
Int J Pharm Pharm Sci, Vol 18, Issue 1, 7-13Review Article
SIRTUINS: POTENTIAL PHARMACOLOGICAL TARGET IN VARIOUS DISORDERS
REEMA MITRA*, PRATIBHA SHARMA
Chandigarh College of Pharmacy, CGC Landran, Mohali, Punjab-140307, India
*Corresponding author: Reema Mitra; *Email: reemamitra.ccp@cgc.edu.in
Received: 24 Jun 2025, Revised and Accepted: 13 Dec 2025
ABSTRAC
Sirtuins are a group of NAD+-dependent protein deacetylases that play an important role in cellular homeostasis, with wide-ranging effects on health and disease. Gene expression, metabolism, DNA repair, stress responses, and longevity are just a few of the biological processes that are affected by these evolutionarily conserved enzymes. Seven sirtuin subtypes (SIRT1–SIRT7) comprise the sirtuin family, and each has unique enzymatic properties and subcellular locations. Sirtuins are essential agents in chromatin remodelling, coordinating epigenetic changes that affect gene transcription and cellular destiny. Additionally, they play a crucial role in controlling pathways for nutrition sensing and energy homeostasis, which regulate the cellular metabolism. Notably, sirtuins participate in DNA repair and affect cell cycle progression, both of which are essential for maintaining genomic stability. This article offers a thorough overview of sirtuin biology, with an emphasis on their numerous roles, cellular localization, and consequences in various physiological and pathological circumstances.
Keywords: Cellular metabolism, Intracellular signalling peptides and proteins, Protein deacetylase, Sirtuins
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijpps.2026v18i1.55201 Journal homepage: https://innovareacademics.in/journals/index.php/ijpps
INTRODUCTION
A novel class of chromatin metabolic enzymes, termed sirtuins, was initially discovered in yeast S. cerevisiae. This group consists of seven members (Sirtuin 1-7), whose enzymatic activity depends upon nicotinamide adenine dinucleotide (NAD). Sirtuins facilitate the post-translational modification of numerous cellular proteins through deacetylation (sirtuins 1, 2, 3, and 5) and the transfer of adenosine diphosphate and ribose (ADP-ribose). Sirtuin activity has been associated with gene repression, metabolic regulation, stress responses, genome stability, apoptosis and cell survival, DNA repair, maturation, inflammation, neuroprotection, and healthy aging. While sirtuin modification may present therapeutic advantages for human diseases, the identification of biomolecules that modulate sirtuin activity is becoming increasingly crucial. Based on sequence-based genomic studies, sirtuins in animals can be categorized into four groups: SIRT1-SIRT3 belong to group I, SIRT 4 to group II, SIRT 5 to group III, and SIRT 6 and SIRT7 to group IV [1, 2]. A comprehensive literature search was conducted using databases such as PubMed, Scopus, and Web of Science using keywords such as ‘sirtuins, subcellular localization of sirtuins,’ ‘sirtuin activators, and ‘sirtuin inhibitors.
Subcellular localisation of sirtuin
Sirtuins exhibit distinct intracellular distributions in mammals. SIRT1 is predominantly localized in the nucleus, although it is also present in the cytoplasm. It plays a crucial role in regulating gene expression and is frequently associated with processes such as DNA repair, chromatin remodelling and transcriptional regulation. SIRT2 is primarily cytoplasmic but can translocate into the nucleus and is involved in a range of cellular activities, such as regulating the cell cycle, managing microtubule dynamics, and organizing the cytoskeleton. SIRT 3, 4, and 5 (SIRT3, SIRT4, and SIRT5) are predominantly located in the mitochondria, which are the energy-generating organelles of the cell. These sirtuins play crucial roles in regulating mitochondrial function, ensuring mitochondrial integrity, and minimizing oxidative stress. Sirtuins play an important role in various processes, such as inflammation, apoptosis, metabolism, and oxidative stress; therefore, they can be targets for disease conditions such as cancer, cardiovascular disease, and respiratory diseases [3]. SIRT6 is primarily nuclear and is involved in chromatin regulation, DNA repair, and maintenance of genomic stability. SIRT7 is localized within the nucleolus, particularly in the nucleolar regions associated with rRNA transcription. It is associated with ribosome biogenesis and plays a role in the regulation of ribosomal RNA (rRNA) transcription [4]. The following table (table 1) summarizes the sirtuin subtypes, their subcellular localization, and their respective physiological functions [5].
Physiological roles of sirtuins
SIRT1 is a crucial metabolic controller whose activity is influenced by dietary intake; it is upregulated throughout the body when food abstinence and calorie deficit are practised. In addition to encouraging the breakdown of cholesterol and lipids in the liver, skeletal muscles, and fatty tissues, SIRT1 upregulates mitochondrial biogenesis in several tissues, promotes gluconeogenic genes, represses glycolytic genes, and broadly activates fatty acid oxidation [5]. The major target of SIRT2 deacetylase in humans is α-tubulin, which is why mammalian SIRT2 is mostly a cytoplasmic catalyst that is closely associated with tubulins. Moreover, SIRT2 interacts with histone H4's Lys16 during the G2/M transition and deacetylates it, causing condensed chromatin formation. Furthermore, SIRT2 levels are diminished in gliomas, implying that SIRT2 removal may promote tumor growth and that SIRT2 replacement may function as a tumor-suppressing therapy [6]. The mitochondria contain SIRT3, SIRT4, and SIRT5, which are associated with the control of fundamental mitochondrial functions, such as adenosine triphosphate (ATP) generation, metabolism, mortality, and cellular signalling [7]. The role of SIRT3 in ameliorating depression has been studied by scientists, where it was concluded that exercise leads to an increase in SIRT3 levels and alleviates depression [8]. SIRT5 may contribute to the regulation of apoptosis and alter oxygenation. In mammalian cells, SIRT 6 integrates stress signalling to fuel the DNA repair process, considering oxidative damage, and plays a critical role in genome repair and preservation of genetic stability. SIRT6 is a desirable target for cancer therapy because it causes significant apoptosis in an assortment of disease cell lines but not in typical cells [9, 10]. SIRT7 interacts with several chromatin-remodelling complexes in the nucleolus and RNA and controls transcription, which is crucial for mammalian cell survival [11].
Sirtuin activators
Sirtuin activators enhance the activity of sirtuin enzymes, influencing various physiological processes, such as metabolism, aging, and age-related diseases. A docking study was conducted using the Indonesian herbal database, and many compounds, such as alpha-carotene, cassiamin C, casuarinin, and lutein, were found to be good SIRT1 activators [12]. The following is a list of compounds that have been reported to be sirtuin activators.
Table 1: Sirtuin subtypes, their subcellular localization, and respective physiological functions
| Sirtuin | Subcellular localization | Physiological functions |
| SIRT1 | Nucleus, Cytoplasm | Regulates transcription, DNA repair, metabolism, cell survival, aging |
| SIRT2 | Cytoplasm, Nucleus | Controls microtubule dynamics, cell cycle, oxidative stress response |
| SIRT3 | Mitochondria | Maintains mitochondrial integrity, regulates energy metabolism, ROS detoxification |
| SIRT4 | Mitochondria | Modulates insulin secretion, fatty acid oxidation, metabolic stress response |
| SIRT5 | Mitochondria | Regulates ammonia detoxification, urea cycle, lysine desuccinylation/demalonylation |
| SIRT6 | Nucleus | DNA repair, telomere maintenance, glucose and lipid metabolism, inflammation control |
| SIRT7 | Nucleolus | Ribosomal RNA transcription, chromatin regulation, stress response |
Resveratrol
Resveratrol (RSV), a polyphenol with two benzyl rings connected via a methylene linkage, was the initial substance shown to imitate calorie restriction (CR) by activating sirtuins. Resveratrol significantly reduced aging symptoms without changing the expression of any sirtuin genes and mimicked CR-induced gene expression patterns in numerous organs. Red wine and other grape products contain resveratrol (RSV), a non-flavonoid polyphenol which prevents the development of malignancy. In addition, RSV possesses anticancer, anti-neurodegenerative, and anti-irritation properties [13]. In vivo studies of RSV have shown improved general health in infected patients. Researchers have explored the role of resveratrol in aging and concluded that resveratrol can play a beneficial role in aging by modulating the sirtuin pathway [14, 15]. Age-related symptoms such as decreased proteinuria and cataract development, improved arterial flexibility, reduced vascular endothelium inflammation and apoptosis, improved motor coordination, and constant mineral density of bones were significantly improved in mice. Owing to RSV's low bioavailability, repackaged varieties with increased bioavailability have been developed. The dietary supplement resVida, which includes 150 mg of resveratrol daily, also produced excellent results in terms of lowering intrahepatic lipid content, blood glucose levels, circulating fatty acids, alanine aminotransferase levels, and inflammatory markers in healthy obese men. Sirtuins enhance the phosphorylation of MAPKs (mitogen-activated protein kinases), AKT, AMPK (5' AMP-activated protein kinase), RhoA (Ras homolog family member A), and BDNF (Brain-Derived Neurotropic Factor) while simultaneously decreasing the activation of the NLRP3 (NLR family pyrin domain containing 3) inflammasome, NF-κB, and STAT3 (Signal Transducer and Activator of Transcription 3). Additionally, they facilitated the SIRT1/SREBP1c pathway, lowered β-amyloid levels via the SIRT1-NF-κB-BACE1 signalling pathway, and alleviated mitochondrial damage by deacetylating PGC-1α [16, 17].
Quercetin
Fruits, vegetables, and nuts contain quercetin (Que; 3,3′,4′,5,7-pentahydroxyflavone), a type of flavonoid polyphenol, which has a characteristic nutritive enhancement that is protected and has mitigating and cell reinforcement qualities. Quercetin is present in organic products, seeds, vegetables, bracken greenery, coffee, tea, and a few other plants, as well as in natural dyes [18]. Quercetin binds to sirtuins (mainly SIRT1 and SIRT6) and increases their deacetylase activity. It plays an important role in inflammation, oxidative stress, mitochondrial health and autophagy by modulating the SIRT1/NF-κB, SIRT1/Nrf2/HO-1, mitochondrial biogenesis and SIRT1/FoxO pathways, respectively [19]. It has several properties that could make it a successful enemy of disease. Quercetin has been shown to play a significant role in the concealment of cancer cells in the breast, colon, prostate, ovary, endometrium, and lungs. Quercetin, which respond to the radicals produced, can prevent the negative effects of lipid peroxidation on the body, such as cardiovascular and neurological diseases [20, 21].
Berberine
Berberine is a yellow alkaloid found in many plants. It interacts mainly with SIRT1 by activating the AMPK pathway which influences lipid breakdown, lowers insulin resistance and decreases oxidative stress in liver, pancreas or heart diseases. Berberine activates SIRT3 in adipose tissue and improves metabolism. It helps in combination of SIRT1 with TFEB (a transcription factor) and induces autophagy. Berberine (BBR) has been linked to analgesic, antitumor, anti-aging, and cardiac preventive effects. It has antioxidative protective properties under various pathological and physiological circumstances [22, 23].
Fisetin
The natural polyphenol fisetin (3,3′,4,7-tetra-hydroxyflavone), is a dietary flavonoid found in apples, persimmons, grapes, onions, kiwi, kale, and strawberries[24]. It activates SIRT1 and 6 and modulates SIRT3. Fisetin has an inhibitory effect on SIRT 2. Activation of SIRT 1 has an anti-inflammatory effect and regulates differentiation of fat cells. SIRT 6 activation helps in inhibiting senescence. The modulation of SIRT3 leads to mitophagy and helps in reduction of oxidative stress. SIRT2 inhibition lead to anticancer potential of Fisetin, especially in cancers of the pancreas [25, 26]. Diabetic animals treated with fisetin showed increased levels of plasma insulin, C peptide, and haemoglobin. These results confirm that fisetin plays a significant role in maintaining glucose homeostasis. Fisetin was found to have antibacterial properties in several tests [27, 28].
Curcumin
Curcumin which is an active compound found in turmeric, can activate and increase the expression of SIRT1, leading to activation of AMPK pathway. Curcumin due its action on SIRT1 has an anti-inflammatory effect and has the ability to decrease age-related stress. Increase in SIRT1 activity has a neuroprotective effect and prevents against Alzheimer's and Parkinson's disease. Through the activation of SIRT, curcumin can affect apoptosis, mitochondrial biogenesis, and malfunction. Diabetes, ischemia/reperfusion injury, cardiac fibrosis, and other disorders can be prevented or treated by curcumin-induced SIRT1 overexpression [29, 30].
Sirtuin inhibitors
Sirtuin inhibitors suppress or reduce the activity of sirtuin enzymes. They are being explored for their potential therapeutic applications in specific contexts, such as cancer treatment [31, 32].
Splitomicin and its derivatives
Splitomicin and its analogues have been studied for cell-based SIRT2 inhibition, and the most effective analogues had hydrolysis rates comparable to that of splitomicin in an in vitro SIRT2 inhibition assay. Splitomicin does not effectively inhibit human sirtuins; therefore, several α-phenyl splitomicin analogues have been developed. The direction of the-phenyl bunch is essential for the role of these chemicals in SIRT2 movement, and a connection between higher catalyst hindrance and anticancer action has been established. These compounds may be helpful in the treatment of cancer and other conditions by activating suppressive properties, inducing cell death, and inhibiting the activity of transcriptional repressors of oncogenes [33, 34].
Sirtinol
Sirtinol is a 2-[(2-hydroxy-naphthalen-1-yl) methylene-amino] compound. In vitro hindrance of yeast SIRT2 and human SIRT2 action by N-(1-phenyl-ethyl)-benzamide shows that this compound directly reduces enzymatic activity and serves as a generic inhibitor of sirtuins. It has been shown that drugs with the 2-hydroxyl-1-napthol moiety belong to another class of sirtuin inhibitors and are effective for restraint. A different class of sirtuin inhibitors, comprising combinations including the 2-hydroxyl-1-napthol moiety grouping, has been shown to be effective at inhibiting sirtuins. Two further analogues have been made: [(2-hydroxy-1-naphthalenylmethylene) amino]-3 and 4-1 phenylethylbenzamide-N-(i. e., m-and p-sirtinol) [35].
AGK2
AGK2 is a chemical compound that acts as a sirtuin inhibitor, specifically targeting SIRT2. AGK2 can alter the deacetylation of SIRT2 target proteins and impact various cellular processes associated with sirtuins. Inhibiting SIRT2 can affect various cellular processes, such as the cell cycle, microtubule dynamics, and cellular stress response, and may have implications for cell proliferation, differentiation, and survival [32].
Cambinol
Cambinol is a chemically stable drug that reduces SIRT1 and SIRT2 levels in vitro. It possesses a sirtinol and splitomicin β-naphtholpharmacophore. SIRT5 was only slightly inhibited by cambinol (42% inhibition at 300 µM). Cambinol decreased the growth of Burkitt lymphoma xenografts in mice and induced apoptosis by hyperacetylating the oncoproteins BCL6 and p53. The phenyl ring of cambinol was modified to enhance the activity and selectivity for SIRT1, and several SIRT2-selective analogues have been discovered. Cambinol has also been explored for its anticancer activity. In certain cancer types, SIRT1 and SIRT2 activities can promote tumor growth and survival. Inhibiting these sirtuins with cambinol may disrupt cancer growth [36].
Suramin
The inhibitor suramin is more powerful than cannabinol in SIRT5 NAD-subordinate deacetylase movement. Originally used to treat trypanosomiasis, suramin is a polyhydrous naphthyl urea that also has antiproliferative and antiviral properties [37].
Protective effects of sirtuin
Sirtuins play an important role in disease pathways. The fig. below illustrates the role of various sirtuins in disease pathways [38].
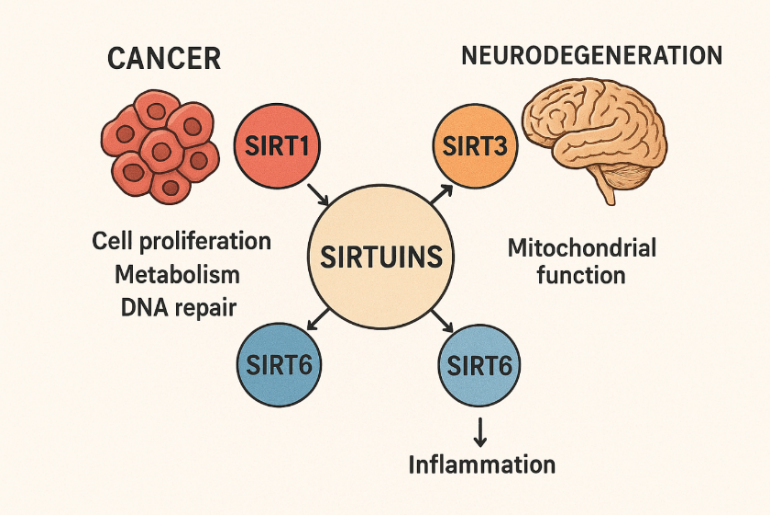
Fig. 1: Roles of different sirtuin subtypes in various diseases
The renal defensive impacts of sirtuin
Acute kidney injury
Different models of intense kidney injury have been used to show the renal protective effects of sirtuins. According to research, SIRT1 protects the kidneys by deacetylating p53 and promoting catalase activity, which prevents apoptosis and oxidative damage, respectively. By deacetylating and activating peroxisome proliferator-initiated receptor gamma coactivator 1-alpha, SRT1720, a specific SIRT1 activator, promotes the biogenesis of mitochondria, mitochondrial proteins, and mitochondrial respiration. Thus, SRT1720 re-establishes separated and enraptured tubule epithelium, decreasing reoxygenation injury. These capabilities are related to the assurance of intense tissue or cell affronts through powerful energy use. By deacetylating nuclear factor-(NF-) p65 and mitogen-activated protein kinase phosphatase-1, SIRT2 further protects the kidney in a lipopolysaccharide-activated severe renal damage model [39].
Cyst formation
SIRT1 has recently been associated with renal cyst formation. Knockout mice SIRT1 and the polycystic kidney disease gene Pkd1 developed cysts less frequently than the other mice. Pkd1 knockout mice were administered a SIRT1 inhibitor, which reduced the rate of cyst formation. Cilia development observed in Pkd1 knockdown was inhibited by SIRT2 overexpression in renal epithelial cells [40, 41].
Diabetic nephropathy
Sirtuin 1 has a role in diabetes [42]. Sirtuins have been investigated for their functions in the initiation and progression of diabetic nephropathy. Through deacetylating and inactivating Nuclear Factor kappa-light-chain-enhancer of activated B cells (NFκB) in mesangial cells, 3,5-diiodothyronine activates SIRT1. This inhibits the phosphorylation of the Jun proto-oncogene (c-JUN), expansion of mesangial cells, and production of urinary proteins. SIRT1 inactivates signal transducer and activator of transcription 3 (STAT3) and NF-’s p65 subunit, which suppresses podocyte destruction and visceral epithelial cell dysfunction in diabetic nephropathy and increases urine protein levels in diabetic nephropathy. As a result, podocyte apoptosis and urinary protein levels are reduced [43].
Neuroprotective impacts of sirtuins
Acute disorder
Cerebral ischemia
According to Raval et al., SIRT1 has a neuroprotective effect. According to their findings, ischemic preconditioning and resveratrol therapy both lessen the neuronal damage caused by N-methyl-D-aspartate (NMDA) challenge to hippocampal CA1 neurons in slices, as well as overall cerebral ischemia in rats. Additionally, they demonstrated that resveratrol and preconditioning have protective effects by enhancing SIRT1 activity. Two recent examinations have shown that sirtuin 1 actuation increases blood supply to the brain and that one potential pathway is the negative impact of the mitochondrial unfasten protein 2, further prove SIRT1's neuroprotective job. In addition to providing direct safety, SIRT1 also works synergistically with other neuroprotective substances, including tetrahydroxy stilbene glucoside and leptinicariin [44].
Depression
Scientists have reported the protective effects of increased SIRT3 expression in depression. They found that swimming increased the expression of SIRT3 and alleviated depression caused by mild stress [45].
Retinal injury
The sensory system's retina is vulnerable to various injuries from UV rays, blood shortages, irritation, and retinal degeneration. SIRT1 is present throughout the normal retinal layers and is essential for retinal health. However, the retinas of SIRT1-deficient animals exhibit disordered cellular patterns, lower cell counts in many layers, and hyperacetylation of p53. SIRT1 has also been linked to neuroprotective functions against retinal damage. For instance, resveratrol upregulates SIRT1, which protects the retina against antibody-induced apoptosis. SIRT1 is absent in retinal degeneration 10 (rd10) mice, an animal model of retinal degeneration, and this absence of SIRT1 causes retinal degeneration. Similarly, the absence of E2Fs, a transcription factor for SIRT1, causes SIRT1 to be downregulated, p53 to become hyperacetylated, and retinal apoptosis to increase. Taken together, these findings show that SIRT1 is helpful for retinal health and maintenance [46].
Neurodegenerative diseases
Alzheimer’s disease
Sirtuins are evolutionarily conserved, NAD⁺-dependent enzymes known for their roles in protecting against age-related disorders. In Alzheimer's disease (AD), various mouse and cell culture models have demonstrated that elevating SIRT1 levels can counteract AD-related pathology. SIRT1 is also a key mediator of the physiological benefits associated with calorie restriction (CR); mice lacking SIRT1 fail to respond normally to CR, while transgenic mice overexpressing SIRT1 show CR-like adaptations. This establishes a strong link between CR and SIRT1 activity. Early evidence of CR’s influence on AD came from findings that calorie-restricted AD mice had fewer amyloid-β plaques. In AD and Amyotrophic Lateral Sclerosis (ALS) mouse models, SIRT1 levels were found to increase, and in cell-based models of both diseases, resveratrol and SIRT1 activation improved neuronal survival [47, 48].
Parkinson's disease (PD)
Is characterized by the loss of dopaminergic neurons in the substantia nigra, which is defined by bradykinesia, postural instability, muscle rigidity, and resting tremor. All organs, including the brain, express SIRT1, particularly in neurons. SIRT1 may be protective against PD. SIRT1 could, on the one hand, avoid α-synuclein aggregation in PD animal and cell models by activating molecular chaperones. To prevent PD pathogenesis, SIRT1 deacetylase activity facilitates α-synuclein elimination via light chain 3 (LC3)-mediated autophagy. Additionally, extracellular α-syn accumulation results in mitochondrial dysfunction and a decrease in SIRT1 production. Resveratrol and another sirtuin-activating compound, quercetin, prevented the loss of dopaminergic neurons caused by the dopaminergic neurotoxin 1-methyl-4-phenyl pyridinium (MPP+), which affects dopaminergic neurons, according to studies on the neuroprotective effects of resveratrol on dopaminergic neurons in organotypic midbrain cut cultures. Whether SIRT2 plays a role in the etiology of neurodegenerative disorders is debatable. Expanded deacetylation of tubulin, which decreases microtubule integrity in neurons, may be the cause of SIRT2 overexpression, promoting neurodegeneration. Repressing SIRT2 resulted in less development of polyglutamine at the N-end of neurons in a mouse model of Huntington's disease. In addition, SIRT2 inhibition increased neuronal cell survival, but SIRT2 expression increased α-synuclein toxicity in PD models. Additionally, SIRT2 expression can lessen brain cell death in animal models after MPP+ treatment. Hence, depending on the circumstances, the role of SIRT2 may change. SIRT2 increases α-syn toxicity and aggregation [49, 50].
Huntington's disease
Huntington’s disease (HD) is an autosomal dominant neurological condition that often affects people in their midlife. Slow-progressive HD is characterized by personality changes as a defining feature, neurological impairment, unusual motor motions, and ultimately patient death. Sirtuin deacetylases have been investigated as potential therapeutic targets for slowing HD progression in human-like cell culture and animal models. The experiments of Parker and colleagues, which revealed that overexpression of Sir1 or therapy with resveratrol reverses Caeno rhabditiselegans' mutant polyglutamine-induced neuronal dysfunction phenotypes, were the first to link SIRT1 to HD. Pre-treatment with resveratrol helped rats recover from the cognitive and motor abnormalities induced by 3-nitropropionic acid in a chemically induced HD model. Nicotinamide, a pharmacological inhibitor that may affect several sirtuin proteins, was administered to these mice. This therapy reduced the motor impairments associated with HD, but had no effect on mice huntingtin aggregation or weight loss. These studies highlight the potential of nicotinamide and resveratrol as potent medicinal agents [51, 52].
Wallerian degeneration
Axonal degradation is an active mechanism in neurodegenerative diseases and peripheral neuropathies. Anterograde degeneration of transected axons is noticeably delayed in a mutant mouse strain called Wallerian degeneration slow (Wld). After axon transection, neurons pretreated with resveratrol or NAD showed less axonal breakdown. Additionally, sirtinol therapy and SIRT1knockdownprevented NAD-dependent axonal protection. Therefore, SIRT1 was suggested as the downstream effector of Nmnat/NAD axonal protective activity. Furthermore, the protective effects of NAD were unaffected by the SIRT1 inhibitor sirtinol or resveratrol in the same experiment [53, 54].
Regulation of cell proliferation, stress resistance, and cancer by sirtuins
Numerous components, such as p53, which control cell proliferation and apoptosis, have been discovered to be sirtuin substrates. SIRT1 may have both tumour-promoting and tumour-suppressive functions in cancer. The dual nature of SIRT1’s role in cancer is influenced by tissue-specific factors and the genetic background of the tumour. As a tumour promoter, SIRT1 can increase cell survival, inhibit apoptosis, and promote cancer cell proliferation and metastasis. In contrast, SIRT1 activation inhibits tumour growth and induces apoptosis. This dual nature of SIRT1 in cancer requires careful consideration. The effect of SIRT1 in cancer depends on the tissue type, stage of cancer progression, and genetic background. It has been suggested that SIRT1 and the tumour-suppressing protein p53 are related. Stopping the cell cycle and cell death are brought on by the hyperacetylation of p53, which stabilizes and activates it due to its many acetylation sites. In contrast, it is predicted that SIRT1's deacetylation of p53 will cause its destruction via an MDM2-dependent ubiquitin-mediated mechanism. Overexpression of the dominant-negative SIRT1 protein reduces p53 transcriptional activity and p53-dependent apoptosis. Except for SIRT5, all mammalian sirtuins have been implicated in carcinogenesis. A key regulator of cancer cell proliferation, DNA restoration, and survivability under various stresses has been identified as SIRT1 [55, 56].
Breast cancer
SIRT1 exhibits both pro-tumorigenic and antitumorigenic effects in breast cancer. SIRT1can facilitate DNA repair and suppress inflammation, which may confer tumor-suppressive properties to it. Nonetheless, SIRT1 can enhance cell survival pathways, potentially contributing to tumor proliferation. It achieves DNA repair by deacetylating essential DNA repair proteins, thereby preserving genomic stability and mitigating the risk of tumorigenesis. Furthermore, SIRT1 can attenuate inflammation by deacetylating and inactivating pro-inflammatory transcription factors such as NF-κB. This anti-inflammatory mechanism may play a role in inhibiting the initiation and progression of breast cancer, while it has also been linked to tumour promotion. It can promote cell survival pathways, potentially supporting cancer cell growth and survival. Additionally, in certain breast cancer subtypes, it enhances oestrogen receptor signalling, which may contribute to tumour progression [57, 58].
Lung cancer
Colon cancer
c-MYC is associated with increased SIRT1 protein levels in colorectal cancer. A positive feedback regulatory loop is created by c-MYC, nicotinamide phosphoribosyl transferase, breast cancer protein c-MYC deletion, and SIRT1. Increased expression of SIRT1 and c-MYC is strongly associated with higher levels of malignancy in colorectal serrated lesions. SIRT 6 can also inhibit colorectal cancer cell growth by regulating glucose metabolism and suppressing inflammation [59, 60].
Thyroid cancer
SIRT1 has been suggested to have tumour-suppressive effects in some studies on thyroid cancer. It can promote DNA repair mechanisms, which helps maintain genomic stability and reduce the risk of cancer development. SIRT1 overexpression and c-MYC protein levels are positively associated with human thyroid tumours. SIRT1 may promote thyroid cell differentiation, which can be disrupted in thyroid cancer cells. Enhanced differentiation may slow down tumour growth. Transgenic SIRT1 expression promotes phosphatase and tensin homolog-induced thyroid carcinogenesis in mice SIRT1 boosts cellular MYC (c-MYC) transcription and stabilizes this protein in thyroid tumours. SIRT 6 can also act as a tumour suppressor in thyroid cancer by promoting DNA repair mechanisms, inhibiting inflammation, and regulating glucose metabolism, all of which may contribute to slowing tumour growth [61].
Pancreatic cancer
Both mRNA and protein upregulation of SIRT1 were observed in pancreatic cancer tissues. In pancreatic cancer cells, apoptosis and senescence are induced by SIRT1 knockdown, which also inhibits invasion and increases chemosensitivity. By deacetylating pancreatic transcription factor-1 and β-catenin, SIRT1 controls acinar-to-ductal metaplasia in pancreatic cancer and promotes cancer cell survival. SIRT3 also exerts tumour-suppressive effects in pancreatic cancer. It can promote mitochondrial function and reduce oxidative stress, which may slow tumour growth. SIRT6 also has a tumour-suppressive role by inhibiting glycolysis, promoting DNA repair mechanisms, and regulating inflammation [61].
Malignancies of the ovary and cervical
SIRT1 is frequently overexpressed in ovarian cancer. It facilitates cancer cell proliferation and inhibits cancer cell apoptosis. SIRT1 stops the transcriptional factor FOXL2 from activating on locations necessary for the cell cycle and DNA repair in granulosa cells. In contrast, nicotinamide's suppression of SIRT1 reduces granulosa cell proliferation by increasing FOXL2 expression. Human papillomavirus E7, an oncogenic viral protein that up regulates SIRT1, can control the pro-survival effect of the virus by inhibiting p53 function in human SiHa cervical cancer cells. SIRT3 may function as a tumour suppressor in ovarian cancer by modulating mitochondrial function and mitigating oxidative stress [62].
Lymphoma and leukaemia
SIRT1 is associated with both pro-tumorigenic and anti-tumorigenic functions in various leukaemia subtypes. It may enhance cell survival pathways and inhibit apoptosis in certain contexts, thereby potentially facilitating leukaemia cell proliferation. SIRT1 is overexpressed in adult T-cell leukaemia cells, and sirtinol inhibition leads to apoptosis. In 104 patients with diffuse large B-cell lymphoma, 74% (77/104) exhibited positive SIRT1 protein expression, which was strongly associated with worse overall survival. SIRT1 expression was also excessive (more than 2-fold). Increased SIRT1 expression appears to be essential for cell survival. Burkitt lymphoma cells undergo apoptosis when cambinol inhibits SIRT1/2. SIRT2 has role in both tumour-promotion and tumour-suppression roles. Some studies suggest that SIRT3 may have tumour-suppressive effects in certain types of leukaemia by regulating mitochondrial function and oxidative stress. SIRT6 can promote DNA repair, inhibit inflammation, and regulate cellular metabolism, all of which may contribute to slowing tumor growth [63].
CONCLUSION
This review highlights the diverse functions of sirtuins, which are crucial enzymes. Although sirtuins show therapeutic potential, their context-dependent roles and limited clinical data necessitate further mechanistic and translational research. Altering sirtuin activity may influence various pathological changes in different diseases. Sirtuins belong to a family of well-preserved enzymes that are vital for numerous cellular activities, such as metabolism, stress response, and aging. These NAD+-dependent deacetylases have attracted considerable interest recently because of their wide-ranging roles and potential therapeutic uses. Sirtuins are key regulators of gene expression, DNA repair, and cellular balance, making them indispensable for sustaining overall cellular health and longevity. Their significance extends beyond basic cellular functions, as they are linked to several pathological conditions, including cardiovascular disease, neurodegenerative disorders, and cancer. Studies have indicated that adjusting sirtuin activity may alleviate or even reverse some of the harmful effects associated with these diseases. For example, activating specific sirtuins has been shown to enhance insulin sensitivity in diabetes, decrease inflammation in cardiovascular conditions, and boost neuroprotection in neurodegenerative diseases. Consequently, targeting sirtuins through pharmacological means or lifestyle changes has emerged as a promising strategy for developing new therapeutic approaches to address a broad spectrum of age-related and metabolic disorders.
ACKNOWLEDGEMENT
We thank the Chandigarh Group of Colleges and Chandigarh College of Pharmacy for providing the necessary facilities for this study.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
Reema Mitra: Conceptualization, Resources, Writing-review, and editing, Supervision, Pratibha Sharma: Resources, Writing Original draft.
CONFLICTS OF INTERESTS
The authors declare no conflicts of interest.
REFERENCES
Carafa V, Rotili D, Forgione M, Cuomo F, Serretiello E, Hailu GS. Sirtuin functions and modulation: from chemistry to the clinic. Clin Epigenet. 2016;8:61. doi: 10.1186/s13148-016-0224-3, PMID 27226812.
Carafa V, Nebbioso A, Altucci L. Sirtuins and disease: the road ahead. Front Pharmacol. 2012;3:4. doi: 10.3389/fphar.2012.00004, PMID 22319497.
Wu QJ, Zhang TN, Chen HH, Yu XF, LV JL, Liu YY. The sirtuin family in health and diseases. Signal Transduct Target Ther. 2022;7(1):402. doi: 10.1038/s41392-022-01257-8, PMID 36581622.
Feldman JL, Dittenhafer Reed KE, Denu JM. Sirtuin catalysis and regulation. J Biol Chem. 2012;287(51):42419-27. doi: 10.1074/jbc.R112.378877, PMID 23086947.
Wu QJ, Zhang TN, Chen HH, Yu XF, Lv JL, Liu YY. The sirtuin family in health and disease. Signal Transduct Target Ther. 2022;7(1):402. doi: 10.1038/s41392-022-01257-8, PMID 36581622.
Gomes P, Fleming Outeiro T, Cavadas C. Emerging role of sirtuin 2 in the regulation of mammalian metabolism. Trends Pharmacol Sci. 2015;36(11):756-68. doi: 10.1016/j.tips.2015.08.001, PMID 26538315.
Chand J, Kandy AT, Prasad K, Mathew J, Sherin F, Subramanian G. In silico preparation and in vitro studies of benzylidene-based hydroxybenzyl urea derivatives as free radical scavengers in Parkinson’s disease. Int J App Pharm. 2024;16(3):217-24. doi: 10.22159/ijap.2024v16i3.50628.
Sowndarya R, Doss VA. Evaluation of sirtuin 3 biomarker before and after exercise regimen in chronic unpredictable mild stress-induced depressed rats. Asian J Pharm Clin Res. 2019;12(1):180-4. doi: 10.22159/ajpcr.2018.v12i1.28669.
Beauharnois JM, Bolivar BE, Welch JT. Sirtuin 6: a review of biological effects and potential therapeutic properties. Mol Bio Syst. 2013;9(7):1789-806. doi: 10.1039/C3MB00001J, PMID 23592245.
Klein MA, Denu JM. Biological and catalytic functions of sirtuin 6 as targets for small-molecule modulators. J Biol Chem. 2020;295(32):11021-41. doi: 10.1074/jbc.REV120.011438, PMID 32518153.
Li XT, Zhang YP, Zhang MW, Zhang ZZ, Zhong JC. Sirtuin 7 serves as a promising therapeutic target for cardiorenal diseases. Eur J Pharmacol. 2022;925:174977. doi: 10.1016/j.ejphar.2022.174977, PMID 35513019.
Villalba JM, Alcain FJ. Sirtuin activators and inhibitors. BioFactors. 2012;38(5):349-59. doi: 10.1002/biof.1032, PMID 22730114.
Aldisa O, Azminah A, Erlina L, Hayun H, Yanuar A. Virtual screening of Indonesian herbal database to find sirtuin 1 activators using the docking method. Asian J Pharm Clin Res. 2017;10(17):158-62. doi: 10.22159/ajpcr.2017.v10s5.23121.
Fremont L. Biological effects of resveratrol. Life Sci. 2000;66(8):663-73. doi: 10.1016/S0024-3205(99)00410-5, PMID 10680575.
Bhat KP, Kosmeder JW, Pezzuto JM. Biological effects of resveratrol. Antioxid Redox Signal. 2001;3(6):1041-64. doi: 10.1089/152308601317203567, PMID 11813979.
Walle T. Bioavailability of resveratrol. Ann NY Acad Sci. 2011;1215(1):9-15. doi: 10.1111/j.1749-6632.2010.05842.x, PMID 21261636.
Szkudelska K, Szkudelski T. Resveratrol obesity and diabetes. Eur J Pharmacol. 2010;635(1-3):1-8. doi: 10.1016/j.ejphar.2010.02.054, PMID 20303945.
Kelly GS. Quercetin. Monograph. Altern Med Rev. 2011;16(2):172-94. PMID 21649459.
Ungurianu A, Zanfirescu A, Margina D. Exploring the therapeutic potential of quercetin: a focus on its sirtuin-mediated benefits. Phytother Res. 2024;38(5):2361-87. doi: 10.1002/ptr.8168, PMID 38429891.
Zhang M, Swarts SG, Yin L, Liu C, Tian Y, Cao Y. Antioxidant properties of quercetin. In: LaManna JC, Puchowicz MA, Xu K, Harrison DK, Bruley DF, editors. Adv Exp Med Biol. Vol 701. Boston, MA: Springer; 2011. p. 283–289. doi: 10.1007/978-1-4419-7756-4_38, PMID 21445799.
Lakhanpal P, Rai DK. Quercetin: a versatile flavonoid. Internet Journal of Medical Update EJOURNAL. 2007;2(2):22-37. doi: 10.4314/ijmu.v2i2.39851.
Shan Y, Zhang S, Gao B, Liang S, Zhang H, Yu X. Adipose tissue SIRT1 regulates insulin sensitizing and anti-inflammatory effects of berberine. Front Pharmacol. 2020;11:591227. doi: 10.3389/fphar.2020.591227, PMID 33390968.
Li D, Yang C, Zhu JZ, Lopez E, Zhang T, Tong Q. Berberine remodels adipose tissue to attenuate metabolic disorders by activating sirtuin 3. Acta Pharmacol Sin. 2022 May;43(5):1285-98. doi: 10.1038/s41401-021-00736-y, PMID 34417576.
Grynkiewicz G, Demchuk OM. New perspectives for fisetin. Front Chem. 2019;7(7):697. doi: 10.3389/fchem.2019.00697, PMID 31750288.
Kim SC, Kim YH, Son SW, Moon EY, Pyo S, Um SH. Fisetin induces Sirt1 expression while inhibiting early adipogenesis in 3T3-L1 cells. Biochem Biophys Res Commun. 2015;467(4):638-44. doi: 10.1016/j.bbrc.2015.10.094, PMID 26499075.
Wang X, Li X, Zhou J, Lei Z, Yang X. Fisetin suppresses chondrocyte senescence and attenuates osteoarthritis progression by targeting sirtuin 6. Chem Biol Interact. 2024 Feb 25;390:110890. doi: 10.1016/j.cbi.2024.110890, PMID 38278314.
Maher P. How fisetin reduces the impact of age and disease on CNS function. Front Biosci (Schol Ed). 2015;7(1):58-82. doi: 10.2741/s425, PMID 25961687.
Prasath GS, Pillai SI, Subramanian SP. Fisetin improves glucose homeostasis through the inhibition of gluconeogenic enzymes in hepatic tissues of streptozotocin-induced diabetic rats. Eur J Pharmacol. 2014;740:248-54. doi: 10.1016/j.ejphar.2014.06.065, PMID 25064342.
Zendedel E, Butler AE, Atkin SL, Sahebkar A. Impact of curcumin on sirtuins: a review. J Cell Biochem. 2018;119(12):10291-300. doi: 10.1002/jcb.27371, PMID 30145851.
Grabowska W, Suszek M, Wnuk M, Lewinska A, Wasiak E, Sikora E. Curcumin elevates sirtuin level but does not postpone in vitro senescence of human cells building the vasculature. Oncotarget. 2016;7(15):19201-13. doi: 10.18632/oncotarget.8450, PMID 27034011.
Rajasekaran A, Sivagnanam G, Xavier R. Nutraceuticals as therapeutic agents: a review. RJPT. 2008;1(4):171-4. doi: 10.5958/0974-360X.
Alcain FJ, Villalba JM. Sirtuin inhibitors. Expert Opin Ther Pat. 2009;19(3):283-94. doi: 10.1517/13543770902755111, PMID 19441904.
Posakony J, Hirao M, Stevens S, Simon JA, Bedalov A. Inhibitors of Sir2: evaluation of splitomicin analogues. J Med Chem. 2004;47(10):2635-44. doi: 10.1021/jm030473r, PMID 15115404.
Neugebauer RC, Uchiechowska U, Meier R, Hruby H, Valkov V, Verdin E. Structure activity studies on splitomicin derivatives as sirtuin inhibitors and computational prediction of binding mode. J Med Chem. 2008;51(5):1203-13. doi: 10.1021/jm700972e, PMID 18269226.
Tsai YF, Yu HP, Chang WY, Liu FC, Huang ZC, Hwang TL. Sirtinol inhibits neutrophil elastase activity and attenuates lipopolysaccharide-mediated acute lung injury in mice. Sci Rep. 2015;5(1):8347. doi: 10.1038/srep08347, PMID 25666548.
Medda F, Russell RJ, Higgins M, McCarthy AR, Campbell J, Slawin AM. Novel cambinol analogs as sirtuin inhibitors: synthesis, biological evaluation and rationalization of activity. J Med Chem. 2009;52(9):2673-82. doi: 10.1021/jm8014298, PMID 19419202.
Trapp J, Meier R, Hongwiset D, Kassack MU, Sippl W, Jung M. Structure–activity studies on suramin analogs as inhibitors of NAD+-dependent histone deacetylases (sirtuins). ChemMedChem. 2007;2(10):1419-31. doi: 10.1002/cmdc.200700003, PMID 17628866.
Carafa V, Nebbioso A, Altucci L. Sirtuins and disease: the road ahead. Front Pharmacol. 2012 Jan 31;3:4. doi: 10.3389/fphar.2012.00004, PMID 22319497.
Funk JA, Schnellmann RG. Accelerated recovery of renal mitochondrial and tubule homeostasis with SIRT1/PGC-1α activation following ischemia–reperfusion injury. Toxicol Appl Pharmacol. 2013;273(2):345-54. doi: 10.1016/j.taap.2013.09.026, PMID 24096033.
Zhou X, Fan LX, Sweeney WE, Denu JM, Avner ED, Li X. Sirtuin 1 inhibition delays cyst formation in autosomal-dominant polycystic kidney disease. J Clin Invest. 2013;123(7):3084-98. doi: 10.1172/JCI64401, PMID 23778143.
Zhou X, Fan LX, Li K, Ramchandran R, Calvet JP, Li X. SIRT2 regulates ciliogenesis and contributes to abnormal centrosome amplification caused by loss of polycystin-1. Hum Mol Genet. 2014;23(6):1644-55. doi: 10.1093/hmg/ddt556, PMID 24203696.
Liu R, Zhong Y, Li X, Chen H, Jim B, Zhou MM. Role of transcription factor acetylation in diabetic kidney disease. Diabetes. 2014;63(7):2440-53. doi: 10.2337/db13-1810, PMID 24608443.
Ashok P, Narenderan ST, Meyyanathan SN, Babu B, Vadivelan R. Development and validation of a RP-HPLC method for estimation of telmisartan in human plasma. Int J App Pharm. 2019;11(1):237. doi: 10.22159/ijap.2019v11i1.30513.
Raval AP, Dave KR, Perez Pinzon MA. Resveratrol mimics ischemic preconditioning in the brain. J Cereb Blood Flow Metab. 2006;26(9):1141-7. doi: 10.1038/sj.jcbfm.9600262, PMID 16395277.
Raval AP, Lin HW, Dave KR, De Fazio RA, Della Morte DD, Kim EJ. Resveratrol and ischemic preconditioning in the brain. Curr Med Chem. 2008;15(15):1545-51. doi: 10.2174/092986708784638861, PMID 18537630.
Shindler KS, Ventura E, Rex TS, Elliott P, Rostami A. SIRT1 activation confers neuroprotection in experimental optic neuritis. Invest Ophthalmol Vis Sci. 2007;48(8):3602-9. doi: 10.1167/iovs.07-0131, PMID 17652729.
Qin W, Chachich M, Lane M, Roth G, Bryant M, De Cabo R. Calorie restriction attenuates Alzheimer’s disease type brain amyloidosis in Squirrel monkeys (Saimiri sciureus). JAD. 2006;10(4):417-22. doi: 10.3233/JAD-2006-10411.
Julien C, Tremblay C, Emond V, Lebbadi M, Salem JR N, Bennett DA. Sirtuin 1 reduction parallels the accumulation of tau in Alzheimer disease. J Neuropathol Exp Neurol. 2009;68(1):48-58. doi: 10.1097/NEN.0b013e3181922348, PMID 19104446.
Duan W, Mattson MP. Dietary restriction and 2-deoxyglucose administration improve behavioral outcome and reduce degeneration of dopaminergic neurons in models of Parkinson’s disease. J Neurosci Res. 1999;57(2):195-206. doi: 10.1002/(SICI)1097-4547(19990715)57:2<195::AID-JNR5>3.0.CO;2-P, PMID 10398297.
Pallas M, Pizarro JG, Gutierrez Cuesta J, Crespo Biel N, Alvira D, Tajes M. Modulation of SIRT1 expression in different neurodegenerative models and human pathologies. Neuroscience. 2008;154(4):1388-97. doi: 10.1016/j.neuroscience.2008.04.065, PMID 18538940.
Okawara M, Katsuki H, Kurimoto E, Shibata H, Kume T, Akaike A. Resveratrol protects dopaminergic neurons in midbrain slice culture from multiple insults. Biochem Pharmacol. 2007;73(4):550-60. doi: 10.1016/j.bcp.2006.11.003, PMID 17147953.
Sorolla MA, Nierga C, Rodriguez Colman MJ, Reverter Branchat G, Arenas A, Tamarit J. Sir2 is induced by oxidative stress in a yeast model of Huntington disease and its activation reduces protein aggregation. Arch Biochem Biophys. 2011;510(1):27-34. doi: 10.1016/j.abb.2011.04.002, PMID 21513696.
Perry VH, Lunn ER, Brown MC, Cahusac S, Gordon S. Evidence that the rate of Wallerian degeneration is controlled by a single autosomal dominant gene. Eur J Neurosci. 1990;2(5):408-13. doi: 10.1111/j.1460-9568.1990.tb00433.x, PMID 12106028.
Avery MA, Sheehan AE, Kerr KS, Wang J, Freeman MR. Wld S requires Nmnat1 enzymatic activity and N16-VCP interactions to suppress Wallerian degeneration. J Cell Biol. 2009;184(4):501-13. doi: 10.1083/jcb.200808042, PMID 19237597.
Chalkiadaki A, Guarente L. The multifaceted functions of sirtuins in cancer. Nat Rev Cancer. 2015;15(10):608-24. doi: 10.1038/nrc3985, PMID 26383140.
Roth M, Chen WY. Sorting out functions of sirtuins in cancer. Oncogene. 2014;33(13):1609-20. doi: 10.1038/onc.2013.120, PMID 23604120.
Choi HN, Bae JS, Jamiyandorj U, Noh SJ, Park HS, Jang KY. Expression and role of SIRT1 in hepatocellular carcinoma. Oncol Rep. 2011;26(2):503-10. doi: 10.3892/or.2011.1301, PMID 21567102.
Zhao G, Cui J, Zhang JG, Qin Q, Chen Q, Yin T. SIRT1 RNAi knockdown induces apoptosis and senescence, inhibits invasion and enhances chemosensitivity in pancreatic cancer cells. Gene Ther. 2011;18(9):920-8. doi: 10.1038/gt.2011.81, PMID 21677689.
Zhang JG, Zhao G, Qin Q, Wang B, Liu L, Liu Y. Nicotinamide prohibits proliferation and enhances chemosensitivity of pancreatic cancer cells through deregulating SIRT1 and Ras/Akt pathways. Pancreatology. 2013;13(2):140-6. doi: 10.1016/j.pan.2013.01.001, PMID 23561972.
Liu C, Huang Z, Jiang H, Shi F. The sirtuin 3 expression profile is associated with pathological and clinical outcomes in colon cancer patients. BioMed Res Int. 2014;2014:871263. doi: 10.1155/2014/871263, PMID 25105144.
Wang S, Chen X, Zhang Z, Wu Z. MicroRNA-1225-5p inhibits the development and progression of thyroid cancer via targeting sirtuin 3. Pharmazie. 2019;74(7):423-7. doi: 10.1691/ph.2019.9411, PMID 31288899.
Li D, Bi FF, Chen NN, Cao JM, Sun WP, Zhou YM. A novel crosstalk between BRCA1 and sirtuin 1 in ovarian cancer. Sci Rep. 2014;4(1):6666. doi: 10.1038/srep06666, PMID 25323003.
Tian J, Yuan L. Sirtuin 6 inhibits colon cancer progression by modulating PTEN/AKT signaling. Biomed Pharmacother. 2018;106:109-16. doi: 10.1016/j.biopha.2018.06.070, PMID 29957460.
Sun X, Wang S, Li Q. Comprehensive analysis of expression and prognostic value of sirtuins in ovarian cancer. Front Genet. 2019;10:879. doi: 10.3389/fgene.2019.00879, PMID 31572453.