
Int J Pharm Pharm Sci, Vol 18, Issue 1, 1-6Review Article
CEREBRAL PALSY: BRIDGING CLINICAL PRACTICE AND RESEARCH ADVANCES
SUTHA PONNUSAMY1*, NITHYAPRIYA KARUPPUSAMY2, SANGAMESWARAN BALAKRISHNAN3, PADMAPRIYA R.4, PALANIBHARATHI T.5, PAVITHRA M.6, PRADEEPKUMAR P.7, MADHIVANAN K.8
1,2Department of Pharmaceutics, SSM College of Pharmacy, Erode-638312, TN, India, 3SSM College of Pharmacy, Erode-638312, TN, India. 4,5,6,7,8SSM College of Pharmacy, Erode-638312, TN, India. The Tamil Nadu Dr. M. G. R Medical University, Guindy, Chennai-600032, TN, India
*Corresponding author: Sutha Ponnusamy; *Email: suthaharini3186@gmail.com
Received: 24 Jun 2025, Revised and Accepted: 13 Dec 2025
ABSTRAC
Permanent movement deficits as an outcome of non-progressive disruptions during the process of cerebral development are the hallmark of cerebral palsy (CP), a common developmental condition. Greater immaturity and lower birth weight are associated with a higher prevalence of CP. CP continues to be a leading cause of childhood disability globally, despite improvements in clinical care. The gap between discovery and application can be closed by combining state-of-the-art research with patient-centered clinical practice, offering new hope for improving the quality of life of individuals with cerebral palsy and their families. Recent advancements also demonstrate how Artificial intelligence (AI) has the potential to revolutionize CP management.-powered technologies are improving early diagnosis by using predictive modeling and sophisticated image analysis. Early treatments are made possible by machine learning algorithms that assist in detecting infants who are at risk even before clinical symptoms manifest. Children with cerebral palsy are benefitting from improved movement, communication, and rehabilitation results thanks to robotics and AI-based assistive technologies. The continuous attempts to connect clinical practice with new research are highlighted in this study. It discusses on early diagnosis, management techniques meant to improve patient outcomes, and the growing understanding of CP risk factors. The way that care is provided is changing as a result of recent scientific developments in fields including Neuron-protective medicines, regenerative medicine, and rehabilitation technology. Furthermore, more effective and customized treatment techniques are being made possible by AI applications in remote patient monitoring and individualized therapy planning.
Keywords: Cerebral palsy, Artificial intelligence, Rehabilitation
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijpps.2026v18i1.55732 Journal homepage: https://innovareacademics.in/journals/index.php/ijpps
INTRODUCTION
Cerebral palsy (CP) affects a person's posture, balance, and movement, in accordance with the Centers for Disease Control and Prevention. Due to damage to the developing brain, the clinical findings are non-progressive and permanent, although they may change over time. CP is a group of permanent movement disorders caused by non-progressive brain injury. In the United States, one out of every 323 children has cerebral palsy, making it the most common childhood physical disability. Over time this fig. has stayed rather steady. It is generally acknowledged that there must be motor impairment, and that this impairment must be caused by brain dysfunction as opposed to spinal cord or muscle issue. Additionally, the brain abnormality must appear early in life and not worsen with time. Low birth weight and prematurity are significant risk factors for cerebral palsy, and other factors, such as multiple gestation and maternal illnesses, have also been linked to increased risk for cerebral palsy. Cerebral palsy symptoms are brought on by issues with brain development, which might include: White matter damage in the brain, disruption of brain development due to genetic changes, brain hemorrhage [bleeding], lack of oxygen, infections during pregnancy, such as CMV or rubella, severe infant jaundice that causes kernicterus, traumatic brain injury in early childhood, maternal conditions such as seizures, thyroid disorders, or toxin exposure, issues during delivery, such as umbilical cord complications or uterine rupture.
Search strategy: The literature for this review was collected using PubMed, Google Scholar, and Scopus databases using keywords like “cerebral palsy,” “artificial intelligence,” “neuroplasticity,” and “robotic rehabilitation.” Articles published between 2013 and 2024 were included.
Aetiology and risk factors
A static neurologic brought on by brain injury that happens before the brain develops fully is cerebral palsy. Since brain development continues during the first two years of life, brain injury that happens during the prenatal, perinatal, or postnatal periods can result in cerebral palsy. Acquired cerebral palsy is the outcome of brain damage that happens in the first few months or years of life. This damage can be caused by brain conditions like viral encephalitis or bacterial meningitis. Additionally, a head injury, typically from a car accident, a fall, or child abuse during the first few years of life, when the brain is still developing can cause it. Cerebral palsy results from a non-progressive (or "static") brain injury that may occur during the prenatal, perinatal, or postnatal period. An individual patient's etiology is frequently complex.
Prenatal causes
Congenital brain deformities, intrauterine infections, intrauterine strokes, and chromosome abnormalities.
Perinatal causes
Hypoxic-ischemic insults, infections of Central nervous system (CNS), stroke, and kernicterus
Postnatal causes
Unintentional and intentional trauma, CNS infections, stroke, and noxious insult.
A number of risk factors, which might exist before, during labor or after pregnancy, soon after the infant is born, are important in the pathophysiology of cerebral palsy. We must differentiate between the known etiology of CP, risk factors, and relationships. In many instances, it was discovered that a sequence of events rather than a single incident caused the motor nerve injury that ultimately led to this illness. Maternal systemic illness, substance use, undernutrition, exposure to harmful substances, reproductive problems, and a history of spontaneous miscarriage are some the risk factors associated with cerebral palsy. Fig. 2 summarizes a number of other risk factors that might result in brain injury before, during, and after delivery.
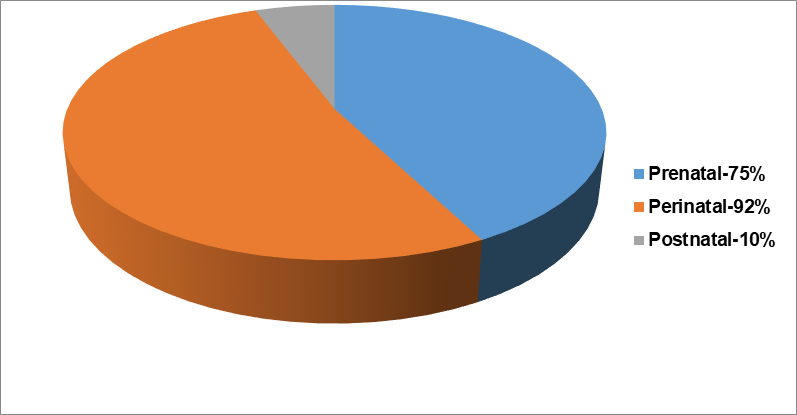
Fig. 1: Causes of cerebral palsy [Ref 8]
Table 1: Risk Factors of Cerebral Palsy
| Category | Examples | References |
| Maternal Factors | Maternal infections (rubella, CMV), thyroid disease, diabetes, poor nutrition | [5, 6] |
| Pregnancy Factors | Prematurity, multiple gestation, placental complications | [5] |
| Environmental Factors | Maternal alcohol use, exposure to toxins, postnatal trauma | [6] |
| Delivery Factors | Prolonged labor, Birth asphyxia, umbilical cord issues | [5, 6] |
| Neonatal Factors | Jaundice, hypoglycemia, meningitis, neonatal stroke | [5] |
Types of cerebral palsy
Cerebral palsy can be divided into three main categories according on the symptoms and treatment strategies used, that is Spastic, ataxic, and athetoid.
Spastic cerebral palsy
The hallmark of this most common type of cerebral palsy is the muscle spasticity, which can be managed with physical and occupational therapy. Every day, the disease is managed by physical activities such as stretching, strengthening, and exercising.
Ataxic cerebral palsy
"Ataxia-type" symptoms that cause some cerebellar damage are the hallmark of ataxic CP. The child may show signs of unstable posture and tremors when attempting to grasp objects. These signs and symptoms are a result of the child's impaired motor skills. When walking, the child may appear unsteady or lack coordination. In ataxic CP, visual and auditory processing may also be impacted.
Athetoid cerebral palsy
It may be difficult for patients with this kind of illness to maintain stable posture. It can be difficult to sit and walk steadily; people may make some unexpected movements. Additionally, patients may become incapable of grasping things, particularly little ones that call for sophisticated or fine motor control. Pens, coins, and other small objects may be difficult for these patients to grasp.
Diagnosis of cerebral palsy
Hand preference rarely occurs before 12 mo of age. Poor muscle tone, delayed motor development, and unusual posture are common early signs of cerebral palsy, in which spasticity is often present. After six months, the Moro reflex is rarely observed in newborns without cerebral palsy, and its persistence may prompt referrals to specialists such as occupational therapists, neurologists, and developmental pediatricians. Depending on which limbs are impacted, there are three types of palsy: can be classified as diplegia, hemiplegic, and quadriplegia.
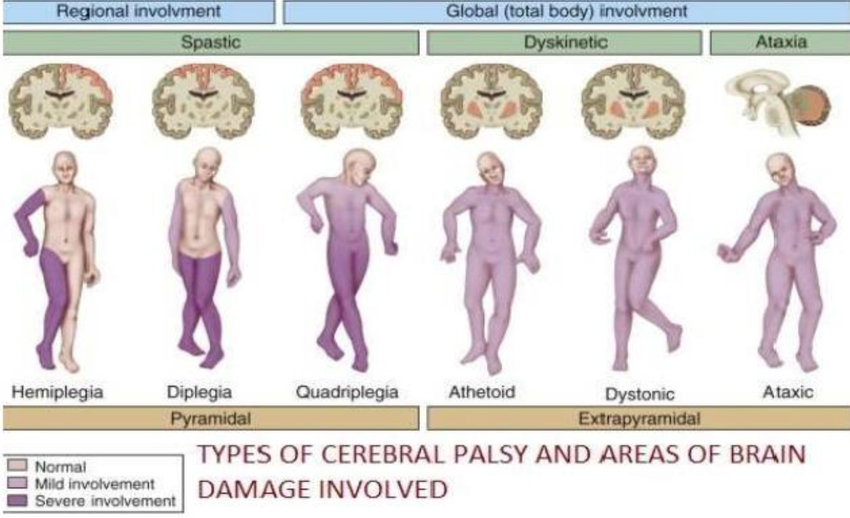
Fig. 3: Types of cerebral palsy
Source: Created by authors
Magnetic resonance imaging (MRI)
An MRI creates finely detailed 3D or cross-sectional images of the brain by using radio waves and a magnetic field. Magnetic resonance imaging (MRI) can often detect changes in your child’s brain. Although this test doesn't hurt, it can take up to an hour to finish and is noisy. Before the procedure, your child will probably be given a sedative or mild general anesthetic.
Cranial ultrasound
This can be done when the child is still a baby. High-frequency sound waves are used in cranial ultrasounds to create images of the brain. Although it cannot produce detailed images, cranial ultrasound is a quick and useful tool for initial brain evaluation.
Electroencephalogram (EEG)
An electroencephalogram (EEG) can be used to further assess your child's potential seizure condition. Children with epilepsy may experience seizures. A number of electrodes are affixed to your child's scalp during an EEG test. The EEG records your child’s brain’s electrical activity. Changes in brain wave patterns are prevalent in epilepsy.
Laboratory tests
To check for genetic or metabolic disorders, blood, urine, or skin tests may be performed.
Additional tests
Your child will probably be referred to experts for testing for additional problems if they are diagnosed with cerebral palsy. These assessments may examine speech, hearing, vision, movement, intellect, and development.
Genetics syndrome
The criteria for CP should be met by certain hereditary diseases, like familial spastic paraplegia. Traditionally, CP registers have eliminated other chromosomal/genetic abnormalities that are easily identifiable (such as typical dysmorphisms) and linked to motor disability. However, it cannot be regarded as a component of the syndrome if the kind or degree of motor abnormalities seen is not a characteristic of the genetic condition, the case ought to be included (for instance, spastic diplegia in a child with Down syndrome). For instance, in a child with Down syndrome, spastic hemiplegia may warrant inclusion in the CP category if not characteristics of the genetic disorder. As previously mentioned, there may be disagreement over whether clinically identifiable disorders should be classified as cerebral palsy.
Management and treatment
The objective of managing cerebral palsy is to maintain mobility, independence, social engagement, and cognitive development while improving functional abilities. The aim is not to cure the condition or restore normalcy. Rather than focusing solely on individual symptoms, a modern team-based approach emphasizes the patient's overall development. Treatment plans include behavioral and physical therapy, medication and surgery, mechanical assistance, and the treatment of related illnesses. One of the biggest challenges facing the treatment team is managing spasticity.
Early actions
For kids who have cerebral palsy, early identification as well as intervention is essential to maximizing results. Neuroplasticity and functional development can be improved by starting therapies like occupational therapy, speech-language therapy, and physical therapy in infancy. Early neurodevelopment evaluations may help predict the severity of CP; enabling timely interventions that support brain rewiring and improve long-term outcomes, according to a Monish Children's Hospital study.
Physiotherapy and occupational therapy
Through focused exercise interventions, physiotherapy seeks to enhance motor function, balance, and mobility; occupational therapy concentrates on improving everyday life skills and encouraging independence; both therapies are most successful when initiated early and customized to meet the individual needs of the child.
Spasticity management
One typical characteristic of CP is spasticity, which is defined by increased muscular tone. Among the management techniques are;
Pharmacological Treatments: Drugs that lower muscular tone include tizanidine, baclofen, diazepam, and dantrolene
Botulinum Toxin Injections: These injections increase function and lessen stiffness by targeting particular muscles. The effects are transient and typically last for three to six months.
Intrathecal Baclofen Therapy: An implanted pump can inject baclofen straight into the spinal fluid to provide more consistent relief from severe spasticity.
Surgical interventions
When conservative measures are not enough, surgical options are taken into consideration:
Selective dorsal rhizotomy (SDR): To lessen spasticity, a neurosurgical technique entails chopping off particular nerve roots. Children aged 3 and 8 benefit best with SDR.
Orthopedic surgery: Techniques such as joint stabilization, osteotomies, and tendon lengthening are used to correct musculoskeletal abnormalities and improve range of motion.
Orthotic devices
Ankle-foot orthoses (AFOs) and other Orthoses are used to: Increase gait efficiency, prevent or treat abnormalities, Provide joint stability. There is evidence supporting the use of AFOs to improve walking ability in ambulant children (GMFCS levels I–III).
Communication and cognitive support
While non-verbal individuals benefit from augmentative and alternative communication (AAC) technology, speech and language therapy treats communication difficulties. Learning and cognitive development are supported by educational interventions and cognitive-behavioral therapy.
Nutritional management
CP patients frequently have trouble feeding. Nutritional examinations and interventions are part of management.
In cases where oral intake is insufficient, enteral feeding (such as via gastrostomy) is recommended. Adequate nutrition is essential for growth, health, and overall well-being.
Assistive technologies
Technology improves life quality and independence: Mobility aids such as wheelchairs and walkers, communication tools like speech-generating devices, rehabilitation technologies including robotics and virtual reality. Equipment that is adaptable for everyday tasks Rehabilitation programs is incorporating new technologies like virtual reality and robots.
Challenges and quality of life
Challenges
Children with cerebral palsy face several challenges in their daily life. They often experience motor impairments that affect movement, balance, and coordination. Spasticity, which involves increased muscle tone, leads to stiffness and spasms. Many children also struggle with communication difficulties that impact speech and language skills. Persistent pain and discomfort are common and may affect overall well-being. In addition, they have a higher risk of developing secondary conditions such as osteoporosis, scoliosis, and seizures.
Table 2: Management and treatment
| Multidisciplinary management group for patients with cerebral palsy | |
| Team member | Role |
| Doctor | Team leader who combines comprehensive, long term strategies and treatments[2] |
| Surgical specialist | Focuses on preventing contractures, hip dislocations, and spinal curvatures in addition to treatment of pain[2] |
| Physical therapist | Develops and implements care plans to improve movement and strength, and administers formal gait analyses[2] |
| Occupational therapist | Develops and implements care plans focused on activities of everyday existence[2] |
| Pathologist for speech language | Creates and carries out care plans to maximize the patient’s communicative abilities.[2] |
| Social worker | Helps the patient’s relatives find local aid opportunities.[2] |
| Psychologist | Helps the patient and their family deal with the responsibilities and stress of their handicap.[2] |
| Educator | Creates plans to deal with learning or cognitive impairments.[2] |
Effect on life quality
Children with cerebral palsy often experience limited mobility, which reduces their freedom and ability to move independently. They may also face social challenges, including difficulty forming relationships and participating in social activities. Emotional health concerns such as increased stress, anxiety, and depression are common. Additionally, many children struggle with activities of daily living, including essential tasks like eating, dressing, and bathing.
Improving quality of life
Rehabilitation therapies such as physical, occupational, and speech therapy play a crucial role in enhancing functional abilities in children with cerebral palsy. Assistive devices, including communication aids, walkers, and wheelchairs, further support mobility and independence. Pain is managed through prescription medications, counselling, and various complementary approaches. Support networks involving families, caregivers, and community groups provide essential emotional and practical assistance. Creating inclusive environments, with accessible public spaces and community support, also helps improve participation and quality of life for children with cerebral palsy.
Importance of support and inclusion
Promoting independence is crucial in cerebral palsy management, as it encourages autonomy and self-advocacy in children. Equally important is fostering inclusion by creating accessible and supportive environments that enable full participation in daily life. Providing adequate support through emotional, physical, and financial assistance further strengthens the overall well-being of individuals with cerebral palsy and their families.
By acknowledging the challenges and promoting support and inclusion, we can enhance the standard of living for those who have cerebral palsy.
Recent advances
Early diagnosis and neuroplasticity
Infants as young as three months can now be diagnosed with CP thanks to advancements in early neurodevelopment assessments. Timely therapies that take advantage of the brain's adaptability are made possible by early identification, which improves functional outcomes. Research has indicated that early therapies can greatly improve children with cerebral palsy's physical and cognitive abilities.
Cell-Based therapies
Recent studies on cell-based treatments, such as and mesenchymal stem cells (MSCs) and neural stem/progenitor cells, have demonstrated promise in enhancing motor abilities and lessening spasticity in CP patients. The results of clinical studies can vary, which emphasizes the need for more research and established procedures.
Technological innovations
New instruments for rehabilitation have been made possible by technological advancements:
Robotics: Tools such as the Lokomat and Innowalk support motor learning and brain restructuring while aiding in gait training.
Virtual reality (VR): interactive environments that promote training and reduce pain.
Augmentative and alternative communication (AAC): AAC devices improve social engagement and involvement by helping non-verbal people communicate.
Pain management
Children with cerebral palsy may experience chronic discomfort. The significance of customized pain assessment instruments and therapies has been emphasized by recent research. An important advancement in the management of this aspect of CP has been made with the creation of age-appropriate and communicative pain scales.
Future directions
Customized healthcare
Personalized strategies that take individual, environmental, and genetic factors into account are the way of the future for managing cerebral palsy. Interventions can be more effective and produce better results if they are customized to each patient's unique needs.
Strategies for neuroprotection
The goal of neuroprotective agent research is to avoid or lessen brain damage during crucial developmental periods. In preclinical research, substances like erythropoietin and resveratrol have demonstrated promise by lowering neuroinflammation and fostering neuroprotection.
Multidisciplinary care integration
It is crucial to have a comprehensive approach that includes psychologists, occupational therapists, neurologists, physiotherapists, and speech therapists. Collaborative care guarantees all-encompassing treatment, attending to the many requirements of people with cerebral palsy.
Global access and equity
It is essential to ensure that advancements in CP management are accessible globally, especially in low-resource settings. Programs that train medical professionals and offer reasonably priced treatments can help close the care gap. Further research into prenatal infections, the function of inflammatory indicators and coagulation, and the application of advanced imaging of brain techniques is expected to deepen our understanding of CP’s etiology. Categorizing different forms of CP based on clinical characteristics and gestational age may also help identify specific risk patterns and guide preventive strategies. The crucial first step in creating prevention methods will be conducting ongoing research on the causes and risk factors of cerebral palsy.
Technological advancement
Children with CP are experiencing a transformation in rehabilitation through advancement in technology, particularly in robotics and artificial intelligence (AI). These developments are meant to improve communication, motor function, and general quality of life. An outline of significant developments is provided below:
Training for robot-assisted gait (RAGT)
In order to enhance lower limb function, robotic devices like Lokomat, Innowalk, Robogait, and Walkbot-K are being included into CP rehabilitation more and more. These tools promote motor learning and brain reconfiguration by providing recurrent, task-specific training with modifiable parameters. The overall effectiveness is still being studied, even though some research show improvements in gait metrics. Comprehensive reviews suggest that these systems are effective as complimentary tools to conventional therapy but are not sufficient as stand-alone treatments.
Robotic assistance for the upper limb
There is growing research on robotic therapies for CP patients' upper limb rehabilitation. Virtual robotic systems that designed to stimulate specific movements can improve motor skills and social interaction in children. These systems aim to enhance engagement by extending therapy session and increasing repetitions.
Socially assistive robots (SARS) and Al integration
Socially assistive robots such as NAO are used to motivate children with CP during therapy sessions. These robots can engage in verbal and non-verbal communication, making therapy more interactive and enjoyable. Al integration allows these robots to adapt to individual needs, providing personalized support
Non-contact rehabilitation systems and virtual reality
Virtual reality and motion capture are two examples of non-contact rehabilitation technologies that provide alternative therapeutic approaches. For kids who are touch-sensitive, these systems allow them to perform rehabilitation exercises without making physical contact. Additionally, they make it possible to personalize therapy sessions, which boost engagement and motivation. These technical developments, which combine Al and robotics, have the potential to improve the therapy results for kids with cerebral palsy. Further research is needed to develop standardized procedures and assess long-term effectiveness.
CONCLUSION
CP remains a complex neurodevelopmental disorder with lifelong implications. However, advancements in research, early diagnosis, and multidisciplinary care are transforming its management. Emphasizing early intervention, personalized therapy, and the integration of innovative technologies—such as robotics, virtual reality, and AI-driven diagnostics—has opened new avenues for improving outcomes. A holistic, patient-centered approach that includes medical, therapeutic, and psychosocial support is essential for enhancing the CP patient’s functional abilities and quality of life. Continued collaboration among researchers, clinicians, families, and communities is crucial to bridging the gap between discovery and clinical application, offering renewed hope and opportunities for those affected by cerebral palsy.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
Sutha Ponnusamy: Conceptualization, supervision, manuscript review, and final approval of the article; Nithyapriya Karuppusamy: Literature search, data acquisition, and drafting of the manuscript; Sangameswaran Balakrishnan: Guidance, critical revision of intellectual content, and proofreading; Padmapriya. R: Literature review, compilation of research findings, and writing support; Palanibharathi. T: Data interpretation, figure/table preparation, and manuscript editing; Pavithra. M: Data organization, formatting, and assistance in manuscript preparation; Pradeepkumar. P: Reference collection, NLM formatting, and technical support; Madhivanan. K: Critical review, corrections, and coordination of the revised manuscript submission.
CONFLICT OF INTERESTS
Declared none
REFERENCES
Wimalasundera N, Stevenson VL. Cerebral palsy. Pract Neurol. 2016 Jun;16(3):184-94. doi: 10.1136/practneurol-2015-001184, PMID 26837375.
Krigger KW. Cerebral palsy: an overview. Am Fam Physician. 2006 Jan 1;73(1):91-100. PMID 16417071.
Vitrikas K, Dalton H, Breish D. Cerebral palsy: an overview. Am Fam Physician. 2020 Feb 15;101(4):213-20. PMID 32053326.
Reddihough DS, Collins KJ. The epidemiology and causes of cerebral palsy. Aust J Physiother. 2003;49(1):7-12. doi: 10.1016/s0004-9514(14)60183-5, PMID 12600249.
Paul S, Nahar A, Bhagawati M, Kunwar AJ. A review on recent advances of cerebral palsy. Oxid Med Cell Longev. 2022 Jul 30;2022:2622310. doi: 10.1155/2022/2622310, PMID 35941906, PMCID PMC9356840.
Centers for Disease Control and Prevention. Cerebral palsy facts. Atlanta: CDC; 2023. Available from: https://www.cdc.gov/cerebral‑palsy/index.html. [Last accessed on 21 Jul 2025].
Cerebral palsy. In: Definitions. Qeios; 2018. doi: 10.32388/671075.
Rosenbaum P, Paneth N, Leviton A. The definition and classification of cerebral palsy. Dev Med Child Neurol. 2007;49(s109):8-14. doi: 10.1111/j.1469‑8749.2007.tb12610.x.
Padmakar S, Kumar KS, Parveen S. Management and treatment for cerebral palsy in children. Indian J Pharm Pract. 2018;11(2):104–9. doi: 10.5530/ijopp.11.2.23.
Kumar S, Sharma R. Recent advancements in interventions for cerebral palsy: a review. J Neurorestoratol. 2023;11(3):100071. doi: 10.1016/j.jnrt.2023.100071.
Harvey A, Smith N, Smith M, Ostojic K, Berryman C. Chronic pain in children and young people with cerebral palsy: a narrative review of challenges advances and future directions. BMC Med. 2024 Jun 11;22(1):238. doi: 10.1186/s12916-024-03458-0, PMID 38862988, PMCID PMC11167894.
Calado CM, Manhaes De Castro R, Pereira SD, Da Silva Souza V, Visco DB, De Silveira BS. Therapeutic advances for treating memory impairments in perinatal brain injuries with implications for cerebral palsy: a systematic review and meta-analysis of preclinical studies. Exp Neurol. 2023 Jul;365:114411. doi: 10.1016/j.expneurol.2023.114411, PMID 37068620.
Makris T, Dorstyn D, Crettenden A. Quality of life in children and adolescents with cerebral palsy: a systematic review with meta-analysis. Disabil Rehabil. 2019;43(3):299-308. doi: 10.1080/09638288.2019.1623852, PMID 31180733.
Schmidt S, Markwart H, Rapp M, Guyard A, Arnaud C, Fauconnier J. Quality of life and mental health in emerging adults with cerebral palsy compared to the general population. Health Qual Life Outcomes. 2022 Apr 2;20(1):61. doi: 10.1186/s12955-022-01961-7, PMID 35366892, PMCID PMC8976958.
Gentile JP. Psychological health and quality of life in cerebral palsy. Theory Clin Pract Pediatr. 2022;4(1):102-5. doi: 10.25082/TCPP.2022.01.001.
Morgan C, Novak I, Badawi N. Enriched environments and motor outcomes in cerebral palsy. Dev Med Child Neurol. 2016;58(6):548-54. doi: 10.1111/dmcn.13105.
Physiopedia contributors. Cerebral Palsy Interventions; 2025. Available from: https://www.physio-pedia.com/cerebral_palsy_interventions. [Last accessed on 21 Jul 2025].
Wikipedia contributors. Selective dorsal rhizotomy. Wikipedia; 2025. Available from: https://en.wikipedia.org/wiki/Selective_dorsal_rhizotomy. [Last accessed on 21 Jul 2025].
Novak I, McIntyre S, Morgan C, Campbell L, Dark L, Morton N. A systematic review of interventions for children with cerebral palsy: state of the evidence. Dev Med Child Neurol. 2013 Oct;55(10):885-910. doi: 10.1111/dmcn.12246, PMID 23962350.
Feil Seifer D, Mataric MJ. A motor learning intervention using a social assistive robot for a child with cerebral palsy. Disabil Rehabil Assist Technol. 2020;15(2):180-8.
Jain M, Bansal V. Cerebral palsy: a neurological disorder-a review. Asian J Pharm Clin Res. 2018;11(2):17-21.
Cardone D, Perpetuini D, Di Nicola M, Merla A, Morone G, Ciancarelli I. Robot-assisted upper limb therapy for personalized rehabilitation in children with cerebral palsy: a systematic review. Front Neurol. 2025 Jan 6;15:1499249. doi: 10.3389/fneur.2024.1499249, PMID 39835154, PMCID PMC11743387.
Conner BC, Remec NM, Lerner ZF. Is robotic gait training effective for individuals with cerebral palsy? A systematic review and meta-analysis of randomized controlled trials. Clin Rehabil. 2022 Jul;36(7):873-82. doi: 10.1177/02692155221087084, PMID 35331027, PMCID PMC10035373.
National Institute for Health and Care Excellence (NICE). Cerebral palsy in under 25s: assessment and management. In: London: NICE; 2017. Available from: https://www.nice.org.uk/guidance/ng62. [Last accessed on 21 Jul 2025].
Novak I, McIntyre S, Morgan C, Campbell L, Dark L, Morton N. A systematic review of interventions for children with cerebral palsy: state of the evidence. Dev Med Child Neurol. 2013 Oct;55(10):885-910. doi: 10.1111/dmcn.12246, PMID 23962350.
Dursun E, Dursun N, Alican D. Ankle-foot orthoses: effect on gait in children with cerebral palsy. Disabil Rehabil. 2002 May 10;24(7):345-7. doi: 10.1080/0963820110090724, PMID 12022783.
Llamas Ramos R, Sanchez Gonzalez JL, Llamas Ramos I. Robotic systems for the physiotherapy treatment of children with cerebral palsy: a systematic review. Int J Environ Res Public Health. 2022 Apr 22;19(9):5116. doi: 10.3390/ijerph19095116, PMID 35564511, PMCID PMC9100658.