phosphate buffer (pH 7.4), sonicated, filtered, diluted, and analyzed spectrophotometrically. The percentage drug content was calculated based on theoretical drug loading.
Protein denaturation assay [23]
The anti-inflammatory potential of the nanoemulgel formulations was evaluated using a protein denaturation method, with bovine serum albumin (BSA) as the protein model. A 5% w/v bovine serum albumin (BSA) solution was prepared in phosphate buffer (pH 6.3). The test formulations were diluted to 100–500 μg/ml, and a marketed indomethacin gel (Inmecin 1%, 100 μg/ml) was used as the reference standard. The percentage inhibition of protein denaturation was calculated as follows:
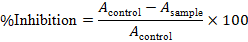
Membrane stabilization assay (HRBC method) [24]
The membrane stabilization method was used to assess the ability of the formulations to protect human red blood cells (HRBCs) from haemolysis. Fresh blood was collected from a healthy volunteer, washed with isotonic saline, and a 10% v/v HRBC suspension was prepared using the blood. The percentage of haemolysis inhibition was calculated as follows:
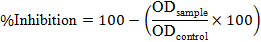
The drug release of the nanoemulgel formulations (F1–F5) and the marketed gel was evaluated using a Franz diffusion cell with a cellophane membrane. The receptor compartment contained phosphate buffer (pH 7.4) at 37 °C with stirring. One g of gel was applied to the donor side, and 1 ml samples were withdrawn at 15–480 min intervals, replaced with fresh buffer, and analyzed using UV spectrophotometry (320 nm for indomethacin and 280 nm for bromelain).
The cumulative drug permeation was plotted against the time. The flux (J) and permeation coefficient (Kp) were calculated as follows:
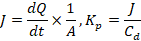
Where A diffusion area, and Cd is the initial drug concentration.
RESULTS AND DISCUSSION
Preformulation studies
Preformulation studies such as melting point determination, UV spectroscopic analysis, and construction of calibration curve were carried out using standard pharmacopeial procedures.
Melting point
The melting points were determined using the capillary method. The melting point of indomethacin was determined to be 162 °C.
Determination of λmax of indomethacin and bromelain
The λmax of Indomethacin and Bromelain was found to be 320 nm and 280 nm, respectively.
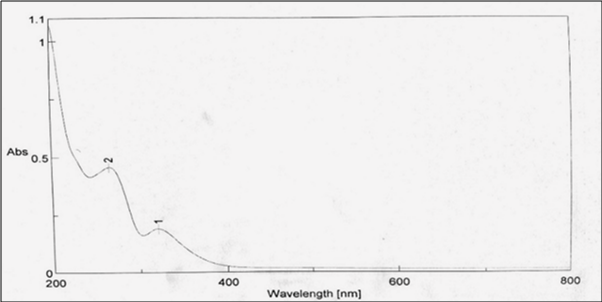
Fig. 1: λmax of indomethacin
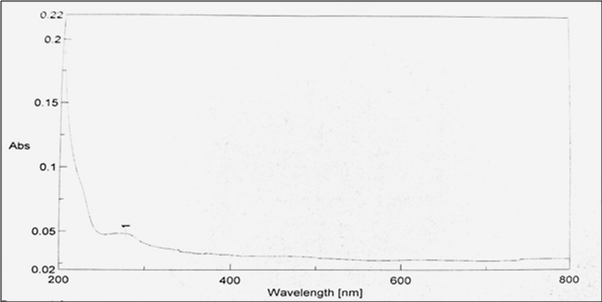
Fig. 2: λmax of bromelain
Calibration curves
For preparation of calibration curves, stock solutions (100 µg/ml) of indomethacin and bromelain were prepared using phosphate buffer pH 7.4. Aliquots were further diluted to obtain concentrations ranging from 5–25 µg/ml.
Solubility of drugs in different vehicles
Indomethacin showed maximum solubility in eucalyptus oil (253.32 mg/ml) and nirgundi oil (214.98 mg/ml), while bromelain exhibited higher solubility in eucalyptus oil (94.16 mg/ml).
Screening of surfactant and Co-surfactant
Surfactants were evaluated for their emulsification efficiency with the selected oil blend (eucalyptus and nirgundi oil) based on the ease of emulsification and the percentage transmittance at 440 nm. Tween 20 exhibited the highest transmittance (82.39%), followed closely by Tween 80 (81.82%).
Co-surfactants were screened in combination with Tween 20 and an oil blend. Propylene glycol exhibited the highest transmittance (94.23%), indicating superior emulsification with the selected surfactant and oil phase.
These results guided the choice of Tween 20 as the surfactant and propylene glycol as the co-surfactant for the nanoemulsion formulation.
Construction of pseudo-ternary phase diagram
Analysis of the pseudo-ternary phase diagrams indicated that a Smix ratio of 1:1 (Tween 20: Propylene glycol) provided the largest nanoemulsion region, reflecting superior emulsification efficiency and stability of the nanoemulsion. Therefore, this ratio, along with the selected oil blend, was chosen as the optimal composition for further nanoemulsion and nanoemulgel formulation.
Drug-excipients compatibility studies
Fourier transformed infrared (FT-IR) spectroscopic analysis (FTIR)
The FTIR spectrum of indomethacin exhibited characteristic peaks corresponding to the C–Cl, C–N, C–O, N–H, and O–H functional groups, confirming its structural integrity.
The FTIR spectrum of bromelain showed characteristic peaks corresponding to the N–H, C–H, C–O, and C=C functional groups, confirming its proteinaceous structure.
The FTIR spectrum of the physical mixture exhibited all the major characteristic peaks of indomethacin and bromelain, confirming their compatibility.
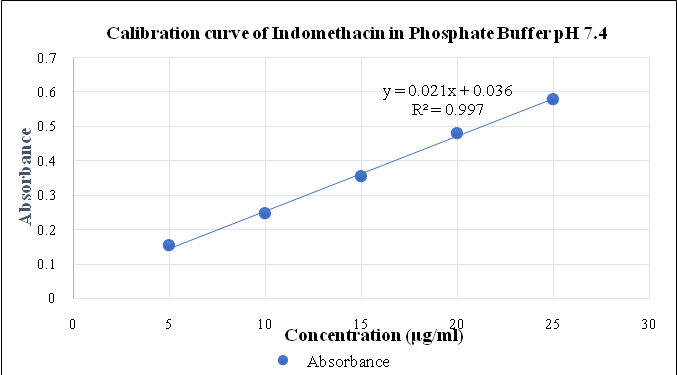
Fig. 3: Calibration curve of indomethacin
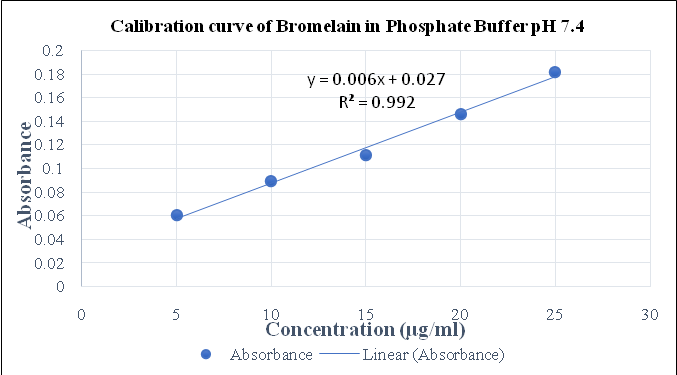
Fig. 4: Calibration curve of bromelain
Table 2: Solubility of indomethacin and bromelain in various vehicles
| S. No. | Vehicle | Solubility of indomethacin (mg/ml) | Solubility of bromelain (mg/ml) |
| Eucalyptus | 253.32 | 94.16 | |
| Nirgundi | 214.98 | 32.75 | |
| Sesame | 82.90 | 22 | |
| Karanja | 111.24 | 14.5 | |
| Tween 20 | 126.27 | 8.35 | |
| Tween 40 | 65.622 | 2.43 | |
| Tween 60 | 62.34 | 2.55 | |
| Tween 80 | 108.90 | 12.35 | |
| Span 20 | 5.06 | 15.86 | |
| Span 80 | 19.72 | 11.33 | |
| PEG 200 | 30.82 | 0.31 | |
| PEG 400 | 23.36 | 0.633 | |
| PEG 600 | 91.42 | 0.25 | |
| Propylene glycol | 102.44 | 17.13 | |
| Isopropyl alcohol | 31.65 | 0.06 | |
| Oleic acid | 6.03 | 26.7 |
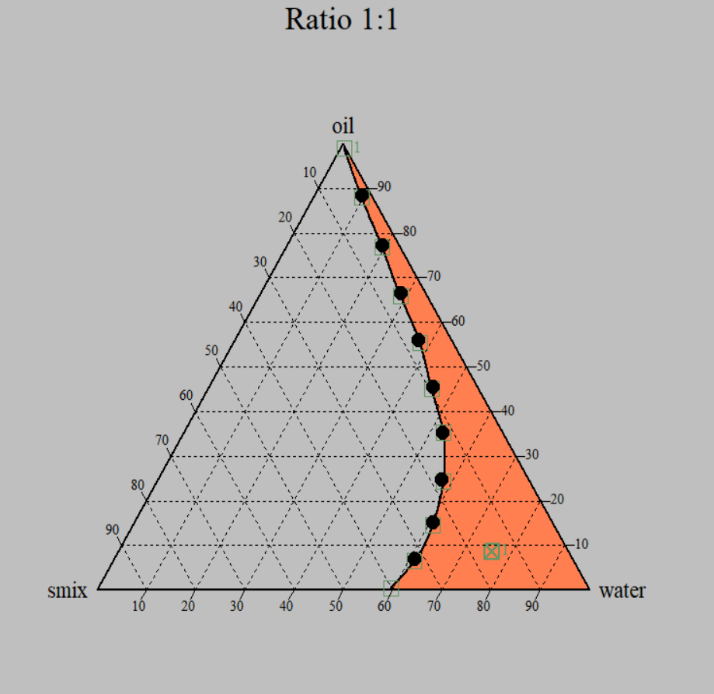
Fig. 5: Pseudo ternary phase diagram 1:1 ratio
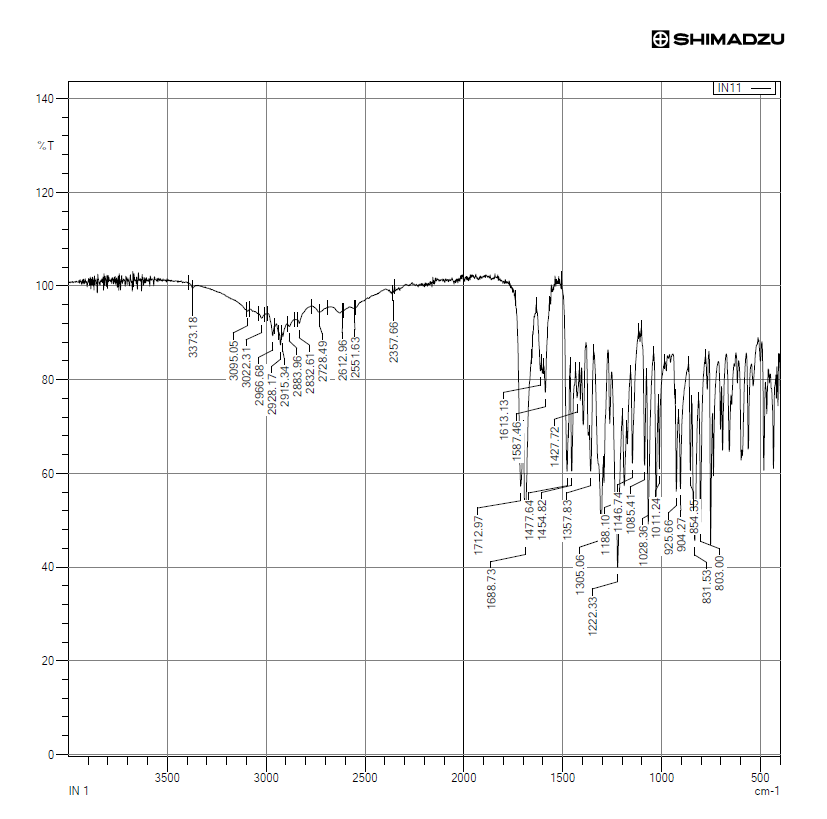
Fig. 6: FTIR spectra of Indomethacin
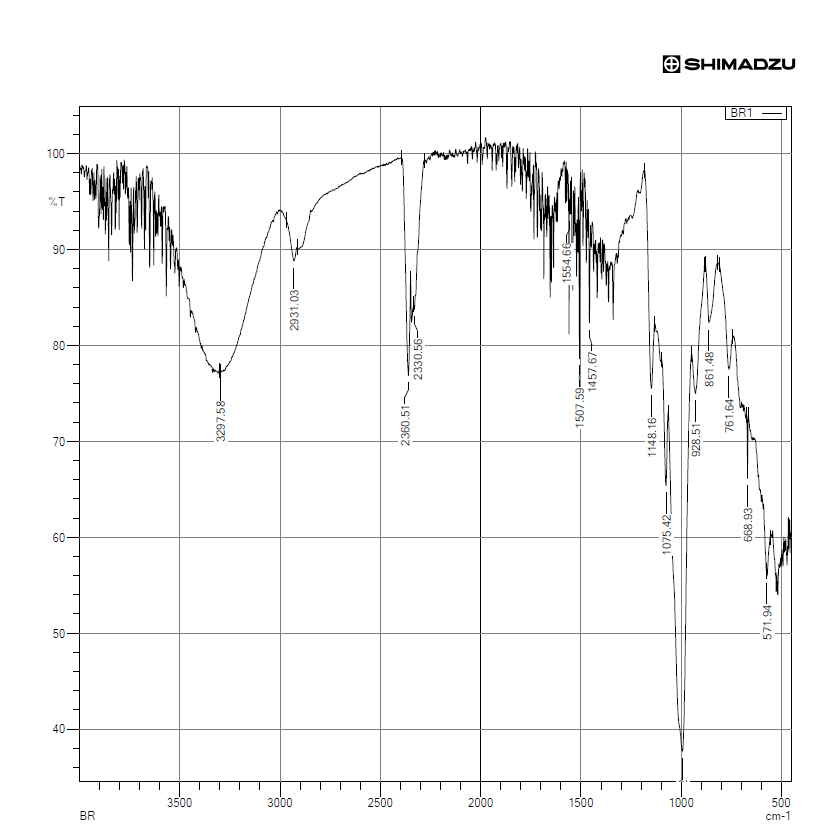
Fig. 7: FTIR spectra of bromelain
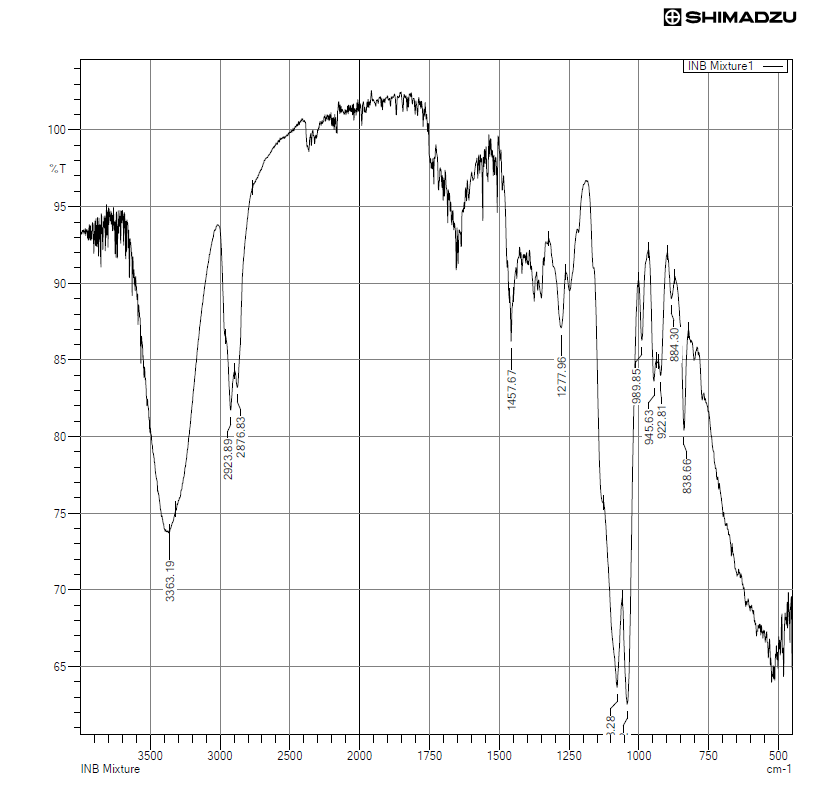
Fig. 8: FTIR spectra of physical mixture
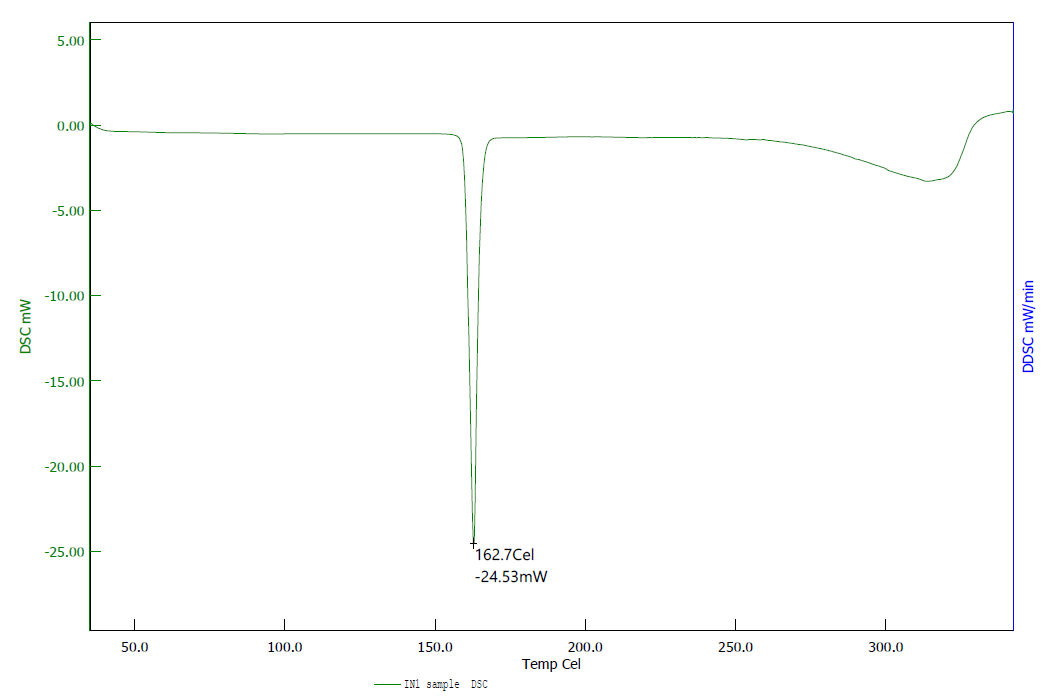
Fig. 9: DSC of indomethacin
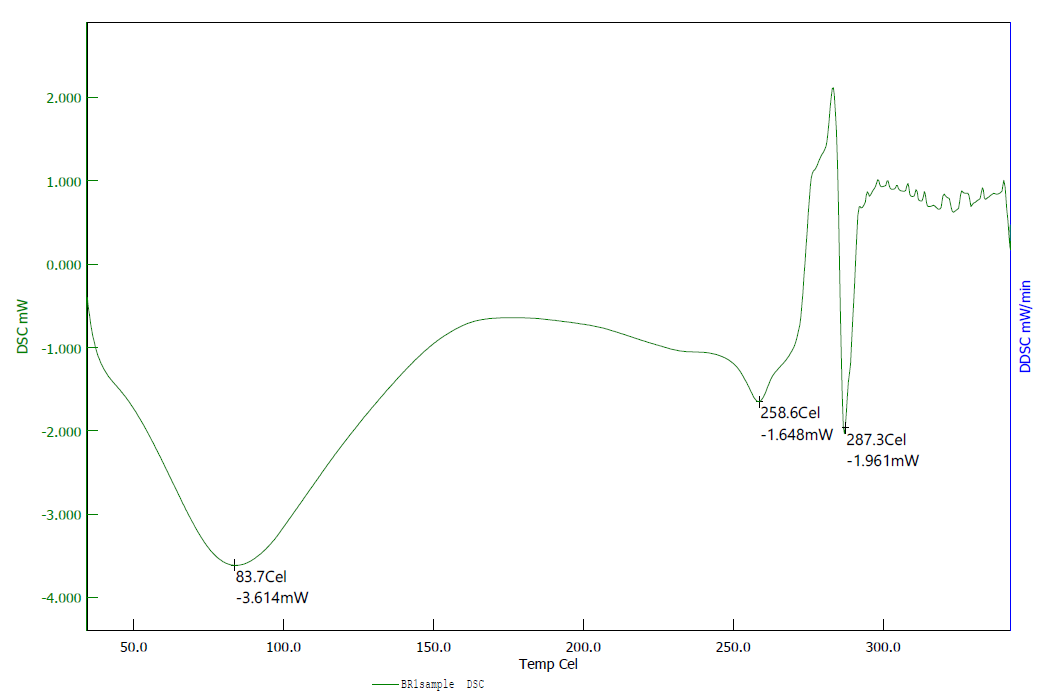
Fig. 10: DSC of bromelain
Differential scanning calorimetry (DSC)
The DSC thermogram of indomethacin showed a sharp endothermic peak at 162.7 °C corresponding to its melting point, confirming its crystalline nature. The DSC thermogram of bromelain exhibited a broad endothermic transition rather than a sharp melting peak, which is characteristic of protein denaturation rather than melting.
Molecular docking study
The binding energies (kcal/mol) represent the stability and strength of the interaction between a drug and a receptor. The docking results suggest that Indomethacin has strong interactions with COX-2 and TNF-α, indicating its potential for anti-inflammatory activity through the inhibition of these key mediators. A similar analysis was performed for bromelain, confirming its complementary binding interactions with the inflammatory target COX-2 (table 3). These results support the rationale for combining both drugs in a nanoemulgel formulation for synergistic localized anti-inflammatory effects.
Evaluation of nanoemulsion and nanoemulgel
Evaluation of nanoemulsion
pH
Droplet size, PDI, and zeta potential
The pH values ranged from 5.60 to 5.97, which is within the acceptable topical range (5.5–7.0) (table 4).
The mean globule size ranged from 119.6 nm to 189.4 nm, with a PDI between 0.462 and 0.753, indicating a nanoscale and moderately uniform distribution of globules.
Zeta potential ranged from –12.0 to –19.2 mV, suggesting good stability (table 4)
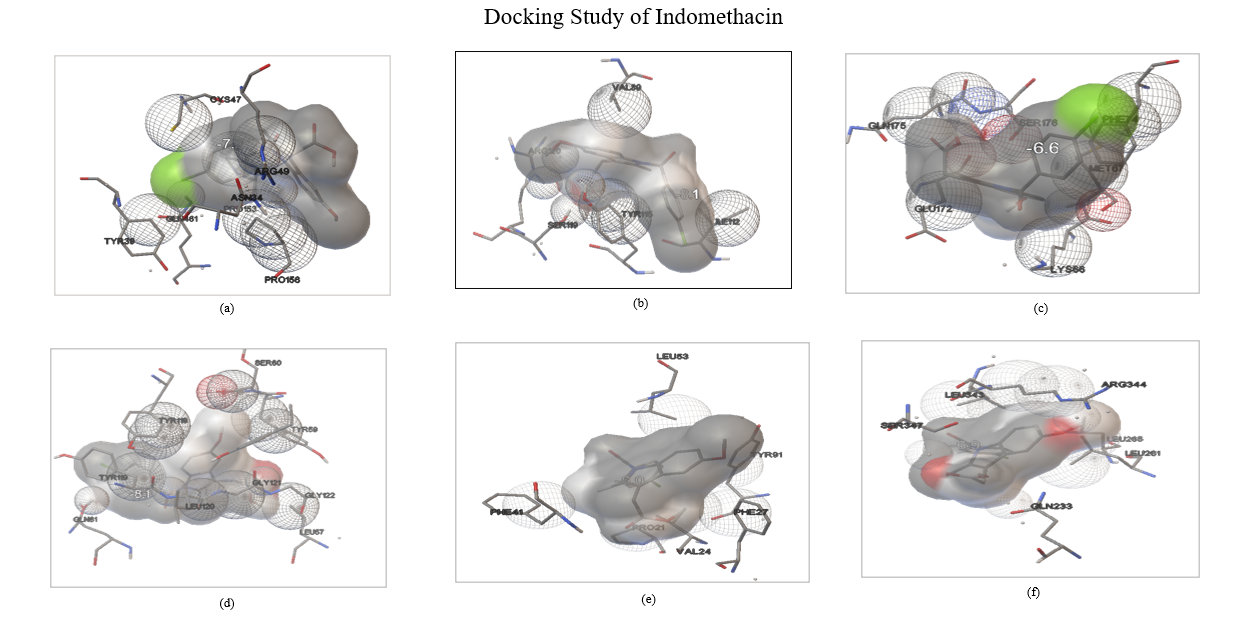
Fig. 11: Docking study of Indomethacin with (a) COX1; (b) COX2; (c) IL 6 (d) TNF Alpha; (e) Phospholipase A2; (f) Phospholipase D2
Table 3: Binding energy of indomethacin and bromelain with receptors
| S. No. | Receptor | Binding energy (kcal/mol) | Binding energy affinity (kcal/mol) |
| 1 | COX-1 | -7.7 | -7.7 |
| 2 | COX-2 | -8.1 | -8.6 |
| 3 | TNF-α | -8.1 | -8.0 |
| 4 | IL-6 | -6.6 | -7.1 |
| 5 | Phospholipase A2 | -8.0 | -7.2 |
| 6 | Phospholipase D2 | -6.9 | -8.1 |
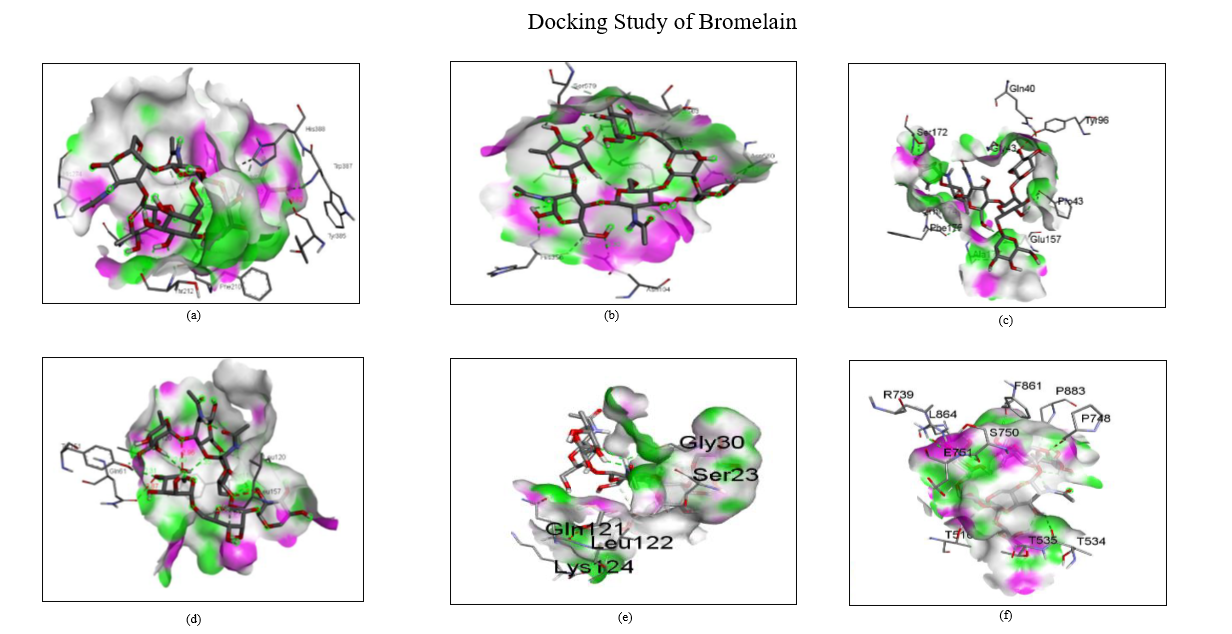
Fig. 12: Docking study of Bromelain with (a) COX1; (b) COX2; (c) IL 6 (d) TNF Alpha; (e) Phospholipase A2; (f) Phospholipase D2
Table 4: Physicochemical properties of nanoemulsion (F1–F5)
| Formulation | pH | Mean globule size (nm) | PDI | Zeta potential (mV) |
| F1 | 5.85 | 119.6 | 0.753 | -13.5 |
| F2 | 5.60 | 189.4 | 0.738 | -19.2 |
| F3 | 5.94 | 154.0 | 0.462 | -12.0 |
| F4 | 6.01 | 182.9 | 0.466 | -16.7 |
| F5 | 5.97 | 146.6 | 0.553 | -15.4 |
Evaluation of nanoemulgel
pH
Viscosity
Spreadability
Extrudability
Drug content
Anti-inflammatory activity
Protein denaturation assay
In vitro permeation study
The pH ranged from 6.50 to 6.73, which is within the acceptable range for topical formulation (table 5).
The viscosity ranged from 21,355 to 21,392 mPa·s, ensuring good skin adherence and ease of application (table 5).
The spreadability values ranged from 33.01 to 39.32 g·cm/sec, indicating acceptable spreading characteristics for the formulations (table 5).
All formulations exhibited satisfactory extrudability, ranging from 61.00% to 74.81%, indicating easy dispensing from the container (table 5).
Table 5: pH, viscosity, spreadability and extrudability of nanoemulgel (F1–F5)
| Formulation | pH | Viscosity (mPa·s) | Spreadability (g·cm/sec) | % Extrudability |
| F1 | 6.53 | 21,359 | 35.00 | 63.37 |
| F2 | 6.50 | 21,355 | 37.23 | 65.02 |
| F3 | 6.55 | 21,361 | 34.32 | 74.81 |
| F4 | 6.65 | 21,369 | 33.01 | 73.00 |
| F5 | 6.73 | 21,392 | 36.45 | 72.64 |
Table 6: % Drug content of nanoemulgel (F1–F5)
| Formulation | % Indomethacin | % Bromelain |
| F1 | 90.0 | - |
| F2 | 86.8 | 91.1 |
| F3 | 95.0 | 93.0 |
| F4 | 92.86 | 93.0 |
| F5 | - | 89.6 |
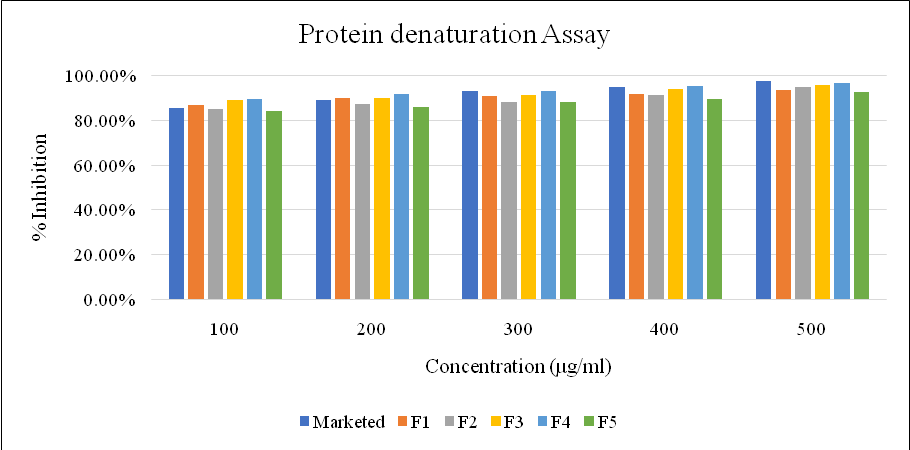
Fig. 13: Graph showing % inhibition for protein denaturation assay
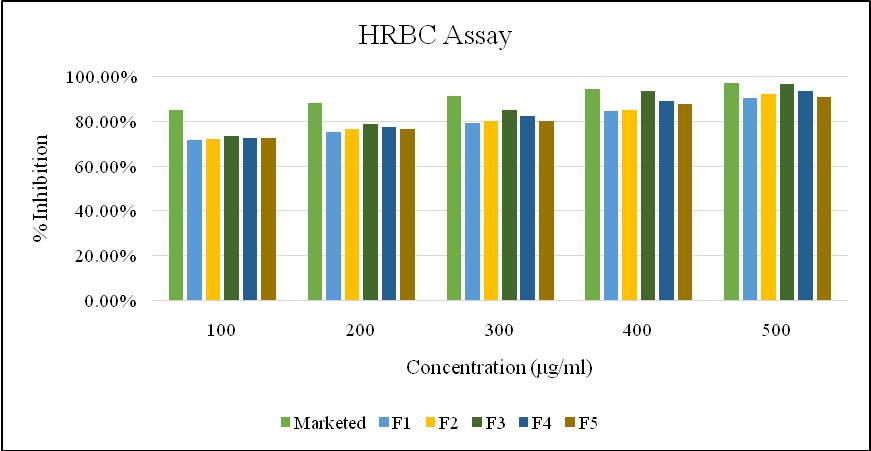
Fig. 14: Graph showing % inhibition for HRBC assay
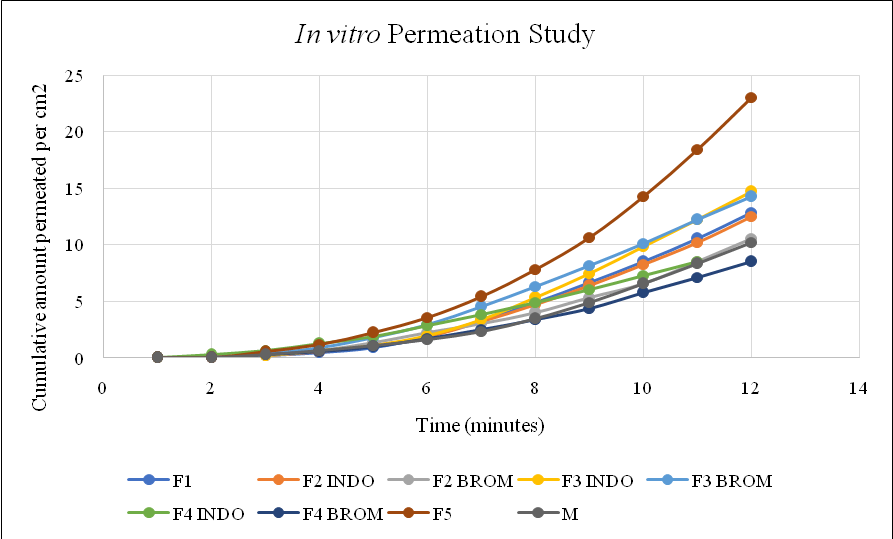
Fig. 15: In vitro permeation study
All formulations showed concentration-dependent inhibition of protein denaturation. F4 exhibited the highest inhibition (96.51% at 500 μg/ml), comparable to the marketed gel (97.23%).
Membrane stabilization assay (HRBC method)
The formulations also showed concentration-dependent membrane stabilization. F3 (96.49% at 500 μg/ml) was most effective, approaching the marketed formulation (97.11%).
Franz diffusion cell studies have demonstrated efficient transdermal drug delivery. The cumulative drug permeation increased over time for all the formulations (table 16). Flux (J) and permeation coefficients (Kp) were high, indicating enhanced skin penetration and potential for localized therapy.
CONCLUSION
In the present study, we successfully formulated and evaluated a topical nanoemulgel containing Indomethacin and Bromelain for localized anti-inflammatory therapy. Preformulation and compatibility studies confirmed the stability of both drugs with the selected excipients. The nanoemulgel formulations demonstrated desirable physicochemical properties, including appropriate globule size, PDI, pH, viscosity, spreadability, and extrudability. Among all formulations, F3 emerged as the most optimized, exhibiting the highest drug content for both actives, effective dual-drug permeation, and a sustained release profile. In vitro anti-inflammatory assays, including HRBC membrane stabilization and protein denaturation, confirmed significant activity comparable to that of the marketed indomethacin gel, likely due to the synergistic action of Indomethacin and Bromelain. Molecular docking studies further supported this synergistic potential by demonstrating the strong binding affinities of both drugs toward key inflammatory targets such as COX-2 and TNF-α. Overall, F3 offers a stable, effective topical formulation with enhanced therapeutic potential for the localized management of arthritis, providing a promising alternative to conventional topical therapies.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
All the authors have contributed equally
CONFLICT OF INTERESTS
The authors declare no conflict of interest
REFERENCES
Tanaka Y. Rheumatoid arthritis. Inflamm Regen. 2020 Sep 7;40(1):20. doi: 10.1186/s41232-020-00133-8.
Nithyashree RS, Deveswaran R. A comprehensive review on rheumatoid arthritis. J Pharm Res Int. 2020 Jul 27;32(12):18-32. doi: 10.9734/jpri/2020/v32i1230556.
Jahnavi K, Pavani Reddy P, Vasudha B, Narender B. Non-steroidal anti-inflammatory drugs: an overview. J Drug Deliv Ther. 2019 Feb 15;9(1-s):442-8. doi: 10.22270/jddt.v9i1-s.2287.
Brown P, Pratt AG, Hyrich KL. Therapeutic advances in rheumatoid arthritis. BMJ. 2024;384:e070856. doi: 10.1136/bmj-2022-070856, PMID 38233032.
Maeda K, Yoshida K, Nishizawa T, Otani K, Yamashita Y, Okabe H. Inflammation and bone metabolism in rheumatoid arthritis: molecular mechanisms of joint destruction and pharmacological treatments. Int J Mol Sci. 2022 Mar 1;23(5):2871. doi: 10.3390/ijms23052871, PMID 35270012.
Singh Malik D, Mital N, Kaur G. Topical drug delivery systems: a patent review. Expert Opin Ther Pat. 2016 Feb 1;26(2):213-28. doi: 10.1517/13543776.2016.1131267, PMID 26651499.
Suza HM, Kamal BA, Abdalrazaq NA, Rashid AM, Tbeekh HT, Hussein RG. A Review article: topical drug delivery system (skin). J Port Sci Res. 2024 Sep 7;7:558-63. doi: 10.36371/port.2024.special.44.
Bhardwaj S, Tiwari A. Nanoemulgel: a promising nanolipoidal-emulsion based drug delivery system in managing psoriasis. Dhaka Univ J Pharm Sci. 2021 Dec 29;20(2):235-46. doi: 10.3329/dujps.v20i2.57174.
Rai S, Singh N, Bhattacharya S. Concepts on smart nano-based drug delivery system. Recent Pat Nanotechnol. 2022;16(1):67-89. doi: 10.2174/1872210515666210120113738, PMID 33494685.
Gokhale JP, Mahajan HS, Surana SJ. Quercetin-loaded nanoemulsion-based gel for rheumatoid arthritis: in vivo and in vitro studies. Biomed Pharmacother. 2019 Apr 1;112:108622. doi: 10.1016/j.biopha.2019.108622, PMID 30797146.
Chen L, Wang Y, Sun L, Yan J, Mao HQ. Nanomedicine strategies for anti-inflammatory treatment of noninfectious arthritis. Adv Healthc Mater. 2021 Jun 1;10(11):e2001732. doi: 10.1002/adhm.202001732, PMID 33870656.
Golatkar SS, Rahate AC, Pomaje MD. Nanotechnology in topical drug delivery: enhancing efficacy and overcoming limitations. Int J Pharm Pharm Sci. 2025 Dec 1;17(12):1-6. doi: 10.22159/ijpps.2025v17i12.54986.
Hajwani A, Khan A, Ansari N, Zerdi N, Baig MS. Fabrication of transdermal gel embedded with solid lipid nanoparticles of indomethacin. Int J Drug Deliv Technol. 2023 Jan 1;13(1):414-7. doi: 10.25258/ijddt.13.1.66.
Badriyya E, Salman SS, Pratiwi AR, Dillasamola D, Aldi Y, Husni E. Topical anti-inflammatory activity of bromelain. Pharmacogn J. 2020 Nov 1;12(6s):1586-93. doi: 10.5530/pj.2020.12.217.
Rathnavelu V, Alitheen NB, Sohila S, Kanagesan S, Ramesh R. Potential role of bromelain in clinical and therapeutic applications. Biomed Rep. 2016 Sep;5(3):283-8. doi: 10.3892/br.2016.720, PMID 27602208.
Kadam PP, Nemade LS, Rahate AC. Herbal and enzymatic therapeutics in modern drug delivery: the case of bromelain. International Journal of Innovative Science and Research Technology (IJISRT). 2025 Sep 3;10(8):2121-8. doi: 10.38124/ijisrt/25aug1258.
Vishwakarma V, Sahu N, Bharadwaj A. Formulation and evaluation of TDDS of indomethacin containing natural penetration enhancer. Int J Adv Res Innov Ideas Educ. 2023;9(1):1375-83.
Jaiswal P, Aggarwal G, Harikumar SL, Singh K. Development of self-microemulsifying drug delivery system and solid-self-microemulsifying drug delivery system of telmisartan. Int J Pharm Investig. 2014;4(4):195-206. doi: 10.4103/2230-973X.143123, PMID 25426441.
Bhattacharya S, Prajapati BG. Formulation and optimization of celecoxib nanoemulgel. Asian J Pharm Clin Res. 2017;10(8):353-65. doi: 10.22159/ajpcr.2017.v10i8.19510.
Trott O, Olson AJ. Autodock vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization and multithreading. J Comput Chem. 2010 Jan 30;31(2):455-61. doi: 10.1002/jcc.21334, PMID 19499576.
Swetha M, Mohan B, Narsimha Rao R, Suresh Kumar JN, Sushma P. Formulation and evaluation of flubiprofen emulgel by using different concentration carbopol 974P. Saudi J Med Pharm. 2018;4(2):215-25. doi: 10.36348/sjmps.2018.v04i02.008.
Giri MA, Bhalke RD. Formulation and evaluation of topical anti-inflammatory herbal gel. Asian J Pharm Clin Res. 2019 May;12(7):252-5. doi: 10.22159/ajpcr.2019.v12i7.33859.
Das PJ, Athira A, Jasna MC, Fathima NM, Ameena A, Kamal BS. Development and evaluation of an anti-inflammatory multi-herbal extract with indigenous medicinal plants. JOPCR. 2025;24(2):89-97. doi: 10.18579/jopcr/v24.i2.51.
Leelaprakash G, Dass SM. In vitro anti-inflammatory activity of methanol extract of Enicostemma axillare. Int J Drug Dev Res. 2011;3(3):189–96.