
Int J Curr Pharm Res, Vol 18, Issue 2, 149-154Original Article
EFFECT OF MINERALOCORTICOID RECEPTOR ANTAGONISTS ON CARDIOVASCULAR MORTALITY IN ADULTS WITH HEART FAILURE: AMETA-ANALYSIS OF RANDOMIZED CONTROLLED TRIALS
PRANAB DAS1, MAHAPRASAD PAL2, WASIM AKRAM3, BODHISATYA DAS4*
1Department of Pharmacology, Pragjyotishpur Medical College and Hospital, Guwahati, Assam, India. 2Department of Paediatrics, IPGMER and SSKM hospital Kolkata, Kolkata-700020, West Bengal, India. 3,4Department of Paediatrics, Burdwan Medical College and Hospital, Burdwan, West Bengal, India
*Corresponding author: Bodhisatya Das; *Email: bodhisatya33@gmail.com
Received: 13 Nov 2025, Revised and Accepted: 09 Jan 2026
ABSTRACT
Objective: To evaluate the effect of mineralocorticoid receptor antagonists on cardiovascular mortality in adults with heart failure using evidence from randomized controlled trials.
Methods: A systematic review and meta-analysis of randomized controlled trials was conducted in accordance with PRISMA 2020 guidelines. Electronic databases, including PubMed, Scopus, Google Scholar, and the Cochrane Central Register of Controlled Trials were searched for eligible studies. Trials enrolling adult patients with heart failure and comparing MRAs with placebo were included. Cardiovascular mortality was the primary outcome. Risk ratios (RRs) with 95% confidence intervals (CIs) were calculated for individual studies and pooled using an inverse-variance fixed-effect model. Statistical heterogeneity was evaluated using Cochran’s Q test and the I² statistic. Risk of bias was assessed using the Cochrane Risk of Bias tool, and certainty of evidence was evaluated using the GRADE framework.
Results: Three large, double-blind randomized controlled trials comprising 11,032 patients met the inclusion criteria. Treatment with MRAs was associated with a significant reduction in cardiovascular mortality compared with placebo. The pooled analysis demonstrated a 20% relative reduction in cardiovascular mortality (pooled RR = 0.80; 95% CI: 0.73–0.86). No significant heterogeneity was observed among the included studies (I² = 0%). All trials were judged to have a low risk of bias, and the certainty of evidence for the primary outcome was rated as high.
Conclusion: Mineralocorticoid receptor antagonists significantly reduce cardiovascular mortality in patients with heart failure. These findings provide high-certainty evidence supporting their use as an essential component of guideline-directed medical therapy.
Keywords: Heart failure, Mineralocorticoid receptor antagonists, Cardiovascular mortality, Randomized controlled trials, Meta-analysis, Spironolactone, Eplerenone
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i2.8086 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Heart failure is a complex clinical syndrome associated with substantial morbidity and mortality and represents a major public health burden worldwide. Despite significant advances in pharmacological therapy and device-based interventions, cardiovascular mortality remains unacceptably high among patients with heart failure, particularly those with reduced ejection fraction [1, 2]. Cardiovascular death, driven by progressive pump failure and fatal arrhythmias, accounts for the majority of adverse outcomes in this population and continues to be a key target for therapeutic intervention.
Activation of the renin–angiotensin–aldosterone system plays a central role in the pathophysiology of heart failure. Aldosterone excess contributes to sodium and water retention, myocardial fibrosis, endothelial dysfunction, inflammation, and adverse ventricular remodeling, all of which promote disease progression and increase the risk of cardiovascular death [3, 4]. Importantly, aldosterone escape may occur despite treatment with angiotensin-converting enzyme inhibitors or angiotensin receptor blockers, leading to persistent mineralocorticoid receptor activation and ongoing cardiovascular risk [5].
Mineralocorticoid receptor antagonists (MRAs), including spironolactone and eplerenone, directly counteract the deleterious effects of aldosterone by blocking mineralocorticoid receptor–mediated signaling. Through antifibrotic, antiarrhythmic, and vasculoprotective mechanisms, MRAs have demonstrated clinically meaningful benefits in patients with heart failure across a range of disease severities [6]. Consequently, contemporary international guidelines recommend MRAs as a cornerstone of guideline-directed medical therapy for patients with heart failure with reduced ejection fraction, and in selected patients with mildly reduced or preserved ejection fraction [7].
Although several large randomized controlled trials have evaluated the efficacy of MRAs in heart failure, individual studies differ with respect to patient populations, clinical settings, and reported outcomes. Cardiovascular mortality is variably reported as a primary or prespecified secondary endpoint, and the magnitude of benefit has not been uniformly quantified across trials. A focused synthesis of randomized evidence is therefore essential to provide a precise and reliable estimate of the effect of MRAs on cardiovascular mortality.
Systematic reviews and meta-analyses restricted to randomized controlled trials offer a robust methodological approach to summarize available evidence, enhance statistical power, and reduce uncertainty surrounding clinically important outcomes [8]. By pooling cardiovascular mortality data from high-quality trials, such analyses can strengthen the evidence base supporting the use of MRAs and inform both clinical practice and guideline development. The objective of this evidence-based synthesis was to evaluate the effect of mineralocorticoid receptor antagonists compared with placebo on cardiovascular mortality in adults with heart failure.
MATERIALS AND METHODS
Research question
This systematic review and meta-analysis was conducted to address the following research question: Do mineralocorticoid receptor antagonists (MRAs) reduce cardiovascular mortality compared with placebo or standard therapy in adults with heart failure? The question was formulated using the Population (adults with heart failure), Intervention (MRAs), Comparator (placebo), and Outcome (cardiovascular mortality) (PICO) framework to ensure clinical relevance and methodological clarity.
Study design and reporting standards
The present study was designed as a systematic review and meta-analysis of randomized controlled trials. The conduct and reporting of this meta-analysis adhered strictly to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 statement, ensuring transparency, completeness, and reproducibility of the review process [9]. Methodological guidance from the Cochrane Handbook for Systematic Reviews of Interventions was followed throughout the study design, data extraction, and statistical analysis phases [8].
Eligibility criteria
Eligible studies were randomized controlled trials enrolling adult patients (≥18 y) diagnosed with heart failure, irrespective of aetiology, provided cardiovascular mortality outcomes were reported. Trials were required to compare a mineralocorticoid receptor antagonist with placebo or standard therapy. Only studies reporting cardiovascular mortality as a primary or prespecified secondary outcome and those published as full-text articles in peer-reviewed journals in English language were included to ensure methodological quality and data completeness. Observational studies, non-randomized trials, crossover studies, post hoc analyses, conference abstracts, and studies without extractable cardiovascular mortality data were excluded. Trials involving paediatric populations or non-heart failure cohorts were also excluded. No restrictions were applied with respect to sex, geographic location, or duration of follow-up, provided that outcome data were available.
Search strategy
A comprehensive literature search was performed using electronic databases, including PubMed, Scopus, Google Scholar, and the Cochrane Central Register of Controlled Trials (central). The search strategy combined Medical Subject Headings (MeSH) terms and free-text keywords related to heart failure, mineralocorticoid receptor antagonists, and cardiovascular mortality. Boolean operators were applied as follows:
(“Heart failure” or “cardiac failure”) and (“mineralocorticoid receptor antagonist” or “aldosterone antagonist” or “spironolactone” or “eplerenone”) and (“cardiovascular mortality” or “cardiovascular death”).
The search was limited to studies published in English and conducted up to the most recent search date. Reference lists of relevant articles and reviews were also manually screened to identify additional eligible studies.
Study selection process
All records identified through the database search were imported into a Microsoft Excel 365 master sheet (data extraction form), and duplicate entries were removed. Two reviewers independently screened titles and abstracts to assess potential eligibility. Full-text articles were retrieved for studies that met the inclusion criteria or where eligibility could not be determined from the abstract alone. Disagreements during the selection process were resolved through discussion and consensus with a third independent reviewer. The final selection of studies was documented using a PRISMA flow diagram.
Data extraction process
Statistical analysis
The primary outcome of interest was cardiovascular mortality. For each included study, risk ratios (RRs) with corresponding 95% confidence intervals (CIs) were calculated. Pooled effect estimates were generated using an inverse-variance fixed-effect model, selected a priori due to the inclusion of large, high-quality randomized controlled trials with similar study designs and outcome definitions. Statistical heterogeneity was assessed using Cochran’s Q statistic and quantified using the I² statistic, with values below 25% considered indicative of low heterogeneity [8]. A significance threshold of p<0.05 was applied for all analyses. Given the small number of included studies, formal assessment of publication bias using funnel plots or Egger’s regression test was not performed, in line with methodological recommendations [10]. All statistical analyses were conducted using Microsoft Excel 365 by following the guidelines and formulas mentioned in the textbook “Meta-Analysis in Excel: A Beginner’s Guide [11].
RESULTS
Study selection and sample characteristics
The systematic literature search identified randomized controlled trials evaluating the effect of MRAs on cardiovascular outcomes in patients with heart failure. After removal of duplicates and full-text screening, three large, multicentre, double-blind randomized controlled trials fulfilled the predefined eligibility criteria and were included in the quantitative synthesis. These trials were the randomized aldactone evaluation study (RALES), the eplerenone post–acute myocardial infarction heart failure efficacy and survival study (EPHESUS), and the eplerenone in mild patients hospitalization and survival study in heart failure (EMPHASIS-HF) [12-14]. PRISMA flow summarizing the study scrutiny process is depicted in table 1.
Table 1: PRISMA summary table
| Stage | Frequency (n) |
| Records identified through database searching | 125 |
| Duplicates removed | 73 |
| Records screened | 52 |
| Records excluded | 39 |
| Full-text articles assessed for eligibility | 13 |
| Full-text articles excluded (due to non-RCT design, non-cardiovascular mortality outcome) | 10 |
| Studies included in qualitative and quantitative synthesis | 3 |
Study characteristics
The three included trials collectively enrolled 11,032 patients with heart failure across a broad spectrum of clinical severity and aetiologies. RALES enrolled patients with severe chronic heart failure (New York Heart Association [NYHA] class III–IV) and reduced left ventricular ejection fraction, evaluating spironolactone versus placebo [12]. EPHESUS included patients with left ventricular systolic dysfunction and heart failure following acute myocardial infarction, comparing eplerenone with placebo [13]. EMPHASIS-HF evaluated patients with mild symptoms of heart failure (NYHA class II) and reduced ejection fraction, also assessing eplerenone against placebo [14]. Details of study characteristics which were included in this meta-analysis are explained in table 2.
Table 2: Characteristics of the chosen randomized controlled trials (RCTs)
| Study ID | First Author (Year) | Study Design | Population | Intervention | Comparator | Intervention dose | Comparator dose | Primary Outcome | Proportion of patients achieving target outcome (Intervention group)* | Proportion of patients achieving target outcome (Comparator group)** | Duration (weeks) |
| RALES | Pitt (1999) [12] | Double-blind RCT | Severe HFrEF (NYHA III–IV, LVEF ≤35%) | Spironolactone | Placebo | 25 mg OD | Matching placebo | Cardiovascular death | 246/822 (0.3) | 338/841 (0.4) | 104 |
| EPHESUS | Pitt (2003) [13] | Double-blind RCT | Post-MI HF (LVEF ≤40%) | Eplerenone | Placebo | 25-50 mg OD | Matching placebo | Cardiovascular death | 407/3319 (0.12) | 482/3313 (0.15) | 72 |
| EMPHASIS-HF | Zannad (2011) [14] | Double-blind RCT | Mild HFrEF (NYHA II, LVEF ≤35%) | Eplerenone | Placebo | Upto 50 mg OD | Matching placebo | Cardiovascular death | 147/1364 (0.11) | 185/1373 (0.13) | 91 |
*Proportion of patients achieving target outcome (cardiovascular death) in intervention group = Sample size of intervention group showing the desired outcome/Total sample size of intervention group. **Proportion of patients achieving target outcome (cardiovascular death) in Comparator group = Sample size of comparator group showing the desired outcome/Total sample size of comparator group.
Cardiovascular mortality outcomes
All three trials demonstrated a reduction in cardiovascular mortality with mineralocorticoid receptor antagonist therapy compared with placebo. In RALES, cardiovascular deaths occurred less frequently in the spironolactone group than in the placebo group, reflecting a substantial relative risk reduction in patients with advanced heart failure [12]. In the EPHESUS trial, eplerenone significantly reduced cardiovascular mortality in patients with heart failure and left ventricular dysfunction following myocardial infarction, with consistent reductions observed across major cardiovascular causes of death, including sudden cardiac death and death due to progressive heart failure [13]. Similarly, EMPHASIS-HF demonstrated a significant reduction in cardiovascular mortality among patients with mild systolic heart failure treated with eplerenone compared with placebo [14].
Pooled effect estimate
When pooled using an inverse-variance fixed-effect model, mineralocorticoid receptor antagonists were associated with a statistically significant reduction in cardiovascular mortality compared with placebo. The pooled risk ratio (pooled RR) was 0.80, corresponding to a 20% relative reduction in mortality, with a 95% confidence interval (CI) ranging from 0.73 to 0.86 (statistically significant) (table 3). The pooled risk ratio, along with narrow 95% CI indicated a consistent and clinically meaningful mortality benefit across trials [12-14].
Table 3: Pooled effect estimate
| Study ID | RR | SE | Lower 95% CI (Individual study) | Upper 95% CI (Individual study) | Pooled RR | SE of pooled Log (RR) | Lower 95% CI (Meta-analysis) | Upper 95% CI (Meta-analysis) |
| RALES | 0.74 | 0.07 | 0.65 | 0.85 | 0.80 | 0.04 | 0.73 | 0.86 |
| EPHESUS | 0.84 | 0.06 | 0.74 | 0.95 | ||||
| EMPHASIS-HF | 0.80 | 0.1 | 0.65 | 0.98 |
RR: Risk Ratio; SE: Standard Error; CI: Confidence Interval.
Forest plot
The forest plot demonstrates a consistent reduction in cardiovascular mortality with MRAs therapy compared with placebo across all included randomized controlled trials. Each study shows a point estimate favouring MRAs, with confidence intervals that largely exclude the line of no effect (RR = 1). The pooled analysis indicates a statistically significant 20% relative reduction in cardiovascular mortality (pooled RR = 0.80, 95% CI = 0.73–0.86). The narrow confidence interval around the pooled estimate reflects the robustness of the finding and the contribution of large, high-quality trials. Visual inspection of the forest plot suggests minimal between-study variability, supporting the appropriateness of a fixed-effect model for the primary analysis (fig. 1).
Risk of bias assessment
Risk of bias assessment was conducted using the Cochrane Risk of Bias tool [15]. All included trials were judged to be at low risk of bias across key methodological domains, including randomization process, allocation concealment, blinding of participants and outcome assessors, completeness of outcome data, and selective reporting. Outcome adjudication committees were blinded to treatment allocation in all trials, further strengthening the internal validity of the findings (table 4).
Heterogeneity assessment
Assessment of between-study heterogeneity using Cochran’s Q statistic and the I² metric demonstrated no significant heterogeneity among the included trials (I² = 0%). This finding indicates a high level of consistency in the direction and magnitude of cardiovascular mortality reduction associated with mineralocorticoid receptor antagonist therapy across diverse heart failure populations.
GRADE assessment
The certainty of evidence for cardiovascular mortality was rated as high. All included studies were well-conducted randomized controlled trials with low risk of bias and consistent effect estimates, showing no meaningful heterogeneity (I² = 0%). The pooled estimate demonstrated a precise and clinically relevant reduction in cardiovascular mortality, with no serious concerns regarding indirectness or imprecision.
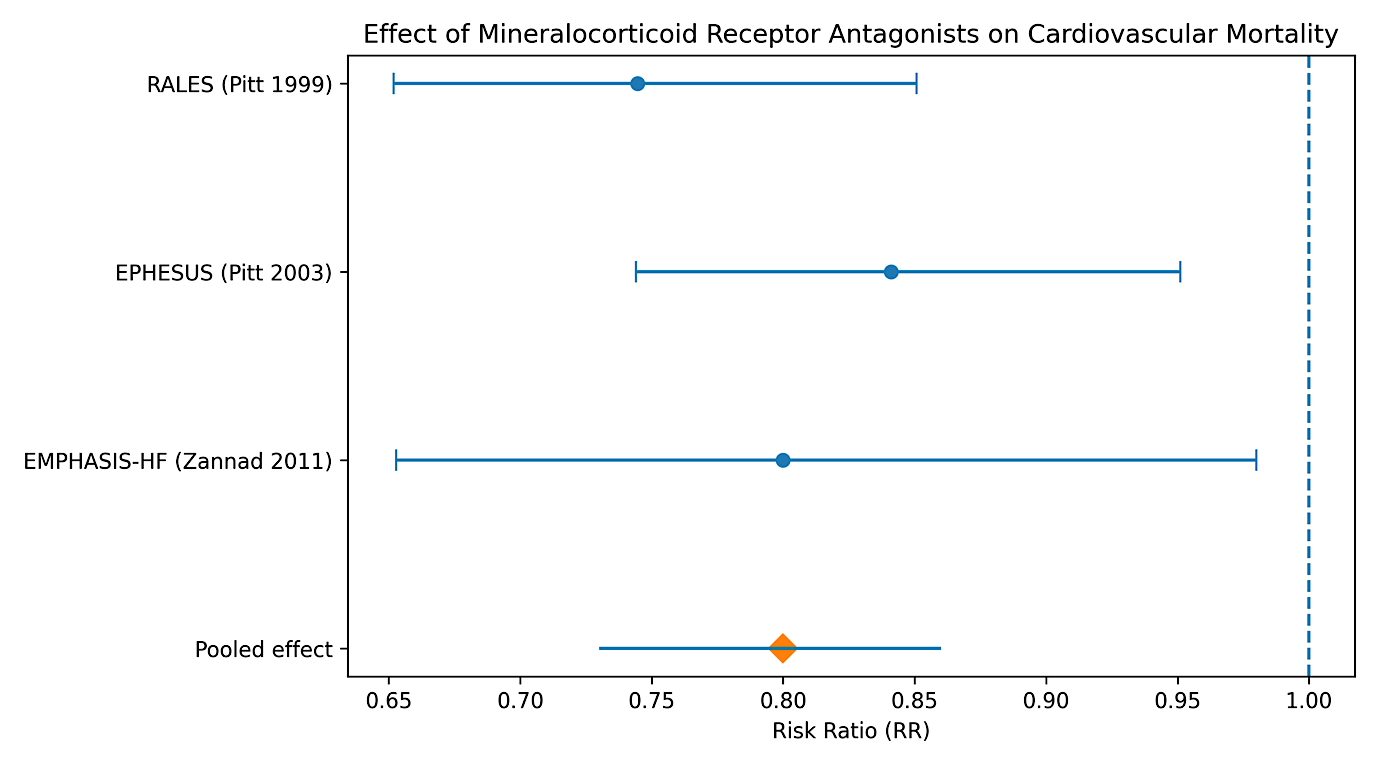
Fig. 1: Forest plot showing the effect of MRAs versus placebo on cardiovascular mortality in patients with heart failure. Individual study estimates are presented as risk ratios with 95% confidence intervals, and the pooled effect is shown as a diamond
Table 4: Risk of bias (RoB) assessment for the included RCTs
| Study ID | Randomization process | Allocation concealment | Blinding of participants and personnel | Blinding of outcome assessment | Incomplete outcome data | Selective reporting | Overall risk of bias |
| RALES | Low | Low | Low | Low | Low | Low | Low |
| EPHESUS | Low | Low | Low | Low | Low | Low | Low |
| EMPHASIS-HF | Low | Low | Low | Low | Low | Low | Low |
Table 5: Heterogeneity assessment of included studies.
| Study ID | ((Log RR-pooled log RR)^2)*weight | Cochran's Q statistic | Degrees of freedom (df) | I2 statistic |
| RALES | 0.966 | 1.74 | 2 | 0% |
| EPHESUS | 0.772 | |||
| EMPHASIS-HF | 0.002 |
Table 6: GRADE assessment for cardiovascular mortality
| Outcome | No. of studies (design) | Risk of bias | Inconsistency | Indirectness | Imprecision | Publication bias | Overall certainty of evidence |
| Cardiovascular mortality | 3 (Randomized controlled trials) | Not serious | Not serious (I² = 0%) | Not serious | Not serious | Unlikely | High |
DISCUSSION
In this meta-analysis of RCTs, mineralocorticoid receptor antagonist therapy was associated with a significant reduction in cardiovascular mortality among patients with heart failure. By pooling data from three large, high-quality trials encompassing more than 11,000 participants, the present analysis provides robust evidence supporting the cardio-protective role of aldosterone blockade across a broad spectrum of heart failure severity. The observed reduction in cardiovascular mortality is clinically meaningful and aligns with the established pathophysiological understanding of aldosterone-mediated myocardial and vascular injury.
The biological plausibility of these findings is well supported. Aldosterone excess contributes to adverse ventricular remodelling, myocardial fibrosis, endothelial dysfunction, and heightened arrhythmic risk, all of which predispose patients with heart failure to cardiovascular death [3, 4]. Mineralocorticoid receptor antagonists mitigate these effects by attenuating fibrosis, reducing inflammation, and improving myocardial electrical stability, thereby lowering the risk of sudden cardiac death and death due to progressive heart failure [16, 17]. These mechanisms are particularly relevant given the phenomenon of aldosterone escape, which may occur despite background therapy with angiotensin-converting enzyme inhibitors or angiotensin receptor blockers [5].
The consistency of cardiovascular mortality reduction across the included trials is a notable strength of this meta-analysis. Despite differences in clinical context ranging from severe chronic heart failure to post–myocardial infarction left ventricular dysfunction and mild symptomatic heart failure, the direction and magnitude of effect were remarkably similar. The absence of statistical heterogeneity (I² = 0%) underscores the reproducibility of benefit with mineralocorticoid receptor antagonists and supports the validity of the pooled estimate. This consistency reinforces current guideline recommendations that position MRAs as a foundational component of guideline-directed medical therapy in heart failure with reduced ejection fraction [7].
Our findings are concordant with prior narrative reviews and secondary analyses that have highlighted the survival benefits of aldosterone antagonism in heart failure populations [6, 18]. However, unlike individual trials or descriptive reviews, the present meta-analysis provides a precise quantitative estimate focused specifically on cardiovascular mortality, a hard clinical endpoint with direct relevance to patients, clinicians, and policymakers. By restricting inclusion to randomized controlled trials and cardiovascular death outcomes, this analysis minimizes confounding and enhances the reliability of the conclusions.
Several strengths merit emphasis. First, only large, double-blind randomized controlled trials with rigorous outcome adjudication were included, resulting in a high overall certainty of evidence. Second, cardiovascular mortality was consistently defined across studies to include cardiac and vascular causes of death, improving endpoint uniformity. Third, the low risk of bias and lack of heterogeneity strengthen confidence in the pooled results. Collectively, these features support the internal validity and clinical applicability of the findings.
Nevertheless, certain limitations should be acknowledged. The meta-analysis included only three trials, reflecting the limited number of large, randomized studies reporting cardiovascular mortality with mineralocorticoid receptor antagonists. Although the trials were substantial in size, the small number of studies precluded formal assessment of publication bias. Additionally, differences trial era and heart failure phenotype may limit extrapolation to contemporary populations receiving newer disease-modifying agents. Finally, patient-level data were not available, preventing exploration of subgroup effects based on age, sex, renal function, or comorbidities. An additional limitation of this meta-analysis is the use of Microsoft Excel for statistical analyses. Although Excel allows transparent calculation of effect estimates and heterogeneity measures when formulas are correctly applied, it lacks the advanced automation, error-checking features, and graphical capabilities available in dedicated meta-analysis software. However, given the small number of included trials, low heterogeneity, and manual cross-verification of calculations, this limitation is unlikely to have materially influenced the results.
From a clinical perspective, these results reinforce the importance of MRAs in reducing cardiovascular mortality among patients with heart failure. Despite strong guideline endorsement, MRAs remain underutilized in routine practice, often due to concerns regarding hyperkalaemia or renal dysfunction [19]. The present findings highlight that, when appropriately prescribed and monitored, MRAs confer substantial survival benefits that should not be overlooked. Improving adherence to guideline-recommended MRA therapy may therefore represent a critical opportunity to reduce cardiovascular mortality in real-world heart failure populations.
Future research should focus on optimizing the safe implementation of mineralocorticoid receptor antagonists in diverse patient groups, including those with chronic kidney disease or borderline hyperkalaemia. In addition, comparative and network meta-analyses evaluating MRAs alongside other contemporary disease-modifying therapies may further clarify their relative and additive benefits. Individual patient data meta-analyses could also provide valuable insights into patient subgroups most likely to derive maximal benefit from aldosterone blockade.
CONCLUSION
This meta-analysis demonstrates that MRAs significantly reduce cardiovascular mortality in patients with heart failure. The observed benefit was consistent across large, well-conducted randomized controlled trials encompassing diverse heart failure populations and clinical settings, with minimal heterogeneity. These findings provide high-certainty evidence supporting the continued and expanded use of mineralocorticoid receptor antagonists as a core component of guideline-directed medical therapy. Optimizing their implementation in routine clinical practice has the potential to substantially improve cardiovascular outcomes in patients with heart failure.
ACKNOWLEDGEMENT
The authors would like to acknowledge all investigators and participants of the original randomized controlled trials whose contributions made this meta-analysis possible. No external editorial or writing assistance was used in the preparation of this manuscript.
FUNDING
This study did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
AUTHORS CONTRIBUTIONS
Pranab Das and Mahaprasad Pal conceived and planned the study, conducted the literature search, performed data extraction and analysis, and prepared the initial draft of the manuscript. Wasim Akram and Bodhisatya Das contributed to data validation, interpretation of findings, and critical revision of the manuscript. All authors reviewed and approved the final manuscript and take full responsibility for the accuracy and integrity of the work.
CONFLICT OF INTERESTS
The authors declare that there are no conflicts of interest related to this work.
REFERENCES
Savarese G, Lund LH. Global public health burden of heart failure. Card Fail Rev. 2017;3(1):7-11. doi: 10.15420/cfr.2016:25:2, PMID 28785469.
Roger VL. Epidemiology of heart failure: a contemporary perspective. Circ Res. 2021;128(10):1421-34. doi: 10.1161/CIRCRESAHA.121.318172, PMID 33983838.
Weber KT. Aldosterone in congestive heart failure. N Engl J Med. 2001;345(23):1689-97. doi: 10.1056/NEJMra000050, PMID 11759649.
Pitt B. Aldosterone blockade in patients with chronic heart failure. Cardiol Clin. 2008;26(1):15-21, doi: 10.1016/j.ccl.2007.12.016, PMID 18312902.
Struthers AD. Aldosterone escape during ACE inhibitor therapy in chronic heart failure. Eur Heart J. 1995;16(Suppl N):103-6. doi: 10.1093/eurheartj/16.suppl_N.103, PMID 8682054.
Zannad F, Gattis Stough W, Rossignol P, Bauersachs J, McMurray JJ, Swedberg K. Mineralocorticoid receptor antagonists for heart failure with reduced ejection fraction: integrating evidence into clinical practice. Eur Heart J. 2012;33(22):2782-95. doi: 10.1093/eurheartj/ehs257, PMID 22942339.
McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A. 2021 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure: developed by the task force for the diagnosis and treatment of acute and chronic heart failure of the European society of cardiology (ESC) with the special contribution of the heart failure association (HFA) of the ESC. Eur J Heart Fail. 2022;24(1):4-131. doi: 10.1002/ejhf.2333, PMID 35083827.
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ. Cochrane handbook for systematic reviews of interventions. Version 6.4. London (UK): Cochrane; 2023. Available from: https://training.cochrane.org/handbook.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. doi: 10.1136/bmj.n71, PMID 33782057.
Sterne JA, Sutton AJ, Ioannidis JP, Terrin N, Jones DR, Lau J. Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomised controlled trials. BMJ. 2011;343:d4002. doi: 10.1136/bmj.d4002, PMID 21784880.
Das P. Meta-analysis in excel: a beginner’s guide: simplifying statistics for medical students and researchers. 1st ed. Chhattisgarh: Shashwat Publication; 2024.
Pitt B, Zannad F, Remme WJ, Cody R, Castaigne A, Perez A. The effect of spironolactone on morbidity and mortality in patients with severe heart failure randomized aldactone evaluation study investigators. N Engl J Med. 1999;341(10):709-17. doi: 10.1056/NEJM199909023411001, PMID 10471456.
Pitt B, Remme W, Zannad F, Neaton J, Martinez F, Roniker B. Eplerenone a selective aldosterone blocker in patients with left ventricular dysfunction after myocardial infarction. N Engl J Med. 2003;348(14):1309-21. doi: 10.1056/NEJMoa030207, PMID 12668699.
Zannad F, McMurray JJ, Krum H, Van Veldhuisen DJ, Swedberg K, Shi H. Eplerenone in patients with systolic heart failure and mild symptoms. N Engl J Med. 2011;364(1):11-21. doi: 10.1056/NEJMoa1009492, PMID 21073363.
Sterne JA, Higgins JP, Elbers RG, Reeves BC. Risk of bias 2: a tool to assess risk of bias in randomised trials. Version 22. In: Cochrane; 2019. Available from: https://www.riskofbias.info/welcome/rob-2-0-tool/current-version-of-rob-2.
Brilla CG, Weber KT. Mineralocorticoid excess dietary sodium and myocardial fibrosis. J Lab Clin Med. 1992;120(6):893-901. PMID 1453111.
Pitt B, Pedro Ferreira J, Zannad F. Mineralocorticoid receptor antagonists in patients with heart failure: current experience and future perspectives. Eur Heart J Cardiovasc Pharmacother. 2017;3(1):48-57. doi: 10.1093/ehjcvp/pvw016, PMID 27530337.
McMurray JJ, Packer M. How should we sequence the treatments for heart failure and a reduced ejection fraction? a redefinition of evidence-based medicine. Circulation. 2021;143(9):875-7. doi: 10.1161/circulationaha.120.052926, PMID 33378214.
Matsumoto S, Kondo T, Jhund PS, Campbell RT, Swedberg K, Van Veldhuisen DJ. Underutilization of mineralocorticoid antagonists in patients with heart failure with reduced ejection fraction. J Am Coll Cardiol. 2023;82(11):1080-91. doi: 10.1016/j.jacc.2023.06.021, PMID 37642608.