
Int J Curr Pharm Res, Vol 18, Issue 2, 65-70Original Article
KNOWLEDGE, ATTITUDE, AND PERCEPTION OF MEDICAL INTERNS TOWARDS ANTIMICROBIAL STEWARDSHIP IN A TERTIARY CARE HOSPITAL
MANSOOR BASHA P.*, PADMA SRAVANI S., SHARON SONIA S.
Department of Pharmacology, Government Medical College, Ananthapuramu, Andhra Pradesh, India
*Corresponding author: Mansoor Basha P.; *Email: mansoorbasha398@gmail.com
Received: 10 Nov 2025, Revised and Accepted: 03 Jan 2026
ABSTRACT
Objective: To assess the knowledge, attitude, and perception of medical interns regarding antimicrobial stewardship programme in Government Medical College, Anathapuramu.
Methods: A Cross-sectional, questionnaire-based study (KAP) was conducted in Medical Interns of 150 members in Government Medical College, Ananthapuramu. Only 128 medical interns participated in the study. The study was carried out between September 2025 and October 2025 among medical interns. Data entered and analysed using Statistical Package for Social Sciences (SPSS) software version 21.0 for Windows.
Results: The total 128 interns are participated in a study in which males are about 42.2% and females are 57.8% Overall, interns showed good awareness and positive attitudes toward antimicrobial stewardship but suboptimal practices. This discrepancy underscores the need for structured training and real-world mentorship to translate knowledge into consistent stewardship behaviour.
Conclusion: The current study demonstrates the dire need of medical interns for further education about antimicrobial stewardship. Hence, it is strongly recommended to provide a comprehensive, regular, standard and up-to-date educational training in all medical institutions, recommended to advocate curriculums and policies that build up antimicrobial stewardship programs. There is a critical need for structured education, training, and supervised clinical exposure to strengthen antimicrobial stewardship practices among medical interns.
Keywords: Antimicrobial stewardship (AMS) Programs, Knowledge, Attitude, Perception (KAP), Healthcare professionals
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i2.8066 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Antimicrobial agents are used to fight against infectious diseases and have saved millions of lives worldwide. The ability of microorganisms to withstand previously effective antimicrobial agents is defined as antimicrobial resistance (AMR) [1]. AMR is a critical global issue [2] and causes over 23,000 deaths annually [3, 4]. Some studies project that this mortality rate could reach over 10 million by 2050 [5, 6]. Although AMR develops naturally, it is accelerated by the misuse of antimicrobials in healthcare, agriculture, and aquaculture [7-9]. Inappropriate use of antibiotics and widespread AMR raise concerns that antimicrobials may lose their sensitivity to a range of microorganisms. Antimicrobial stewardship programs (ASPs) aim to reduce AMR. Typically, a multidisciplinary team within a healthcare institution directs an ASP to ensure patient safety and promote optimum antimicrobial treatment by selecting the proper antibiotics and their dosage and length of treatment. The ASP team may consist of a variety of healthcare professionals (HCPs), including physicians, pharmacists, infection control specialists, microbiologists, nurses, allied health professionals, insurers, researchers, and government representatives.
Antimicrobials are the most commonly prescribed drugs in developing as well as many developed countries to treat and control infectious disease [1, 2]. Inappropriate use of antibiotics results in bacteria resistant to antibiotics in the community and hospital. Antibiotic resistance is a worldwide problem, with a negative consequence on patient treatment outcomes [4]. Furthermore, patients infected with drug-resistant bacterial infection need more expensive intervention, so antimicrobial resistance results in increased health care costs to families and societies [5]. Reducing the burden of antimicrobial resistance is highly challenging in low-income countries than developed countries due to high prevalence of infection, irrational uses of antimicrobials, over-the-counter availability of antibiotics and lack of clinical microbiology laboratories for antimicrobial susceptibility testing in the developing countries [6].
In most countries, healthcare system physicians are responsible for prescribing antibiotics. However, junior medical doctors feel unprepared for the complexity of antibiotic prescribing in their daily practice [7]. Several international studies have identified areas of lack of confidence and gaps in antibiotic prescribing knowledge, and have shown that medical students perceive a need for further education and training on antibiotic prescribing and antimicrobial stewardship program [8]. Education can influence prescribing behaviour and can provide knowledge that will enhance the acceptance of stewardship strategies among health care practitioners [9]. In most countries education systems, there are no enough antimicrobial resistance education programs at undergraduate level and are still being conducted at the postgraduate level in healthcare institutions [10]. Evidence suggests that a multifaceted approach is favoured at improving the organization of the healthcare system and changing physicians’ prescribing behaviour and practice [11]. Reduction of antimicrobial resistance requires change in antimicrobial prescribing behaviour of health workers, antimicrobial prescribing patterns and physicians’ behaviour towards the magnitude of antimicrobial resistance problem [12]. The World Health Organization has recommended training for medical undergraduates students regarding appropriate prescription of antimicrobials [13]. It is necessary that our future doctors are better equipped with better knowledge, attitude towards antimicrobial use and resistance [14]. Unlike the senior physicians or infectious specialists who have a large amount of experience and knowledge in infection treatment, the junior doctors usually have limited knowledge and skills to reduce the potential risk of antimicrobial resistance [15]. Therefore, antimicrobial stewardship efforts should be made to standardize the prescribing behaviours of medical interns [16]. In India, limited studies have assessed the knowledge, attitude, and perception of medical interns regarding antimicrobial stewardship, highlighting the need for further research [17, 18]. Hence, the purpose of this study is to assess the attitude and perception of medical interns on antimicrobial resistance at teaching hospitals in Ananthapuramu. The information generated in this study would be instrumental in planning and implementing preventive and control interventions on antimicrobial resistance and applying antimicrobial stewardship program at regional and national levels. Therefore, this study was undertaken to assess the knowledge, attitude, and perception of medical interns toward antimicrobial stewardship in a tertiary care teaching hospital.
MATERIALS AND METHODS
A observational, cross-sectional questionnaire based study was conducted to assess the knowledge, attitude, and perception (KAP) of healthcare professionals towards Antimicrobial Stewardship Programs (ASP). The study was carried out between September 2025 and October 2025 amongthemedical interns of total 150 membersin Government Medical College, Ananthapuramu. Out of 150 interns, only 128 are given consent form for willing to participate in the study. The questionnaire and the consent forms were sent online to the participants via google forms. Ethical clearance was taken from the Institutional Ethics Committee prior to beginning of the study.
Out of 150 medical interns, the sample size of 128 participants were included in the study. The structured and self-administered questionnaire was developed by comparing various studies and the questions adapted from WHO guidelines on AMR. Total of 25 MCQs included in the study. Knowledge based questionnaire (10 MCQ s) which include knowledge in Antimicrobial stewardship programme and Anti-microbial Drugs. Perception (10 MCQs) and Attitude (5 MCQs) are given through google forms to interns. Evaluation done based on a 5-point Likert scale
The responses were exported from Google Forms to an Excel spreadsheet, cleaned and decoded, and then analyzed using IBM SPSS Statistics Version 24.0 for Windows. Descriptive statistics were used to analyze the demographic data and survey responses.
RESULTS
Participants socio-demographic and other characteristics
Table 1 provides an overview of the demographics of the participants who participated in the study. The study involved among 128 participants (out of 150) ranging from various specialities who completed the questionnaire, yielding a response rate of 85.3%. The participants were medical interns as per their designation, with medical sub-specialties, surgical and surgical sub-specialties, and critical medicine departments as their primary specialties.
Table 1: Distribution of medical interns by specialty
| Characteristics | Frequency (n=128) | Percentage (%) |
| Primary Speciality | ||
| Medicine and Medical sub-specialties | 67 | 52.3% |
| Surgery and Surgical sub-specialties | 43 | 33.5% |
| Critical care medicine | 18 | 14% |
| Designation | ||
| Interns | 128 | 100% |
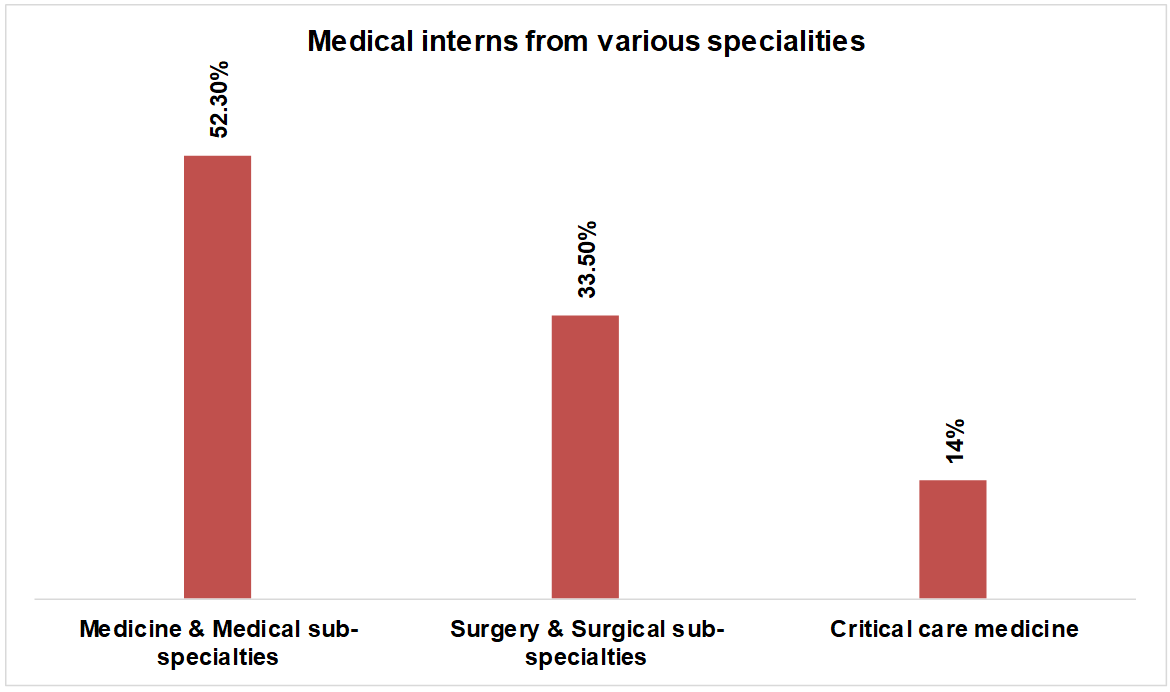
Fig. 1: Fig. showing medical interns from various specialities, the majority of participants were within the age group of 23-24 y (n = 92, 71.9%), followed by 21-22 y (n = 22, 17.2%) depicting in the following table 2 and fig. 2
Table 2: Age (in years) of the participants
| Age (in years) | Frequency (n) | Percentage (%) |
| <20 yrs | 2 | 1.6% |
| 21-22 yrs | 22 | 17.2% |
| 23-24 yrs | 92 | 71.9% |
| >25 yrs | 12 | 9.4% |
| Total | 128 | 100% |
Table 3: table representing the gender distribution among the participants
| Gender | Frequency (n) | Percentage (%) |
| Male | 54 | 42% |
| Female | 74 | 58% |
| Total | 128 | 100% |
The table 3 and fig. 3 represents the gender distribution among the medical interns (n=128) where males (n=54) comprises of 42% and females (n=74) about 58% involved in the study.
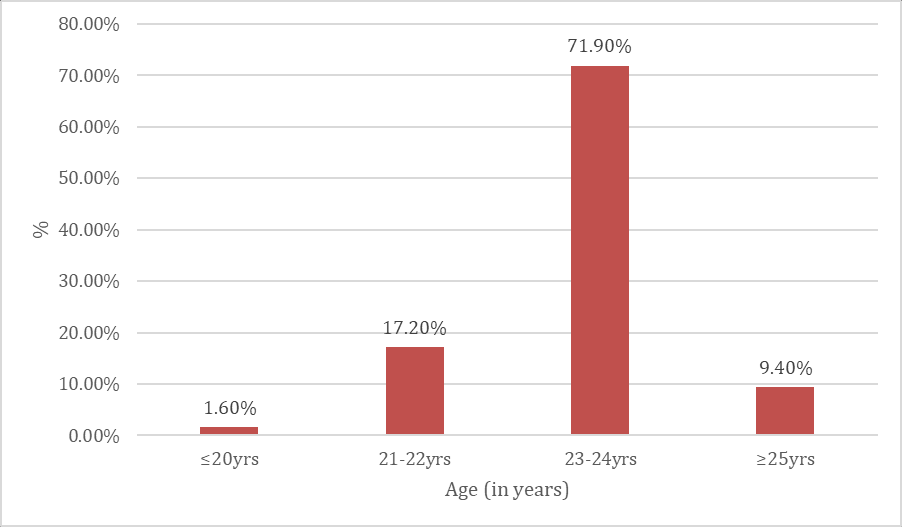
Fig. 2: Fig. depicted in the Age (in years) of participants
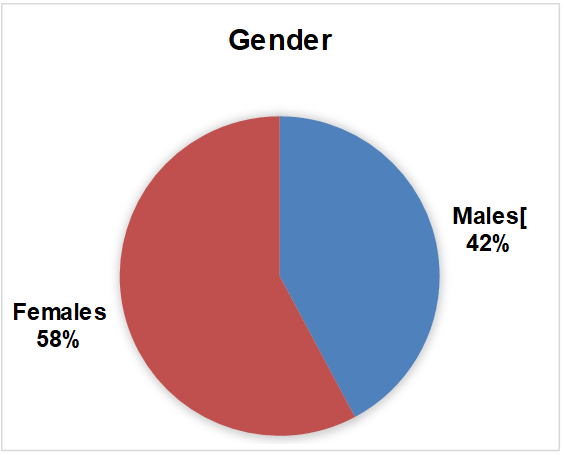
Fig. 3: Fig. representing the gender distribution among the participants
Table 4: Knowledge of health care professionals towards antimicrobial stewardship
| Knowledge of health care professionals towards antimicrobial stewardship | % Correct answer | % Wrong answer |
|
81.3 | 18.7 |
|
65.6 | 34.4 |
|
59.4 | 40.6 |
|
89.1 | 10.9 |
|
48.4 | 51.6 |
| 6. Which of the following is a broad-spectrum antibiotic? | 54.7 | 45.3 |
|
60.9 | 39.1 |
| 8. What does the term "antibiotic de-escalation" mean? | 73.4 | 26.6 |
| 9. Which of the following is NOT a benefit of antimicrobial stewardship? | 60.9 | 39.1 |
| 10. What is the best initial step in diagnosing the cause of fever in a hospitalized patient? | 76.6 | 23.4 |
The knowledge assessment revealed variability in understanding across different domains of antimicrobial stewardship. Correct response rates ranged from 48.4% to 89.1%, demonstrating areas of strong knowledge as well as significant gaps.
Participants showed the highest knowledge in identifying antibiotic misuse (89.1%) and understanding the primary goal of AMS (81.3%). Knowledge related to proper diagnostic approaches for fever in hospitalized patients was also relatively high (76.6%). Additionally, 73.4% correctly understood the concept of antibiotic de-escalation.
Moderate performance was observed in questions regarding cornerstone elements of stewardship (65.6%), the recommended duration of therapy for uncomplicated urinary tract infections (60.9%), and the benefits of AMS (60.9%). Understanding of the “5 Rs” of stewardship (59.4%) and recognition of broad-spectrum antibiotics (54.7%) also reflected only partial knowledge.
The lowest-scoring question concerned the most common mechanism of antibiotic resistance, with only 48.4% answering correctly, indicating a major knowledge gap in microbiological fundamentals.
Overall, the results indicate that while participants possess foundational knowledge, several essential areas of AMS require targeted educational reinforcement.
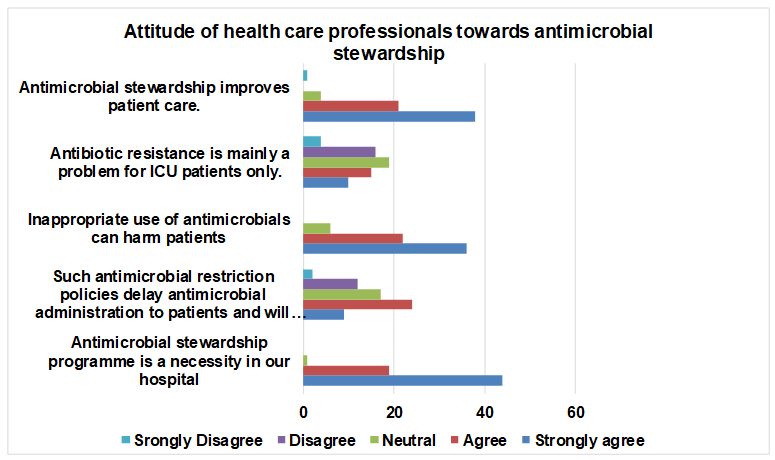
Fig. 4: Representing attitude of medical interns towards antimicrobial stewardship
The attitude of interns toward antimicrobial stewardship was assessed using a 5-point Likert scale ranging from strongly disagree (1) to strongly agree (5). Overall, interns demonstrated a predominantly positive attitude toward antimicrobial stewardship and its role in improving patient care and hospital practice.
A substantial majority of interns agreed or strongly agreed that antimicrobial stewardship improves patient care. Based on visual estimation from the response distribution, approximately 65–70% of interns expressed agreement, yielding an estimated mean Likert score of 3.8±0.9, indicating strong positive attitudes.
Regarding the statement that antibiotic resistance is mainly a problem confined to ICU patients, responses were more heterogeneous. Nearly 40–45% of interns disagreed or strongly disagreed, while around 30% expressed agreement and the remainder were neutral. The estimated mean score of 2.9±1.1 suggests partial misconceptions and uncertainty among interns regarding the broader scope of antimicrobial resistance.
A high level of awareness was observed for the statement that inappropriate use of antimicrobials can harm patients. Approximately 70–75% of interns selected agree or strongly agree, corresponding to an estimated mean score of 4.0±0.8, reflecting strong recognition of patient safety concerns.
Perceptions regarding antimicrobial restriction policies were comparatively mixed. About 45–50% of interns agreed that such policies may delay antimicrobial administration, while a considerable proportion remained neutral. The estimated mean score of 3.2±1.0 reflects ambivalence toward the operational impact of stewardship policies.
Notably, the statement that an antimicrobial stewardship programme is a necessity in the hospital received the strongest endorsement. Approximately 75–80% of interns strongly agreed, resulting in an estimated mean likert score of 4.4±0.7, highlighting strong institutional acceptance among interns.
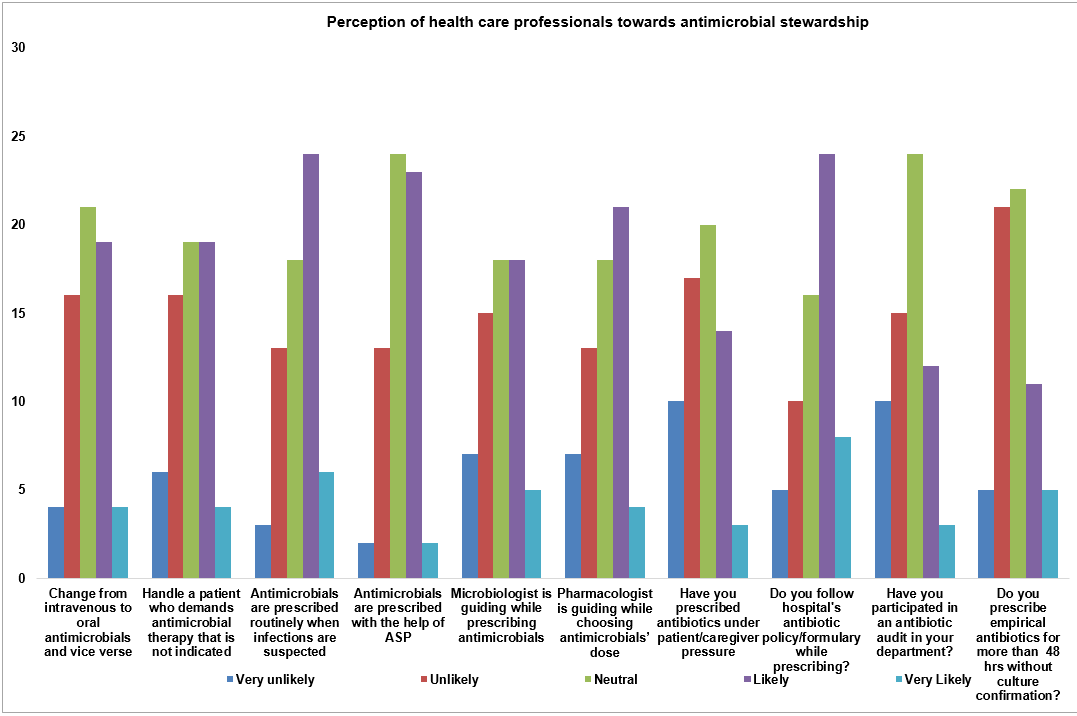
Fig. 5: Representing perception of medical interns towards antimicrobial stewardship
Perception of interns toward antimicrobial stewardship–related practices was assessed using a 5-point Likert scale ranging from very unlikely (1) to very likely (5). Overall, interns demonstrated a moderately positive perception toward key stewardship practices, guideline use, and multidisciplinary support, although neutral responses were common for several items.
Approximately 55–60% of interns reported being likely or very likely to change from intravenous to oral antimicrobials when clinically appropriate, with an estimated mean Likert score of 3.4±1.1, indicating moderate acceptance of de-escalation strategies.
Regarding confidence in handling patients requiring antimicrobial therapy, around 50–55% of interns selected likely or very likely, yielding an estimated mean likert score of 3.3±1.0. Similarly, perceptions that antimicrobials are used appropriately in clinical practice showed a modestly positive trend, with approximately 55% expressing favorable responses (mean ≈ 3.5±1.0).
Consultation with microbiologists for antimicrobial guidance was perceived as likely or very likely by about 45–50% of interns (mean ≈ 3.2±1.1). Slightly higher endorsement was observed for seeking pharmacologist guidance, with approximately 50–55% indicating favorable perceptions (mean ≈ 3.4±1.0).
Adherence to hospital antimicrobial guidelines demonstrated relatively stronger perception, with nearly 60% of interns reporting they were likely or very likely to follow institutional protocols (mean ≈ 3.6±0.9). Participation in antimicrobial stewardship activities was reported by approximately 45–50% of interns (mean ≈ 3.3±1.1).
Empirical prescribing practices were commonly perceived, with about 50–55% of interns indicating they were likely to prescribe antimicrobials empirically (mean ≈ 3.4±1.0), while a substantial proportion remained neutral.
DISCUSSION
This KAP study demonstrates that healthcare professionals possess good foundational knowledge, positive attitudes, and moderately favorable perceptions toward antimicrobial stewardship. The high level of correct responses regarding stewardship goals and antibiotic misuse suggests that core concepts are well understood. However, gaps in knowledge related to mechanisms of resistance and spectrum of antibiotics highlight areas requiring targeted educational reinforcement. Similar findings have been reported in previous studies assessing knowledge and attitudes toward antimicrobial stewardship among junior doctors and healthcare professionals [8, 11, 14].
The overwhelmingly positive attitude toward antimicrobial stewardship—particularly its perceived necessity and role in improving patient care—is encouraging and aligns with global efforts to integrate stewardship into routine hospital practice. International guidelines and systematic reviews have consistently demonstrated that effective antimicrobial stewardship programs improve clinical outcomes and reduce antimicrobial resistance [14-16]. Recognition of patient harm resulting from inappropriate antimicrobial use further reinforces readiness for stewardship implementation.
Nevertheless, misconceptions persist, particularly regarding antibiotic resistance being confined to ICU settings. Such beliefs may limit stewardship engagement in general wards and outpatient settings, underscoring the need for education emphasizing the hospital-wide and community-wide nature of antimicrobial resistance. Studies have shown that antimicrobial resistance is prevalent across all hospital wards and is not limited to intensive care units, highlighting the importance of institution-wide stewardship interventions [2, 15].
Perception findings suggest that while respondents are receptive to stewardship practices such as guideline adherence and IV-to-oral switch strategies, operational concerns remain. Neutral responses regarding restriction policies and moderate reliance on multidisciplinary consultation indicate perceived barriers related to workflow, accessibility, and clinical autonomy. Operational barriers, diagnostic uncertainty, and limited microbiology support have been identified as major factors contributing to continued empirical antibiotic prescribing in hospital settings [7, 10, 17].
The continued reliance on empirical prescribing reflects diagnostic uncertainty and time constraints inherent in clinical practice. Strengthening access to rapid diagnostics, reinforcing de-escalation principles, and integrating stewardship teams into daily clinical workflows may help bridge the gap between positive attitudes and consistent practice.
Overall, the findings indicate a favourable environment for strengthening antimicrobial stewardship programs, particularly through targeted education addressing identified knowledge gaps, structured intern and clinician training, and enhanced multidisciplinary collaboration. Evidence suggests that targeted educational interventions and hands-on stewardship training during internship significantly improve antimicrobial prescribing behaviour and guideline adherence [13, 16, 18].
This study evaluated the KAP of medical interns regarding antimicrobial stewardship, revealing moderate knowledge, strong attitudes, but inconsistent practice. The finding that 68% recognized inappropriate antibiotic use as a driver of resistance suggests basic conceptual understanding. However, the limited awareness (40%) of WHO stewardship elements highlights a knowledge gap regarding structured ASP frameworks.
Positive attitudes toward ASPs were noted, consistent with global findings emphasizing young clinicians’ awareness of AMR threats. Nevertheless, only 55% expressed confidence in applying stewardship guidelines, suggesting that theoretical understanding does not always translate to practical capability. Practice-related deficiencies were evident, especially in antibiotic review at 48 h—a core stewardship step shown to reduce unnecessary antibiotic exposure. The disparity between knowing and doing may be attributed to time constraints, inadequate supervision, or lack of structured stewardship exposure during internship.
These results reinforce the need for strengthened, competency-based stewardship training, integration of interns into ASP teams, and mandatory hands-on sessions during clinical rotations.
CONCLUSION
Medical interns demonstrated adequate knowledge and positive attitudes toward antimicrobial stewardship; however, practical implementation was suboptimal. Structured educational interventions, enhanced clinical supervision, and active involvement in antimicrobial stewardship programs are essential to improve rational antibiotic use and reduce antimicrobial resistance.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
All authors have contributed equally
CONFLICT OF INTERESTS
Declared none
REFERENCES
Antimicrobial Resistance Collaborators. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet. 2022;399(10325):629-55. doi: 10.1016/S0140-6736(21)02724-0, PMID 35065702.
Cassini A, Hogberg LD, Plachouras D, Quattrocchi A, Hoxha A, Simonsen GS. Attributable deaths and disability-adjusted life-years caused by infections with antibiotic-resistant bacteria in the EU and the European economic area in 2015: a population-level modelling analysis. Lancet Infect Dis. 2019;19(1):56-66. doi: 10.1016/S1473-3099(18)30605-4, PMID 30409683.
Vincent JL, Sakr Y, Singer M, Martin Loeches I, Machado FR, Marshall JC. Prevalence and outcomes of infection among patients in intensive care units in 2017. JAMA. 2020;323(15):1478-87. doi: 10.1001/jama.2020.2717, PMID 32207816.
Magill SS, O’Leary E, Ray SM, Kainer MA, Evans C, Bamberg WM. Assessment of the appropriateness of antimicrobial use in U.S. hospitals. JAMA Netw Open. 2021;4(3):e212007. doi: 10.1001/jamanetworkopen.2021.2007, PMID 33734417.
Roger PM, Montero E, Lesselingue D, Perez P, Jouve E, Fosse JP. Risk factors for unnecessary antibiotic therapy in hospitalized patients: a major role for clinical management. Clin Infect Dis. 2019;69(3):466–72. doi: 10.1093/cid/ciy921.
Prescott HC, Iwashyna TJ. Improving sepsis treatment by embracing diagnostic uncertainty. Ann Am Thorac Soc. 2019;16(4):426-9. doi: 10.1513/AnnalsATS.201809-646PS, PMID 30883190.
Trivedi KK, Bartash R, Letourneau AR, Abbo L, Fleisher J, Gagliardo C. Opportunities to improve antibiotic appropriateness in U.S. ICUs: a multicenter evaluation. Crit Care Med. 2020;48(7):968-76. doi: 10.1097/CCM.0000000000004344, PMID 32317600.
Barlam TF, Cosgrove SE, Abbo LM, MacDougall C, Schuetz AN, Septimus EJ. Implementing an antibiotic stewardship program: guidelines by the infectious diseases society of America and the society for healthcare epidemiology of America. Clin Infect Dis. 2016;62(10):e51-77. doi: 10.1093/cid/ciw118, PMID 27080992.
Baur D, Gladstone BP, Burkert F, Carrara E, Foschi F, Dobele S. Effect of antibiotic stewardship on the incidence of infection and colonisation with antibiotic-resistant bacteria and clostridium difficile infection: a systematic review and meta-analysis. Lancet Infect Dis. 2017;17(9):990-1001. doi: 10.1016/S1473-3099(17)30325-0, PMID 28629876.
Davey P, Marwick CA, Scott CL, Charani E, McNeil K, Brown E. Interventions to improve antibiotic prescribing practices for hospital inpatients. Cochrane Database Syst Rev. 2017;2(2):CD003311. doi: 10.1002/14651858.CD003543.pub4, PMID 28178770.
Ashiru Oredope D, Hopkins S, Vasandani S, Umoh E, Oloyede O, Nilsson A. Healthcare workers’ knowledge attitudes and behaviours with respect to antibiotics antibiotic use and antibiotic resistance across 30 EU/EEA countries in 2019. Euro Surveill. 2021;26(12):1900633. doi: 10.2807/1560-7917.ES.2021.26.12.1900633, PMID 33769250.
Rawson TM, Wilson RC, O’Hare D, Herrero P, Kambugu A, Lamorde M. Optimizing antimicrobial use: challenges advances and opportunities. Nat Rev Microbiol. 2021;19(12):747-58. doi: 10.1038/s41579-021-00578-9, PMID 34158654.
World Health Organization. Global action plan on antimicrobial resistance. Geneva: World Health Organization; 2015.
Dyar OJ, Huttner B, Schouten J, Pulcini C, ESGAP. What is antimicrobial stewardship? Clin Microbiol Infect. 2017;23(11):793-8. doi: 10.1016/j.cmi.2017.08.026, PMID 28882725.
Charani E, Castro Sanchez E, Holmes A. The role of behavior change in antimicrobial stewardship. Infect Dis Clin North Am. 2014;28(2):169-75. doi: 10.1016/j.idc.2014.01.004, PMID 24857386.
Tamma PD, Avdic E, Li DX, Dzintars K, Cosgrove SE. Association of adverse events with antibiotic use in hospitalized patients. JAMA Intern Med. 2017;177(9):1308-15. doi: 10.1001/jamainternmed.2017.1938, PMID 28604925.
Labi AK, Obeng Nkrumah N, Nartey ET, Bjerrum S, Adu-Aryee NA, Ofori-Adjei YA. Antibiotic use in a tertiary healthcare facility in Ghana: a point prevalence survey. Antimicrob Resist Infect Control. 2018;7:15. doi: 10.1186/s13756-018-0299-z, PMID 29423190.
World Health Organization. Global antimicrobial resistance surveillance system (GLASS) report: early implementation 2020. Geneva: World Health Organization; 2020. Available from: https://iris.who.int/handle/10665/332081.