
Int J Pharm Pharm Sci, Vol 17, Issue 4, 12-17Original Article Article
A RETROSPECTIVE ANALYSIS OF DRUG UTILIZATION STUDY IN DRUG-RESISTANT TUBERCULOSIS AT TERTIARY CARE HOSPITAL, GUJARAT
VIJITA SHAHa, CHETNA PATELb, JIGAR CHOTALIYAc, BHARGAVI PATELd*
aDepartment of Pharmacology, Dr Kiran C Patel Medical College and Research Institute, Bharuch, Gujarat, India. bDepartment of Pharmacology, Government Medical College, Surat, Gujarat, India. cDepartment of Pharmacology, Pacific Medical College and Hospital, Udaipur, Rajasthan, India. dDepartment of Pharmacology, Dr M K Shah Medical College and Research Centre, Ahmedabad, Gujarat, India
*Corresponding author: Bhargavi Patel; *Email: [email protected]
Received: 24 Dec 2024, Revised and Accepted: 02 Mar 2025
ABSTRACT
Objective: The point of this observational study was to look at how inpatients at our tertiary care facility used medications. The goal was to find ways to improve prescription practices for people with drug-resistant tuberculosis.
Methods: The respiratory ward medical record department serves as the retrospective source of data. We enrolled a total of 301 patients with drug-resistant tuberculosis. We assess drug utilization data using the World Health Organization Anatomical Therapeutic Chemical/Defined Daily Dose approach. We analyzed prescriptions using World Health Organization core drug indicators.
Results: Adherence to international standards was demonstrated by the prescription of most antibiotics at dosages consistent with the recommended daily dose. Antibiotic polypharmacy (100%) was common with Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) (64.1%), proton pump inhibitors (61.5%), vitamins and minerals (77.7%), and generic names (92%) that were frequently provided with oral medications (77%). The National List of Essential Medicines 2022 contained information on approximately 95% of all prescribed drugs.
Conclusion: Drug costs were significantly impacted by the fact that antibiotics were the most often-given drugs, accompanied by vitamins and minerals, NSAIDs, and proton pump inhibitors. Limiting the use of more costly, recent antibiotics, branded medications, and the number of medications given can drastically lower the cost of drug therapy.
Keywords: Tuberculosis, Drug utilization, Defined daily dose, Essential medicines
© 2025 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijpps.2025v17i4.53513 Journal homepage: https://innovareacademics.in/journals/index.php/ijpps
INTRODUCTION
"The marketing, distribution, prescription, and use of drugs in a society, with special emphasis on the resulting medical, social, and economic consequences," is how the World Health Organization described drug use in 1977 [1]. Despite the declining TB epidemic, India continues to have the highest TB burden in the world in terms of the sheer number of incidences. Healthcare failure has led to the resurgence of tuberculosis and the development of antibiotic resistance. To prolong the usefulness of effective antibiotics and slow the establishment of resistance, the prudent use of antibiotics in healthcare is crucial [2]. Since antibiotics are a valuable resource, pharmaceutical corporations, healthcare executives, and healthcare professionals must work together to maintain their usefulness. Rapid infection and outbreak detection and control, as well as regional initiatives to limit transmission within community and healthcare settings, are also necessary for resistance prevention. Misuse and overuse of antibiotics in healthcare continue to hasten the development of bacterial drug resistance, leading to loss of efficacy of existing antibiotics [3]. To reduce the likelihood of medication abuse, minimize antibiotic resistance, and give a general overview of drug use, a drug utilization study is proposed for drug-resistant tuberculosis.
The World Health Organization Anatomical Therapeutic Chemical/ Defined Daily Dose method is the standard for drug use [4]. The assumed average maintenance adult dose per day for each drug and route of administration is known as the defined daily dose, according to the World Health Organization Collaborating Centre for Drug Statistics and Methodology.
Because of this, the Defined Daily Dose is a universal unit that can be used for regional or global comparisons. Defined Daily Dose, on the other hand, does not always represent the recommended or prescribed daily dose. Indeed, several studies have demonstrated differences between Defined Daily Dose and Prescribed Daily Dose for different drug groups [5-8].
Numerous research have examined drug use patterns in different Indian healthcare sectors [9–19]. Finding out about drug use among hospital patients in respiratory ward was the aim of this study. In particular, they compared the average Prescribed Daily Dose to the World Health Organization Defined Daily Dose and examined prescriptions for the World Health Organization core parameters. The study's conclusions would provide the basis for identifying possible areas to improve the hospital's prescription practices and medication dispensing guidelines.
MATERIALS AND METHODS
The Medical Record Department (MRD) of a tertiary care hospital in Gujarat served as the site of this retrospective inpatient drug utilization investigation. The study was started after prior permission from the Institutional Human Research Ethical Committee (HREC) and the approval number is GMCS/STU/ETHICS/Approval/6603/21.
We gathered information by reviewing inpatient case files kept in the hospital's medical records division. We selected 301 case reports of individuals with drug-resistant TB from December 2018 to December 2021. The government drugstore provided the medications, while the treating physicians wrote the prescriptions. The national tuberculosis elimination program provided free drug comes to central drug store, from where patients procured all anti-tubercular medications. We carried out the data extraction and collection procedure. Utilizing a template that inquired about the patient's age, gender, diagnosis, length of hospital stay, and prescription information, including the name, strength, dosage form, frequency of administration, and quantity of units administered, it also inquired as to whether the medications were prescribed under a generic name. Additionally, we examined the World Health Organization core drug prescribing indicators. The average number of prescription drugs, the percentage of generic drugs, the percentage of prescriptions with antibiotics, the percentage of prescriptions with injections, the percentage of prescription drugs from an essential medication list, and the average cost of prescription drugs were some of the things that were looked at [1].
We coded all medications using the World Health Organization anatomical code as a guide. For all medications, we employed the Anatomical and Therapeutic Chemical Classification categorization scheme. To determine how many medications were prescribed from the essential list, we examined India's National List of Essential Medicines 2022 [20]. From prescription data for groups of commonly prescribed drugs based on the 2024 version of the Anatomical Therapeutic Chemical/Defined Daily Dose index, the number of drugs used was turned into the number of Defined Daily Dose. We determined Defined Daily Dose using the World Health Organization Defined Daily Dose measure and the quantity of medications issued for each item. We determined the number of Defined Daily Dose per 100 bed days.
To calculate the predicted Prescribed Daily Dose in grams for antibiotics, multiply the Defined Daily Dose by the number of treatment days and divide it by the number of Defined Daily Doses.
Statistical analysis
We present the data as mean, SD, and median (IQR), along with numbers and percentages. We entered all the data into a Microsoft Excel 2016 database and used jamovi version 2.3.18 software for statistical analysis. We evaluated no statistical hypothesis.
Inclusion criteria
Confirmed cases of drug-resistant tuberculosis have been admitted to the respiratory medicine department of a tertiary care hospital, involving patients aged 18 to 85 years, regardless of gender.
Exclusion criteria
The case paper does not provide sufficient details, nor does it address drug-resistant tuberculosis in individuals who are also co-infected with HIV.
RESULTS
During the study period, we reviewed a total of 301 case records. The patients included were admitted mainly due to drug resistance (treatment, adverse effect, and complication). 63.12% of the inpatients were male, while 36.88% were female. The average age of the patients was 36.2 years, with a standard deviation of 12.6. Hospital stays ranged from 1 to 23 d, with a median of 9 d.
Table 1 displays the various drug classes used in inpatients. All drugs administered to inpatients were antitubercular drugs. Following them were Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) (64.1%), proton pump inhibitors (61.5%), and vitamins and minerals (77.7%).
Table 1: Various classes of drugs are utilized in inpatient settings
| Drug class | Number of prescription |
| Anti-tubercular drugs | 301 (100 %) |
| Proton pump inhibitors | 185 (61.5 %) |
| H2 blockers | 116 (38.5 %) |
| Antiemetics | 128 (42.5 %) |
| NSAIDs | 193 (64.1%) |
| Corticosteroids | 36 (12 %) |
| Antihypertensive drugs | 72 (24 %) |
| Anti-diabetic drugs | 116 (38.5 %) |
| Vitamins and Minerals | 234 (77.7 %) |
People who were receiving Rifampicin-Resistant Tuberculosis (RR TB) treatment or Extreme Drug-Resistant (XDR) TB treatment were primarily the ones who came in with problems related to RR TB drug treatment. We noted complications such as pulmonary effusion, emphysema, bronchiectasis, pneumonia, lung abscess, pericarditis, liver or kidney failure, and psychosis. Common adverse reactions included vomiting, fever, tingling, numbness, jaundice, abdominal pain, and blurred vision. Following the assessment of the Q-T interval, liver function tests, renal function tests, X-ray imaging, and baseline optic and neurological examinations, we initiated TB treatments.
The drug prescriptions were analyzed for World Health Organization prescribing indicators and findings as per World Health Organization prescribing drug use was:
The average number of medicines prescribed per prescription was 11.52+2.34.
The percentage of medicines prescribed by a generic name was 92.42%.
The proportion of prescriptions that contain an antibiotic was 100%.
The percentage of prescriptions that included an injection was 22.69%.
The percentage of medicines prescribed by NLEM in 2022 was 95.33%.
Average drug cost per prescription was 675 INR.
A total of 3,469 medications were prescribed throughout 301 prescriptions, with 1,835 (52.9%) being antitubercular agents. Levofloxacin was the antitubercular drug that was taken in the most Defined Daily Doses. It was followed by ethionamide, clofazimine, and pyrazinamide. In contrast, levofloxacin had the most Defined Daily Doses per 100 bed days, followed by bedaquiline, delamanid, and moxifloxacin.
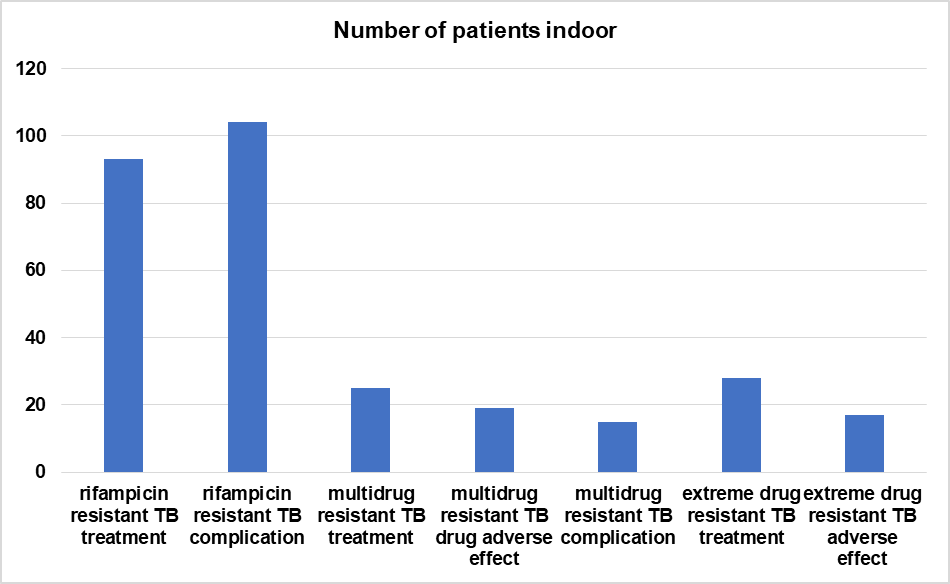
Fig. 1: Displays the number of inpatients with the specified diagnosis
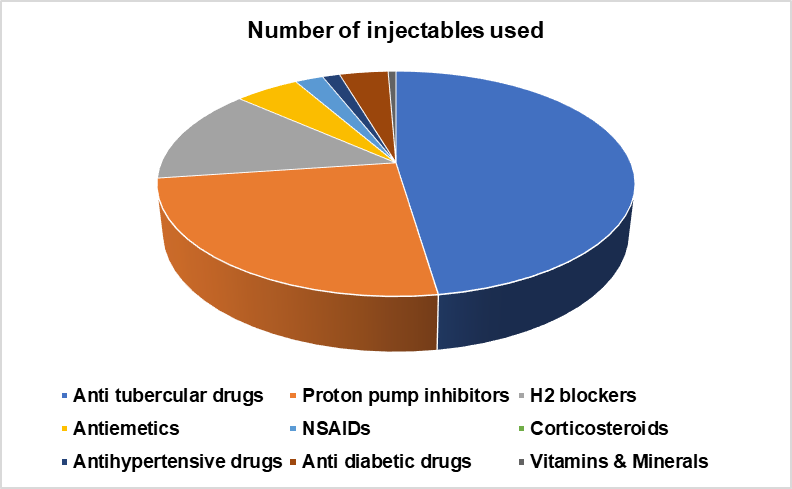
Fig. 2: Illustrates the number of injectables utilized in inpatient settings, Twenty-three percent of prescriptions included injectables, with antitubercular drugs being the most common. Proton pump inhibitors (5.7%), H2 blockers (3.1%), antiemetics (1.2%), and insulin (0.9%) were among the frequently utilized injectables
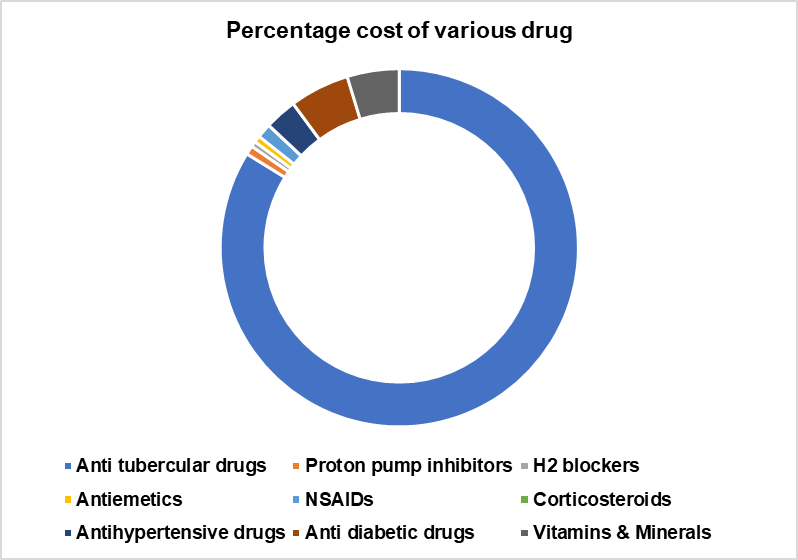
Fig. 3: Illustrates the percentage cost associated with different groups of drugs, Antitubercular drugs accounted for the majority of the total cost of prescribed medications, representing 83.8% of the expenditure. Recent anti-tubercular drugs like bedaquiline, delamanid, and linezolid incurred the primary expenses
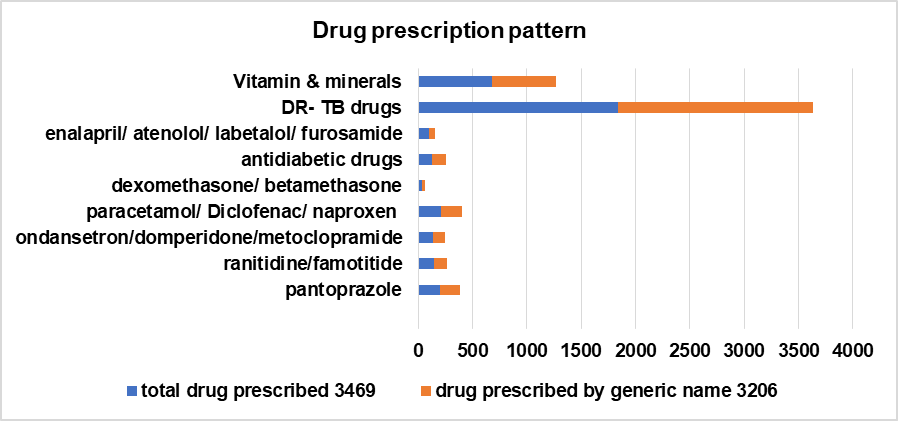
Fig. 4: Illustrates the drug prescription pattern, approximately 92.42% of prescriptions are issued using their generic names. Prescribed branded medications included anti-tubercular agents, vitamins and minerals, anti-inflammatory agents, proton pump inhibitors, and anti-diabetic medications
Table 2: Analysis of estimated prescribed daily doses versus defined daily doses for various anti-tubercular medications
| Drug name | Anatomical therapeutic chemical code | Defined daily dose (g) | Bed days | Defined daily dose per 100 bed days | World health organisation defined daily dose (g) | Prescribed daily dose (g) |
| Levofloxacin (oral) | J01MA12 | 769.5 | 207 | 372 | 0.5 | 1.86 |
| Levofloxacin (intravenous) | J01MA12 | 145.5 | 114 | 128 | 0.5 | 0.64 |
| Moxifloxacin (oral) | J01MA14 | 194 | 93 | 209 | 0.4 | 0.84 |
| Bedaquiline (oral) | J04AK05 | 263.72 | 101 | 261 | 0.86 | 2.24 |
| Linezolid (intavenous) | J01XX08 | 188.5 | 192 | 98 | 1.2 | 1.18 |
| Clofazimine (oral) | J04BA01 | 413 | 337 | 123 | 0.1 | 0.12 |
| Cycloserine (oral) | J04AB01 | 183.48 | 218 | 84 | 0.75 | 0.63 |
| Terizidone (oral) | J04AK03 | 124.66 | 115 | 108 | 0.75 | 0.81 |
| Ethambutol(oral) | J04AK02 | 227.33 | 158 | 144 | 1.2 | 1.73 |
| Ethambutol (intravenous) | J04AK02 | 60.66 | 41 | 148 | 1.2 | 1.78 |
| Delamanid (oral) | J04AK06 | 214 | 100 | 214 | 0.2 | 0.43 |
| Pyrazinamide (oral) | J04AK01 | 409.33 | 421 | 97 | 1.5 | 1.46 |
| Imipenem – Cilastatin (intravenous) | J01DH51 | 105.5 | 111 | 95 | 2.0 | 1.9 |
| Meropenem (intravenous) | J01DH02 | 55 | 73 | 75 | 3.0 | 2.25 |
| Amikacin (intravenous) | J01GB06 | 28.7 | 29 | 99 | 1.0 | 0.99 |
| Streptomycin (intramuscular) | J01GA01 | 17.5 | 25 | 70 | 1.0 | 0.70 |
| Ethionamide (oral) | J04AD03 | 538.66 | 439 | 123 | 0.75 | 0.92 |
| Prothionamide (oral) | J04AD01 | 201.33 | 207 | 97 | 0.75 | 0.73 |
| Para amino salicylic acid (PAS) (oral) | J04AA01 | 54 | 83 | 65 | 12 | 7.8 |
DISCUSSION
Our study records the utilization pattern of tertiary care inpatients in India. In a study carried out at a tertiary care hospital's emergency department and other wards, antibiotics came into existence as the most regularly administered class of medications [19]. A study on the changing paradigm for treating drug-resistant tuberculosis found that fluoroquinolones were the most common daily doses. Following these were newer antitubercular medications like bedaquiline and delamanid [3]. The most often given medications for drug-resistant tuberculosis were injectable aminoglycosides and fluoroquinolones. Its wider assortment of action, shifting resistance patterns, and prescription practices might have contributed to this.
The average Prescribed Daily Dose was higher than Defined Daily Dosefor fluoroquinolones, bedaquiline, and delamanid, but lower than Defined Daily Dose for other anti-tubercular medications. This could be because the safety profiles of these two categories differ. A few other studies found big differences between Prescribed Daily Dose and Defined Daily dose for most antibiotics. This study, on the other hand, found that Prescribed Daily Dose and Defined Daily Dose were generally the same for most antibiotics, which is in line with what the whole world recommends [8, 21].
Like a study where antibiotics accounted for almost 50% of drug cost spending, anti-tubercular drugs had the highest percentage expenditures (83.8%) of all drug classes [18, 19]. Drugs like linezolid, delamanid, and bedaquiline were mostly responsible for these exorbitant expenses. Another factor contributing to the rise of germ resistance is the inappropriate use of antibiotics [22]. To decrease the quantity of needless and frequent prescriptions for antibiotics, especially the more current and costly ones, this situation calls for modifications to the hospital's antibiotic policy. Designing a program to limit the use of antibiotics might be a major first step in implementing this strategy. The hospital must compile a list of prohibited antibiotics in compliance with this policy, taking into account current resistance trends and expenses. Before any antibiotics are initiated or distributed, a member of the infectious diseases team, a multidisciplinary team of doctors and pharmacists, must give their prior consent. This tactic may be invaluable to combat rising healthcare costs and the emergence of resistance to novel, vulnerable agents. Past studies have demonstrated the effectiveness of this program in reducing antibiotic use [23]. Another tried-and-true method for enhancing prescribing practices is to make practice guidelines accessible to doctors during clinical decision-making [24]. Furthermore, the use of preventive antibiotics needs to be limited. Antiemetics, proton pump inhibitors, H2-blocking medications, vitamins, and minerals remained across the other regularly prescribed medications. We need to conduct a more comprehensive investigation to confirm the medical recommendation for using these supplements.
Even though generic prescribing has many benefits, such as lower drug costs, better patient adherence, and therapeutic effects like brand-name drugs, it is not commonly used in India [25, 26]. In our survey, generic names accounted for almost 92% of prescriptions. About 23 percent of prescriptions were for injectable drugs, primarily antibiotics. Given that the trial took place in an inpatient setting with mostly critically sick patients, the large proportion of injectables was understandable. Injectable drugs are associated with administration issues and dosage mistakes [27]. Therefore, it is essential transition to alternative administration methods as quickly as feasible. Approximately 95% of prescriptions were for the Indian National List of Essential Medicines, 2022, which is based on the World Health Organization Essential Drugs List [28]. It is necessary to examine the idea of essential medicines and their accessibility to different societal groups.
On average, there were 11.5 medications in prescriptions. Prior studies [29, 30] have demonstrated a comparable pattern. The drawbacks of polypharmacy include high medical expenses, poor patient adherence, a greater frequency of adverse events [31, 32], and drug-drug interactions [17, 19]. Consequently, determining predictors of polypharmacy and adjusting prescribing procedures are always required. The median cost per prescription in the respiratory ward of the tertiary care hospital ranges from 349 INR to 3388 INR, whereas the average cost of a prescription for drug-resistant tuberculosis was 675 INR. In a nation like India, where the government provides for most the population's medical needs, this puts a significant financial strain on the government. Therefore, we must modify the prescription patterns, focusing on providing generic medications, restricting the use of injectables, and reducing polypharmacy. We also need to make a few changes to the hospital's antibiotic policy. Furthermore, creating a system that enables patients to obtain prescription drugs at a reduced cost may be a beneficial first step in reducing the overall burden of medical expenses.
CONCLUSION
Drug costs were significantly impacted by the fact that antibiotics were the most often-given drugs, accompanied by vitamins and minerals, NSAIDs, and proton pump inhibitors. Limiting the use of more costly, recent antibiotics, branded medications, and the number of medications given can drastically lower the cost of drug therapy. We worked diligently to provide the tuberculosis elimination program with the expertise and experience it needed to make decisions and use its resources in an efficient manner to achieve cost-effective healthcare in response to the demands of policymakers who sought quantitative analysis and improved TB control policy and implementation.
LIMITATIONS OF STUDY
Multicentric studies would provide a more comprehensive understanding of the prevalence and treatment outcomes of drug-resistant tuberculosis across different regions in India. This would allow for more targeted interventions and strategies to combat this public health issue effectively.
FUNDING
The National Tuberculosis Elimination Program's operational research provided funding for this study.
AUTHORS CONTRIBUTIONS
In addition to helping with data collection, analysis, and result interpretation, Dr. Vijita Shah was involved in the study's inception and design. Drs. Vijita Shah, Jigar Chotaliya, and Bhargavi Patel all contributed to the manuscript's preparation, critical revision for noteworthy intellectual substance, and publication approval. Throughout the research procedure, Dr. Chetna Patel offered direction, guaranteeing the correctness and integrity of the study. The cooperation of all authors allowed for the successful completion of this project.
CONFLICT OF INTERESTS
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
REFERENCES
Introduction to drug utilization research. WHOCC. 2003;92(4):156234.
National strategic plan for tuberculosis elimination 2017-2025. New Delhi; 2017. Available from: https://tbcindia.gov.in/WriteReadData/NSPDraft20.02.20171.pdf. [Last accessed on 11 Mar 2025].
Dookie N, Ngema SL, Perumal R, Naicker N, Padayatchi N, Naidoo K. The changing paradigm of drug-resistant tuberculosis treatment: successes, pitfalls, and future perspectives. Clin Microbiol Rev. 2022;35(4):e0018019. doi: 10.1128/cmr.00180-19, PMID 36200885.
WHO Collaborating Centre for Drug Statistics Methodology. Guidelines for ATC classification and DDD assignment; 2024. Available from: http://www.whocc.no/filearchive/publications/2024guidelines.pdf. [Last accessed on 11 Mar 2025].
Koristkova B, Grundmann M, Brozmanova H. Differences between prescribed daily doses and defined daily doses of antiepileptics-therapeutic drug monitoring as a marker of the quality of the treatment. Int J Clin Pharmacol Ther. 2006;44(9):438-42. doi: 10.5414/cpp44438, PMID 16995332.
Muller A, Monnet DL, Talon D, Henon T, Bertrand X. Discrepancies between prescribed daily doses and WHO defined daily doses of antibacterials at a university hospital. Br J Clin Pharmacol. 2006;61(5):585-91. doi: 10.1111/j.1365-2125.2006.02605.x, PMID 16669851.
Duarte Ramos F, Cabrita J. Using a pharmaco-epidemiological approach to estimate diabetes type 2 prevalence in portugal. Pharmacoepidemiol Drug Saf. 2006;15(4):269-74. doi: 10.1002/pds.1186, PMID 16294365.
Grimmsmann T, Himmel W. Discrepancies between prescribed and defined daily doses: a matter of patients or drug classes? Eur J Clin Pharmacol. 2011;67(8):847-54. doi: 10.1007/s00228-011-1014-7, PMID 21544512.
Ghosh BN, Mitra J, Das KK. Prescription habits of physicians in an urban locality. Indian J Public Health. 1987;31(2):120-8. PMID 3453359.
Ahmad SR, Bhutta ZA. A survey of paediatric prescribing and dispensing in Karachi. J Pak Med Assoc. 1990;40(6):126-30. PMID 2118192.
Phadke AR. The quality of prescribing in an Indian District. Natl Med J India. 1996;9(2):60-5. PMID 8857039.
Das NP, Saha NN, Roy ND. Drug utilization study of psychotropic drugs in psychiatry outdoor patients at A Tertiary Care Hospital. Asian J Pharm Clin Res. 2024:56-60. doi: 10.22159/ajpcr.2024v17i7.51781[PubMed].
Mandal A, Gangopadhyay T, Mandal S. Drug utilization study in a radiotherapy unit of a tertiary care teaching hospital in rural West Bengal, India. Asian J Pharm Clin Res. 2022b;15(11):126-30. doi: 10.22159/ajpcr.2022.v15i11.45709.
Naik PS, Kandra NV, Bai KS. A prospective, observational drug utilization study in the dermatology outpatient department of a government hospital, nandyal, Andhra Pradesh. Int J Curr Pharm Sci. 2024;16(6):89-92. doi: 10.22159/ijcpr.2024v16i6.5087.
Hazra A, Tripathi SK, Alam MS. Prescribing and dispensing activities at the health facilities of a non-governmental organization. Natl Med J India. 2000;13(4):177-82. PMID 11002683.
Biswal S, Mishra P, Malhotra S, Puri GD, Pandhi P. Drug utilization pattern in the intensive care unit of a Tertiary Care Hospital. J Clin Pharmacol. 2006;46(8):945-51. doi: 10.1177/0091270006289845, PMID 16855079.
Choure MK, Jadhav RR, Padwal SLP. Drug utilization study in neonatal intensive care unit at rural Tertiary Care Hospital. Asian J Pharm Clin Res. 2017 Apr 1;10(4):102-4. doi: 10.22159/ajpcr.2017.v10i4.16111.
Singh I, Mittal R, Shafiq N, Bharati B, Nigah RK, Pandhi P. A drug utilization study to provide background data for bringing amendments in the drug dispensing policy of a pediatric referral center. Pharmacoepidemiol Drug Saf. 2010;19(4):393-9. doi: 10.1002/pds.1832, PMID 20020440.
Dhamija P, Bansal D, Srinivasan A, Bhalla A, Hota D, Chakrabarti A. Patterns of prescription drug use and incidence of drug-drug interactions in patients reporting to medical emergency. Fundam Clin Pharmacol. 2013;27(2):231-7. doi: 10.1111/j.1472-8206.2011.00990.x, PMID 21895763.
National List of Essential Medicine; 2022. http://www.cdsco.nic.in/nedl.pdf. [Last accessed on 11 Mar 2025]
Bro F, Mabeck CE. Use of antibiotics in general practice in Denmark. Prescribed daily dose, duration of treatment and number of treatments in general practice. Scand J Prim Health Care. 1986;4(2):101-4. doi: 10.3109/02813438609014811, PMID 3726323.
Gyssens IC. Quality measures of antimicrobial drug use. Int J Antimicrob Agents. 2001;17(1):9-19. doi: 10.1016/s0924-8579(00)00208-9, PMID 11137643.
Mansouri MD, Cadle RM, Agbahiwe SO, Musher DM. Impact of an antibiotic restriction program on antibiotic utilization in the treatment of community-acquired pneumonia in a veterans affairs medical center. Infection. 2011;39(1):53-8. doi: 10.1007/s15010-010-0078-0, PMID 21318422.
Westphal JF, Jehl F, Javelot H, Nonnenmacher C. Enhanced physician adherence to antibiotic use guidelines through increased availability of guidelines at the time of drug ordering in hospital setting. Pharmacoepidemiol Drug Saf. 2011;20(2):162-8. doi: 10.1002/pds.2078, PMID 21254287.
Haas JS, Phillips KA, Gerstenberger EP, Seger AC. Potential savings from substituting generic drugs for brand-name drugs: medical expenditure panel survey, 1997-2000. Ann Intern Med. 2005;142(11):891-7. doi: 10.7326/0003-4819-142-11-200506070-00006, PMID 15941695.
Shrank WH, Hoang T, Ettner SL, Glassman PA, Nair K, DeLapp D. The implications of choice: prescribing generic or preferred pharmaceuticals improves medication adherence for chronic conditions. Arch Intern Med. 2006;166(3):332-7. doi: 10.1001/archinte.166.3.332, PMID 16476874.
Taxis K, Barber N. Ethnographic study of incidence and severity of intravenous drug errors. BMJ. 2003;326(7391):684. doi: 10.1136/bmj.326.7391.684, PMID 12663404.
WHO Essential medicine list. Available from: http://www.who.int/medicines/publications/08_english_indexfinal_EML22.pdf. [Last accessed on 11 Mar 2025].
Chaudhury RR, Parameswar R, Gupta U, Sharma S, Tekur U, Bapna JS. Quality medicines for the poor: experience of the Delhi programme on rational use of drugs. Health Policy Plan. 2005;20(2):124-36. doi: 10.1093/heapol/czi015, PMID 15746221.
Junius Walker U, Theile G, Hummers Pradier EH. Prevalence and predictors of polypharmacy among older primary care patients in germany. Fam Pract. 2007;24(1):14-9. doi: 10.1093/fampra/cml067, PMID 17164234.
Kojima T, Akishita M, Nakamura T, Nomura K, Ogawa S, Iijima K. Association of polypharmacy with fall risk among geriatric outpatients. Geriatr Gerontol Int. 2011;11(4):438-44. doi: 10.1111/j.1447-0594.2011.00703.x, PMID 21545384.
Lai SW, Liao KF, Liao CC, Muo CH, Liu CS, Sung FC. Polypharmacy correlates with increased risk for hip fracture in the elderly: a population-based study. Med (Baltim). 2010;89(5):295-9. doi: 10.1097/MD.0b013e3181f15efc, PMID 20827106.