
Int J Curr Pharm Res, Vol 18, Issue 2, 143-148Original Article
COMPARATIVE EFFICACY OF SODIUM–GLUCOSE COTRANSPORTER-2 INHIBITORS VERSUS PLACEBO ON CARDIOVASCULAR MORTALITY IN ADULTS WITH HEART FAILURE: A META-ANALYSIS OF RANDOMIZED CONTROLLED TRIALS
PRANAB DAS1, MAHAPRASAD PAL2, WASIM AKRAM3, BODHISATYA DAS4*
1Department of Pharmacology, Pragjyotishpur Medical College and Hospital, Guwahati, Assam, India. 2Department of Paediatrics, IPGMER and SSKM hospital Kolkata, Kolkata-700020, West Bengal, India. 3,4Department of Paediatrics, Burdwan Medical College and Hospital, Burdwan, West Bengal, India
*Corresponding author: Bodhisatya Das; *Email: bodhisatya33@gmail.com
Received: 11 Nov 2025, Revised and Accepted: 06 Jan 2026
ABSTRACT
Objective: To evaluate the effect of sodium–glucose cotransporter-2 (SGLT2) inhibitors compared with placebo on cardiovascular mortality in adults with heart failure using evidence derived exclusively from randomized controlled trials.
Methods: A systematic review and meta-analysis of randomized controlled trials was conducted in accordance with PRISMA 2020 guidelines. Electronic databases were searched to identify eligible trials enrolling adult patients with heart failure that directly compared an SGLT2 inhibitor with placebo and reported cardiovascular mortality. Data were extracted from full-text publications, and risk ratios (RRs) with 95% confidence intervals (CIs) were pooled using an inverse-variance fixed-effect model. Statistical heterogeneity was assessed using Cochran’s Q test and the I² statistic.
Results: Four large, double-blind randomized controlled trials comprising 20,725 participants met the inclusion criteria. Across individual studies, SGLT2 inhibitor therapy was consistently associated with a lower risk of cardiovascular death compared with placebo. The pooled analysis demonstrated a statistically significant reduction in cardiovascular mortality with SGLT2 inhibitors (pooled RR = 0.88; 95% CI: 0.81–0.96). Statistical heterogeneity was negligible (I² = 0%), indicating high consistency across trials.
Conclusion: This meta-analysis demonstrates that SGLT2 inhibitors significantly reduce cardiovascular mortality in adults with heart failure. The consistency of benefit across heart-failure phenotypes and the low heterogeneity support the robustness of these findings and reinforce the role of SGLT2 inhibitors as a foundational component of contemporary heart-failure therapy.
Keywords: Heart failure, SGLT2 inhibitors, Cardiovascular mortality, Randomized controlled trials, Meta-analysis
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i2.8085 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Heart failure remains a major global public health challenge, affecting more than 64 million individuals worldwide and accounting for substantial morbidity, mortality, and health-care expenditure [1]. Despite advances in pharmacological and device-based therapies, cardiovascular mortality among patients with heart failure remains unacceptably high, particularly in those with chronic symptomatic disease [2]. The heterogeneity of heart-failure phenotypes and the complex interplay of hemodynamic, metabolic, renal, and neurohormonal pathways contribute to persistent adverse outcomes, underscoring the need for therapies that provide consistent prognostic benefit across diverse patient populations.
Sodium–glucose cotransporter-2 (SGLT2) inhibitors were initially developed as glucose-lowering agents for type 2 diabetes mellitus; however, accumulating evidence has demonstrated that their cardiovascular benefits extend well beyond glycaemic control [3]. Experimental and clinical studies suggest that SGLT2 inhibitors exert pleiotropic effects, including osmotic diuresis, natriuresis, reduction in preload and afterload, improvement in myocardial energetics, attenuation of cardiac remodelling, and favourable renal–cardiac interactions [4, 5]. These mechanisms have positioned SGLT2 inhibitors as an important therapeutic class in contemporary heart-failure management, irrespective of diabetic status.
Although multiple large randomized controlled trials have evaluated SGLT2 inhibitors in patients with heart failure, individual trials were primarily powered for composite outcomes rather than cardiovascular mortality alone [6]. Consequently, estimates of treatment effect on cardiovascular death have varied modestly across studies and heart-failure phenotypes. In clinical practice, cardiovascular mortality remains one of the most clinically meaningful endpoints, directly reflecting disease severity and therapeutic effectiveness. From a decision-making perspective, clinicians and guideline developers require precise and consolidated estimates of mortality benefit to guide therapy selection and prioritize foundational treatments.
Meta-analysis offers a robust methodological approach to synthesize evidence across randomized trials, increase statistical power, and provide more stable estimates of treatment effects. Importantly, when restricted to large, high-quality randomized controlled trials with comparable designs and outcome definitions, pooled analyses can clarify clinically relevant benefits while minimizing bias and heterogeneity [7]. Given the growing incorporation of SGLT2 inhibitors into guideline-directed medical therapy and their expanding indications across the heart-failure spectrum, a focused meta-analysis examining cardiovascular mortality is both timely and clinically relevant.
Therefore, the present meta-analysis was undertaken to quantitatively evaluate the effect of SGLT2 inhibitors compared with placebo on cardiovascular mortality in adults with heart failure using evidence derived exclusively from randomized controlled trials.
MATERIALS AND METHODS
Research question
The present study was designed to address the following research question using the PICO framework: In adults with heart failure (Population), does treatment with sodium–glucose cotransporter-2 (SGLT2) inhibitors (Intervention), compared with placebo (Comparator), reduce cardiovascular mortality (Outcome)? This focused question was formulated to ensure clinical relevance and methodological clarity, with an emphasis on a hard clinical endpoint that is directly meaningful to patients and clinicians.
Study design and reporting standards
This investigation was conducted as a systematic review and meta-analysis of randomized controlled trials. The study methodology and reporting were developed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 statement to ensure transparency, reproducibility, and completeness of reporting [8]. The overall analytical approach followed established recommendations outlined in the Cochrane Handbook for Systematic Reviews of Interventions (version 6.4), particularly with respect to study selection, data synthesis, and assessment of heterogeneity [7].
Eligibility criteria
Eligible studies were selected based on predefined inclusion and exclusion criteria structured around the PICO framework. Randomized controlled trials enrolling adult participants (≥18 y) with a clinical diagnosis of heart failure were considered eligible. Trials were required to directly compare an SGLT2 inhibitor with placebo in addition to standard background therapy. Only studies reporting cardiovascular mortality as an outcome and those published as full-text articles in peer-reviewed journals in English language were included to ensure methodological quality and data completeness. To maintain methodological rigor and comparability, observational studies, non-randomized trials, post hoc analyses, subgroup analyses, conference abstracts, editorials, and review articles were excluded. No restrictions were applied with respect to sex, geographic location, or duration of follow-up, provided that outcome data were available.
Search strategy
A comprehensive literature search was performed using electronic databases including PubMed, Scopus, and Google Scholar. The search strategy combined Medical Subject Headings (MeSH) terms and free-text keywords related to heart failure, SGLT2 inhibitors, and randomized trials. The following Boolean search string was used for PubMed:
(“heart failure” or “cardiac failure” or “heart failure with reduced ejection fraction” or “heart failure with preserved ejection fraction”) and (“SGLT2 inhibitor” or “dapagliflozin” or “empagliflozin”) and (“randomized controlled trial” or “randomised controlled trial”) and (“cardiovascular mortality” or “cardiovascular death”).
Searches were limited to full-text articles published in English and involving human participants. Reference lists of relevant reviews and guidelines were also manually screened to identify additional eligible studies. The final search was completed prior to data extraction.
Study selection process
All records identified through the electronic search were initially screened by title and abstract to exclude clearly irrelevant studies. Full-text versions of potentially eligible articles were then retrieved and independently assessed for inclusion based on the predefined eligibility criteria. The study selection process was conducted systematically by two independent reviewers, and only trials meeting all eligibility requirements were included in the final analysis. Discrepancies during the study selection process were resolved through discussion and consensus with a third independent reviewer. The overall study selection process followed a structured, stepwise approach consistent with PRISMA recommendations [9].
Data extraction process
Statistical analysis
Statistical analyses were conducted using standard meta-analytic techniques as recommended by the Cochrane Handbook [8]. Cardiovascular mortality was treated as a dichotomous outcome, and treatment effects were expressed as risk ratios (RRs) with corresponding 95% confidence intervals (CIs). Log-transformed RRs were pooled using an inverse-variance fixed-effect model. This model was selected a priori due to the inclusion of a small number of large, high-quality randomized trials with similar study designs and minimal clinical and methodological heterogeneity. Statistical heterogeneity was assessed using Cochran’s Q statistic and quantified with the I² statistic. In accordance with established convention, negative I² values were truncated to zero, reflecting negligible heterogeneity [8]. Sensitivity analyses and publication bias assessments, including funnel plot visualization and Egger’s regression test, were not performed because fewer than ten studies were included, a threshold below which such analyses are considered unreliable and potentially misleading [8]. All calculations were performed using a predefined Excel-based analytical framework consistent with standard meta-analysis methodology.
RESULTS
Study selection and sample characteristics
The systematic literature search yielded a total of 154 records from electronic databases. After removal of duplicate 91 records, the remaining articles were screened based on titles and abstracts. Full-text articles were retrieved for studies that met the predefined eligibility criteria. Following detailed full-text evaluation, four randomized controlled trials (RCTs), with n = 20725 participants, fulfilled all inclusion criteria and were included in both qualitative and quantitative synthesis. These trials provided outcome data suitable for pooled analysis of cardiovascular mortality and formed the final evidence base for this meta-analysis [9–12]. PRISMA flow diagram summarizing the study selection process is presented in table 1.
Table 1: PRISMA summary table
| Stage | Frequency (n) |
| Records identified through database searching | 154 |
| Duplicates removed | 91 |
| Records screened | 63 |
| Records excluded | 48 |
| Full-text articles assessed for eligibility | 15 |
| Full-text articles excluded | 11 |
| Studies included in qualitative and quantitative synthesis | 4 |
Study characteristics
The four included randomized controlled trials were large, multicenter, double-blind studies enrolling adult patients with chronic heart failure across the spectrum of left ventricular ejection fraction. Two trials primarily enrolled patients with heart failure with reduced ejection fraction, while the remaining trials included patients with mildly reduced or preserved ejection fraction. All trials compared an SGLT2 inhibitor with matching placebo on top of guideline-directed medical therapy and reported cardiovascular mortality as a prespecified outcome [9–12]. Details of study attributes which were included in this meta-analysis are depicted in table 2.
Table 2: Attributes of the included randomized controlled trials (RCTs)
| Study ID | First author (Year) | Study design | Population | Intervention | Comparator | Intervention dose | Comparator dose | Primary outcome | Percentage of patients achieving target outcome (Intervention)* | Percentage of patients achieving target outcome (Comparator)** | Duration (weeks) |
| Dapa-HF | McMurray (2019) [9] | Double-blind RCT | Chronic HFrEF (LVEF ≤40%) | Dapagliflozin | Placebo | 10 mg OD | Matching placebo | Cardiovascular death | 227/2373 (9.6%) | 273/2371 (11.5%) | 79 |
| Emperor-Reduced | Packer (2020) [10] | Double-blind RCT | Chronic HFrEF (LVEF ≤40%) | Empagliflozin | Placebo | 10 mg OD | Matching placebo | Cardiovascular death | 187/1863 (10.0%) | 202/1867 (10.8%) | 69 |
| Emperor-Preserved | Anker (2021) [11] | Double-blind RCT | Chronic HFpEF (LVEF>40%) | Empagliflozin | Placebo | 10 mg OD | Matching placebo | Cardiovascular death | 219/2997 (7.3%) | 244/2991 (8.9%) | 114 |
| Deliver | Solomon (2022) [12] | Double-blind RCT | HFmrEF/HFpEF (LVEF>40%) | Dapagliflozin | Placebo | 10 mg OD | Matching placebo | Cardiovascular death | 231/3131 (7.4%) | 261/3132 (8.3%) | 120 |
*Percentage of patients achieving target outcome (cardiovascular death) in intervention group = Sample size of intervention group showing the desired outcome/Total sample size of intervention group. **Percentage of patients achieving target outcome (cardiovascular death) in Comparator group = Sample size of comparator group showing the desired outcome/Total sample size of comparator group.
Meta-analysis findings
All four trials demonstrated a numerically lower risk of cardiovascular death in patients treated with SGLT2 inhibitors compared with placebo. When pooled using an inverse-variance fixed-effect model, treatment with an SGLT2 inhibitor was associated with a statistically significant reduction in cardiovascular mortality. The pooled risk ratio (pooled RR) was 0.88, corresponding to a 12% relative reduction in mortality, with a 95% confidence interval (CI) ranging from 0.81 to 0.96 (statistically significant). The effect estimates of the meta-analysis are summarised below in table 3. The pooled risk ratio indicated a consistent benefit across studies, with narrow confidence intervals reflecting the large sample size and precision of the included trials [9–12].
Table 3: Summary of effect estimates of the meta-analysis
| Study ID | RR | SE | Lower 95% CI (Individual study) | Upper 95% CI (Individual study) | Pooled RR | SE of pooled Log(RR) | Lower 95% CI (Meta-analysis) | Upper 95% CI (Meta-analysis) |
| DAPA-HF | 0.83 | 0.085 | 0.70 | 0.98 | 0.88 | 0.04 | 0.81 | 0.96 |
| Emperor-reduced | 0.93 | 0.096 | 0.77 | 1.12 | ||||
| Emperor-preserved | 0.90 | 0.089 | 0.75 | 1.07 | ||||
| Deliver | 0.88 | 0.087 | 0.75 | 1.05 |
RR: Risk Ratio; SE: Standard Error; CI: Confidence Interval.
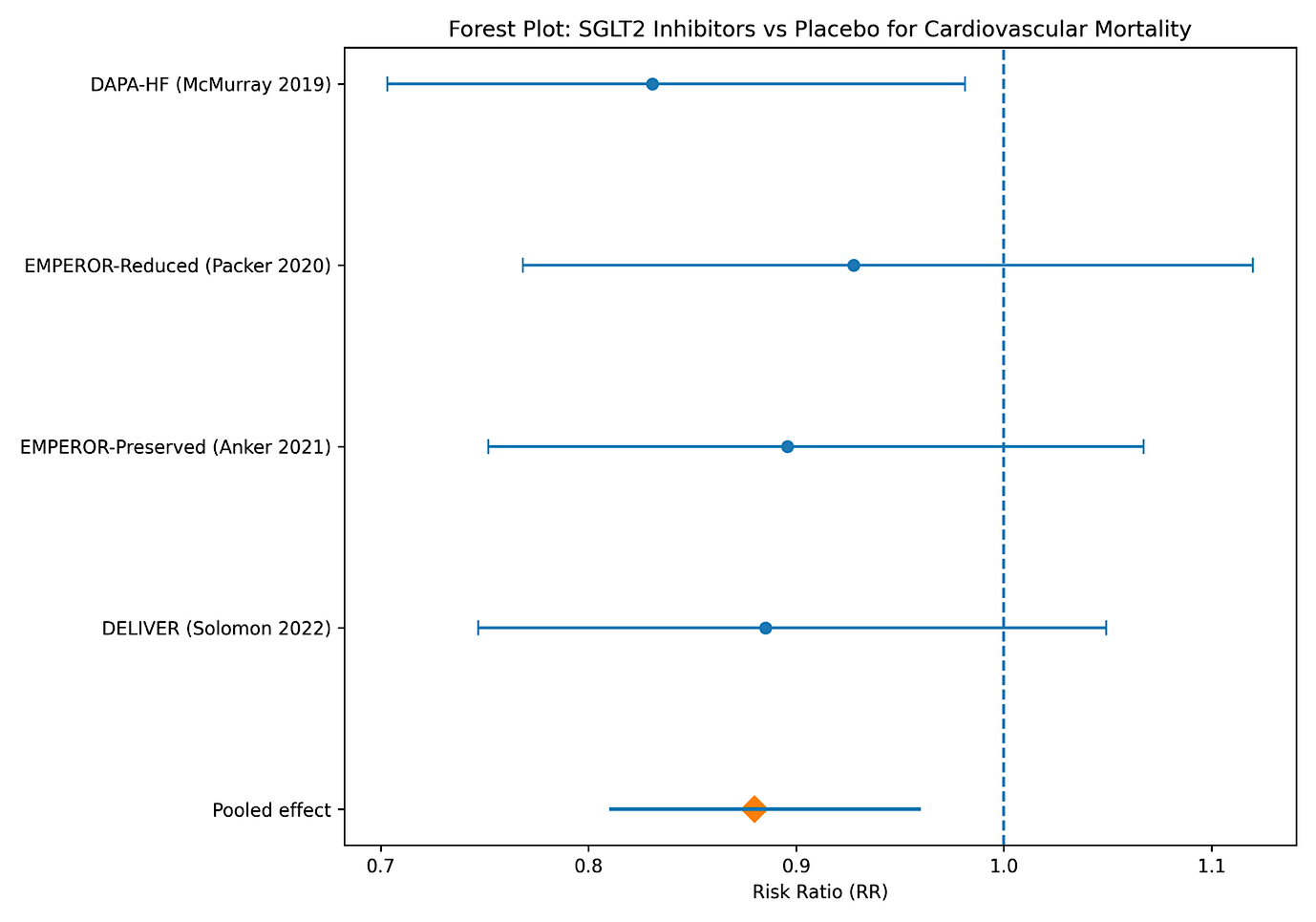
Fig. 1: Forest plot
Forest plot
The forest plot demonstrates a consistent trend toward reduced cardiovascular mortality with SGLT2 inhibitor therapy compared with placebo across all included randomized controlled trials. Although the confidence intervals of individual studies vary in width and, in some cases, cross the line of no effect (EMPEROR-Reduced, EMPEROR-Preserved and DELIVER trials), the direction of effect uniformly favours SGLT2 inhibition. The pooled estimate, represented by the diamond along with the line of confidence interval (CI), lies entirely to the left of unity, indicating a statistically significant overall reduction in cardiovascular mortality. The relatively narrow pooled confidence interval reflects improved precision achieved through aggregation of large, well-conducted trials. Collectively, the visual alignment of individual study estimates with the pooled effect supports the robustness of the overall finding and suggests minimal between-study heterogeneity.
Risk of bias assessment
Risk of bias assessment was conducted using the Cochrane Risk of Bias tool [13]. All included trials employed appropriate random sequence generation, allocation concealment, and double-blinding of participants and personnel. Outcome assessment was adjudicated by blinded clinical events committees. Attrition rates were low and balanced between groups, and all trials reported prespecified outcomes. Overall, the risk of bias was judged to be low across all domains for each included study [9–12].
Heterogeneity assessment
Statistical heterogeneity among the included studies was minimal. Cochran’s Q statistic was lower than the degrees of freedom, and the I² statistic was calculated as 0% after truncation of negative values, indicating negligible between-study variability.
Table 4: Risk of bias (RoB) assessment for the included RCTs
| Study ID | Randomization process | Allocation concealment | Blinding of participants and personnel | Blinding of outcome assessment | Incomplete outcome data | Selective reporting | Overall risk of bias |
| DAPA-HF | Low | Low | Low | Low | Low | Low | Low |
| EMPEROR-Reduced | Low | Low | Low | Low | Low | Low | Low |
| EMPEROR-Preserved | Low | Low | Low | Low | Low | Low | Low |
| DELIVER | Low | Low | Low | Low | Low | Low | Low |
Table 5: Heterogeneity assessment of included studies
| Study ID | ((Log RR-Pooled log RR)^2)*weight | Cochran's Q statistic | Degrees of freedom (df) | I2 statistic |
| DAPA-HF | 0.48 | 2.77 | 3 | 0% |
| EMPEROR-Reduced | 0.28 | |||
| EMPEROR-Preserved | 0.03 | |||
| DELIVER | 1.97 |
GRADE assessment
Using the GRADE framework, the certainty of evidence for cardiovascular mortality was rated as high. This rating was supported by the inclusion of multiple large, well-conducted randomized controlled trials with low risk of bias, directness of evidence, consistency of findings, and precise effect estimates.
Table 6: GRADE assessment of the meta-analysis
| Domain | Assessment |
| Risk of bias | Not serious |
| Inconsistency | Not serious |
| Indirectness | Not serious |
| Imprecision | Not serious |
| Publication bias | Undetected |
| Overall certainty of evidence | High |
DISCUSSION
This meta-analysis provides a comprehensive and methodologically robust synthesis of randomized evidence evaluating the impact of SGLT2 inhibitors on cardiovascular mortality in adults with heart failure. By restricting inclusion to large, contemporary randomized controlled trials and focusing on cardiovascular death as a definitive endpoint, the present study addresses a critical evidence gap in heart-failure therapeutics. The pooled findings demonstrate a statistically significant and clinically meaningful reduction in cardiovascular mortality, reinforcing the role of SGLT2 inhibitors as a foundational therapy across the heart-failure spectrum.
Despite major advances in neurohormonal modulation and device therapy, heart failure remains associated with high residual cardiovascular mortality, even under optimal guideline-directed medical therapy [1, 2]. The present meta-analysis demonstrates that SGLT2 inhibitors confer a survival benefit that is additive to established therapies. While individual trials were not uniformly powered to detect cardiovascular mortality differences as isolated endpoints, the consistent direction of effect across trials and the statistically significant pooled estimate strongly support a true mortality benefit.
Importantly, the observed reduction in cardiovascular death was evident across heterogeneous patient populations, including those with reduced, mildly reduced, and preserved ejection fraction. This finding challenges the traditional paradigm that effective mortality-reducing therapies in heart failure are limited primarily to reduced ejection fraction and highlights the evolving understanding of heart failure as a systemic cardiometabolic disorder rather than a purely hemodynamic condition.
The mortality benefit observed with SGLT2 inhibitors is supported by substantial mechanistic plausibility. Beyond their glucose-lowering effects, SGLT2 inhibitors induce osmotic diuresis and natriuresis, resulting in reductions in interstitial fluid volume and ventricular wall stress without the neurohormonal activation commonly seen with conventional diuretics [3, 4]. These effects may translate into improved cardiac efficiency and reduced myocardial oxygen demand.
At the myocardial level, SGLT2 inhibitors have been shown to alter cardiac metabolism by promoting ketone body utilization, which may enhance myocardial energy efficiency under conditions of chronic stress [4, 14]. Experimental data further suggest reductions in myocardial fibrosis, inflammation, and oxidative stress, processes that are central to adverse ventricular remodelling and progressive heart-failure deterioration [5, 15].
Renal protection represents another critical mechanism linking SGLT2 inhibition to improved cardiovascular outcomes. Chronic kidney disease is a powerful predictor of mortality in heart failure, and stabilization of renal function may interrupt the deleterious cardiorenal cycle that accelerates disease progression [5, 16]. The integration of cardiac and renal protective effects likely contributes to the consistent mortality benefit observed across diverse patient populations.
Several prior meta-analyses have demonstrated reductions in heart-failure hospitalization and composite cardiovascular outcomes with SGLT2 inhibitors [3, 6]. However, many of these analyses pooled heterogeneous endpoints or included observational data, limiting their ability to provide definitive conclusions regarding cardiovascular mortality. By contrast, the present meta-analysis isolates cardiovascular death and restricts inclusion to randomized controlled trials, thereby enhancing internal validity and clinical relevance.
The findings are highly concordant with contemporary international guidelines, which now recommend SGLT2 inhibitors as core therapy for heart failure irrespective of diabetic status [6]. Importantly, this analysis provides quantitative mortality data that strengthen the evidentiary foundation underlying these recommendations and may facilitate broader clinical adoption.
This study has several notable strengths. First, the exclusive inclusion of large, double-blind randomized controlled trials minimizes selection bias and confounding. Second, the focus on cardiovascular mortality ensures clinical relevance and avoids ambiguity associated with composite endpoints. Third, the low degree of statistical heterogeneity supports the validity of the pooled estimate and justifies the use of a fixed-effect model. Fourth, adherence to PRISMA 2020 reporting standards and Cochrane methodological guidance enhances transparency and reproducibility [7, 8]. Finally, the use of contemporary trials reflecting modern background therapy improves the generalizability of the findings to current clinical practice.
Several limitations warrant consideration. The analysis was conducted using trial-level data, which precluded patient-level subgroup analyses based on age, sex, renal function, or comorbidity burden. The small number of included trials limited the feasibility of formal sensitivity analyses and publication-bias assessment. Differences in follow-up duration and background therapy may have influenced absolute event rates, although relative treatment effects remained consistent across trials.
From a clinical standpoint, the findings of this meta-analysis have immediate implications. The demonstrated reduction in cardiovascular mortality supports early initiation of SGLT2 inhibitors as part of foundational therapy in heart-failure management. Given their favourable safety profile, oral administration, and compatibility with existing therapies, SGLT2 inhibitors represent a practical and impactful intervention capable of improving survival in a broad patient population. These findings may also inform shared decision-making and reinforce clinician confidence in prioritizing SGLT2 inhibitors early in the treatment algorithm.
Future research should prioritize individual patient-level meta-analyses to identify subgroups that derive the greatest mortality benefit. Longer follow-up studies are needed to evaluate the durability of benefit and potential late effects. Additionally, mechanistic studies integrating cardiac imaging, biomarker profiling, and metabolic assessment may further elucidate pathways of benefit and guide precision-based therapeutic strategies. Finally, pragmatic and implementation-focused studies will be essential to translate trial efficacy into real-world effectiveness at the population level.
CONCLUSION
This meta-analysis demonstrates that treatment with SGLT2 inhibitors is associated with a significant reduction in cardiovascular mortality among adults with heart failure. By synthesizing evidence exclusively from large, high-quality randomized controlled trials, the present study provides a precise and clinically meaningful estimate of mortality benefit across a broad spectrum of heart-failure phenotypes. The consistency of findings, coupled with minimal statistical heterogeneity, supports the robustness of the pooled effect. These results reinforce the role of SGLT2 inhibitors as a foundational component of guideline-directed medical therapy and highlight their importance in improving long-term cardiovascular outcomes in patients with heart failure.
ACKNOWLEDGEMENT
The authors would like to acknowledge the investigators and participants of the original randomized controlled trials included in this meta-analysis, whose contributions made this work possible. No additional individuals or organizations contributed directly to the conduct or reporting of this study.
FUNDING
This study did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
AUTHORS CONTRIBUTIONS
Pranab Das and Mahaprasad Pal conceptualized and designed the study, performed the literature search, extracted and analysed the data, and drafted the manuscript. Wasim Akram and Bodhisatya Das assisted with data verification, interpretation of results, and critical revision of the manuscript. Both authors reviewed and approved the final version and accept full responsibility for the accuracy and integrity of the work.
CONFLICT OF INTERESTS
The authors declare no conflicts of interest relevant to this study.
REFERENCES
Savarese G, Lund LH. Global public health burden of heart failure. Card Fail Rev. 2017;3(1):7-11. doi: 10.15420/cfr.2016:25:2, PMID 28785469.
Roger VL. Epidemiology of heart failure: a contemporary perspective. Circ Res. 2021;128(10):1421-34. doi: 10.1161/circresaha.121.318172, PMID 33983838.
Zelniker TA, Braunwald E. Clinical benefit of cardiorenal effects of sodium-glucose cotransporter 2 inhibitors: JACC state-of-the-art review. J Am Coll Cardiol. 2020;75(4):435-47. doi: 10.1016/j.jacc.2019.11.036, PMID 32000956.
Verma S, McMurray JJ. SGLT2 inhibitors and mechanisms of cardiovascular benefit: a state-of-the-art review. Diabetologia. 2018;61(10):2108-17. doi: 10.1007/s00125-018-4670-7, PMID 30132036.
Packer M. Critical reanalysis of the mechanisms underlying the cardiorenal benefits of SGLT2 inhibitors and reaffirmation of the nutrient deprivation signaling/autophagy hypothesis. Circulation. 2022;146(18):1383-405. doi: 10.1161/circulationaha.122.061732, PMID 36315602.
McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Bohm M. 2021 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2021;42(36):3599-726. doi: 10.1093/eurheartj/ehab368, PMID 34447992.
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, editors. Cochrane handbook for systematic reviews of interventions. Version 6.4. London (UK): Cochrane; 2023. Available from: https://training.cochrane.org/handbook.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. doi: 10.1136/bmj.n71, PMID 33782057.
McMurray JJ, Solomon SD, Inzucchi SE, Kober L, Kosiborod MN, Martinez FA. Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med. 2019;381(21):1995-2008. doi: 10.1056/NEJMoa1911303, PMID 31535829.
Packer M, Anker SD, Butler J, Filippatos G, Pocock SJ, Carson P. Cardiovascular and renal outcomes with empagliflozin in heart failure. N Engl J Med. 2020;383(15):1413-24. doi: 10.1056/NEJMoa2022190, PMID 32865377.
Anker SD, Butler J, Filippatos G, Ferreira JP, Bocchi E, Bohm M. Empagliflozin in heart failure with a preserved ejection fraction. N Engl J Med. 2021;385(16):1451-61. doi: 10.1056/NEJMoa2107038, PMID 34449189.
Solomon SD, McMurray JJ, Claggett B, De Boer RA, De Mets D, Hernandez AF. Dapagliflozin in heart failure with mildly reduced or preserved ejection fraction. N Engl J Med. 2022;387(12):1089-98. doi: 10.1056/NEJMoa2206286, PMID 36027570.
Sterne JAC, Higgins JPT, Elbers RG, Reeves BC. Risk of bias 2: a tool to assess risk of bias in randomised trials. Version 2.0. London (UK): Cochrane; 2019. Available from: https://www.riskofbias.info/welcome/rob-2-0-tool/current-version-of-rob-2.
Ferrannini E, Mark M, Mayoux E. CV protection in the EMPA-REG outcome trial: a “thrifty substrate” hypothesis. Diabetes Care. 2016;39(7):1108-14. doi: 10.2337/dc16-0330, PMID 27289126.
Santos Gallego CG, Requena Ibanez JA, San Antonio R, Ishikawa K, Watanabe S, Picatoste B, Flores E, Garcia Ropero A, Sanz J, Hajjar RJ, Fuster V, Badimon JJ. Empagliflozin ameliorates adverse left ventricular remodeling in nondiabetic heart failure by enhancing myocardial energetics. J Am Coll Cardiol. 2019;73(15):1931-44. doi: 10.1016/j.jacc.2019.01.056, PMID: 30999996.
Rangaswami J, Bhalla V, Blair JE, Chang TI, Costa S, Lentine KL. Cardiorenal syndrome: classification pathophysiology diagnosis and treatment strategies: a scientific statement from the American heart association. Circulation. 2019;139(16):e840-78. doi: 10.1161/CIR.0000000000000664, PMID 30852913.