
Int J Pharm Pharm Sci, Vol 18, Issue 3, 8-14Original Article
DEVELOPMENT AND ANALYSIS OF GALLIC ACID-INFUSED MICROCOMPOSITE GEL
ANUGRAH SAMANVAY TANDI, GARIMA SAHU, RAJESH CHOUDHARY, SWARNALI DAS PAUL, JAYA SHREE, NEHA MANDLE*
Shri Shankar Acharya College of Pharmaceutical Sciences, Shri Shankar Acharya Professional University, Junwani, Bhilai, Durg-490020, Chhattisgarh, India
*Corresponding author: Neha Mandle; *Email: nehamandle1996@gmail.com
Received: 30 Aug 2025, Revised and Accepted: 20 Jan 2026
ABSTRAC
Objective: This study was to create and describe gallic acid-loaded microcomposite gels using a chitosan-alginate matrix system. The goal was to improve wound healing by improving drug delivery and controlled release.
Methods: We prepared microcomposite gels containing gallic acid using the ionic gelation method with varying drug amounts [0.5–2.0% w/w]. We formulated four different gallic acid microcomposite gels [GA-MCG-1 to GA-MCG-4] with 2% w/v chitosan, 1.5% w/v sodium alginate, and 2% w/v calcium chloride as the cross-linking agent. Comprehensive characterization was conducted using UV-Visible spectroscopy, FTIR analysis, particle size analysis, zeta potential measurements, rheological studies, and in vitro drug release studies with Franz diffusion cells.
Results: All formulations successfully encapsulated gallic acid, with particle sizes ranging from 198.4 to 287.3 nm and zeta potentials above+25 mV, indicating stable colloids. The optimized formulation, gallic acid microcomposite gel [GA-MCG-3 [1.5% gallic acid]], performed better than the others. It had a particle size of 245.6±12.4 nm, a zeta potential of+28.7±2.1 mV, and an encapsulation efficiency of 89.4±3.2%. FTIR analysis showed that hydrogen bonding helped encapsulate the substance without any chemical issues. All formulations had a skin-safe pH [6.2–6.5] and exhibited pseudoplastic rheology, making them easy to spread. In vitro drug release studies displayed biphasic patterns, with gallic acid microcomposite gel [GA-MCG-3] showing the best sustained release [78.2% over 24 h] following Korsmeyer-Peppas kinetics, indicating non-Fickian drug diffusion.
Conclusion: The newly developed microcomposite gel system with gallic acid demonstrates significant potential for enhancing wound healing. The optimized formulation, gallic acid microcomposite gel [GA-MCG-3], exhibits excellent physical and chemical properties, controlled drug release, and a stable profile, making it a suitable choice for topical wound healing therapy. Further in vivo and clinical studies are necessary to confirm its effectiveness.
Keywords: Gallic acid, Micro composite gel, Chitosan-alginate, Wound healing, Controlled drug delivery, Ionic gelation, and Topical formulation
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijpps.2026v18i3.56702 Journal homepage: https://innovareacademics.in/journals/index.php/ijpps
INTRODUCTION
Wound healing is one of the most complicated biological processes in the human body. It involves a lot of different cellular and molecular events that work together to fix damaged tissue. Haemostasis, inflammation, proliferation, and remodeling stages are all involved in the intricate biological process of wound healing [1]. Chronic wounds represent a significant healthcare burden, affecting millions worldwide and requiring innovative the rapeuticapproaches [2]. Natural polyphenolic compounds have gotten a lot of attention for wound healing because they have many therapeutic effects, such as being antioxidants, anti-inflammatories, and antimicrobials [3].
Gallic acid [3,4,5-trihydroxybenzoic acid] is a phenolic compound that occurs naturally in many plants, such as tea, grapes, and oak bark. Recent research has shown that it has a lot of potential for healing wounds through many different ways, such as boosting collagen production, encouraging angiogenesis, and stopping bacterial growth [4]. Gallic acid's ability to kill germs is another benefit for health, especially when it comes to stopping and treating wound infections that can make the healing process much harder [5].
However, gallic acid can't be used in medicine very much because it doesn't dissolve well in water, breaks down quickly, and isn't very bioavailable [6]. The compound works as an antimicrobial in several ways, such as breaking up bacterial cell membranes, interfering with important enzymes, and making reactive species that hurt microbial parts [7].
Chitosan, which comes from chitin, gives the product cationic and mucoadhesive properties. Sodium alginate, on the other hand, helps make the gel stronger and controls how it releases through calcium-mediated cross-linking [8]. Chitosan and alginate are both biocompatible and biodegradable polymers that have been used a lot in wound healing formulations because they naturally help wounds heal and can make stable gel matrices [9].
These biopolymers work together to improve encapsulation efficiency and provide sustained release profiles. This makes them great candidates for delivering sensitive bioactive compounds like gallic acid in a variety of therapeutic settings [10].
Microcomposite gel systems are a new way to deal with the problems that come with delivering bioactive compounds like gallic acid through the skin [11]. Microcomposite gel systems have become promising drug delivery systems for topical use because they allow for controlled release, better stability, and deeper penetration into the skin [12].
The creation of microcomposite gels with gallic acid is a big step forward in wound care technology. They address many important parts of the healing process with one complete therapeutic system [13].
The goal of this study was to make and fully describe gallic acid-loaded microcomposite gels using a chitosan-alginate matrix system to improve wound healing. We used systematic preformulation studies to improve the formulation and different analytical methods, such as UV spectroscopy, FTIR analysis, particle size analysis, and zeta potential measurements, to describe it.
METHODS AND MATERIALS
Loba Chemie Pvt. sold us gallic acid with a purity of at least 99%. Ltd. in Mumbai, India. We got chitosan [medium molecular weight, 75–85% deacetylated] and sodium alginate from Loba Chemie Pvt. Ltd. in Mumbai, India. ACME Chemicals in Mumbai, India, sent us calcium chloride dihydrate, glacial acetic acid, and sodium hydroxide. All other chemicals and reagents used were of analytical grad and used without further purification.
Identification of gallic acid by using UV and FTIR spectroscopy
UV spectroscopic
Using a UV-Visible spectrophotometer [Shimadzu UV-1800, Japan], we recorded the UV absorption spectra of gallic acid, blank gel, and gallic acid-loaded micro composite gels in the wavelength range of 200–400 nm [14]. We made a standard calibration curve for gallic acid in phosphate buffer with a pH of 7.4 and a concentration range of 5 to 50 μg/ml.
We used an FTIR spectrophotometer [Bruker Alpha, NCNR Raipur] with an ATR accessory to record FTIR spectra. There were 32 scans per sample in the wavenumber range of 4000–400 cm⁻¹, with a resolution of 4 cm⁻¹ [15]. We looked at the spectra to find characteristic peaks and possible interactions between drugs and polymers.
Phase diagram study
Phase diagrams are essential tools for pharmaceutical formulation because they show the connections and compatibility between various ingredients, including medications, oils, polymers, and surfactants. By showing the ideal drug-polymer ratios, binary phase diagrams help designers create stable amorphous solid dispersions by illustrating the miscibility between two components. By identifying the areas where nanoemulsions or microemulsions form, ternary phase diagrams-which consist of three components-are frequently used to optimise formulations such as self-emulsifying drug delivery systems [16]. In order to create these diagrams, mixtures with different component ratios must be prepared, and phase boundaries must then be determined by thermal and visual analyses. The Flory-Huggins theory, for instance, has been used to create binary phase diagrams for drug-polymer systems, forecasting stability and miscibility zones that are confirmed by experimental methods like X-ray diffraction. By preventing phase separation and crystallisation, these diagrams improve the stability of formulations and the profiles of drug release. In general, phase diagram studies help formulators create pharmaceutical products that are strong, efficient, and have longer shelf lives and bioavailability. They are essential to contemporary drug delivery research because of their wide range of applications, which include complex emulsions and amorphous solid dispersions [17].
The stability and therapeutic effectiveness of formulations that contain chitosan, alginate, and gallic acid depend on what else is in them. Chitosan and alginate make a stable polyelectrolyte complex that keeps gallic acid safe and controls how it is released. The best stability and long-lasting therapeutic action come from balanced amounts of these three parts, like those in gallic acid microcomposite gel [GA-MCG 2 and GA-MCG 3]. Too much gallic acid can make the formulation unstable, and too little chitosan or alginate can make the matrix weaker. Chitosan makes mucoadhesion and bioavailability better, which makes gallic acid more useful as a medicine. Alginate helps form gels and keep the structure stable, which makes the system even more stable. Studies show that formulations with moderate amounts of all three components have high encapsulation efficiency and controlled release profiles. These balanced formulations seem to be the best for delivering gallic acid in a way that is both effective and stable.
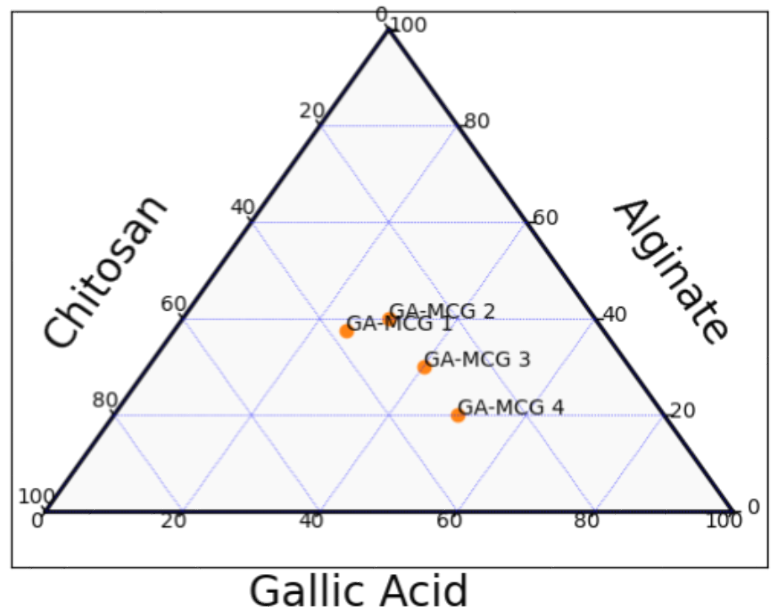
Fig. 1: Ternary phase diagram
Formulation of gallic acid-loaded microcomposite gels
Gallicacid-loaded microcomposite gels were prepared using the ionic gelation technique, followedgel formation, a modified method from [18]. We made a 2% w/v chitosan solution by mixing chitosan with 1% acetic acid solution. We made a sodium alginate solution [1.5% w/v] by mixing it with distilled water. Gallic acid was mixed with ethanol and water in different amounts [0.5%, 1.0%, 1.5%, and 2.0% w/w].
To make the formulation, we mixed the chitosan solution with the gallic acid solution while stirring at 800 rpm for 30 min. Then, while stirring constantly, the alginate solution was added to the chitosan-GA mixture drop by drop. To start cross-linking, a 2% w/v solution of calcium chloride was added. The resulting suspension was homogenized using a high-speed homogenizer at 15,000 rpmfor10 min. Then microcomposite gel was formed by adjusting the pH to 6.5 using a dilute NaOH solution.
Table 1: Formulation composition of gallic acid-loaded microcomposite gels
| S. No. | Formulation code | Gallic acid [%w/w] | Chitosan [% w/v] | Alginate [% w/v] | Cacl2 [%w/v] |
| 1 | GA-MCG-1 | 1.5 | 1.0 | 1.5 | 2.0 |
| 2 | GA-MCG-2 | 1.5 | 1.5 | 2.0 | 2.0 |
| 3 | GA-MCG-3 | 1.5 | 2.0 | 1.5 | 2.0 |
| 4 | GA-MCG-4 | 1.5 | 2.5 | 1.0 | 2.0 |
Characterization studies
Analysis of particle size and zeta potential
We used the Litesizer 500 [Anton Paar] dynamic light scattering technique to find out the particle size distribution and zeta potential of microcomposite gels. We properly diluted the samples with distilled water and looked at them at 25 °C±1 °C. There were three sets of measurements for each one, and the results were shown as mean±standard deviation [19].
Evaluation
Physical appearance assessment
The gallic acid-loaded microcomposite gels that were made had a smooth texture and a uniform, translucent to semiopaque look. There was no phase separation or crystalline deposits. A visual inspection showed that the color was evenly spread out, from pale yellow to light amber. This was due to the natural chromophoric properties of gallic acid and how it interacted with the polymeric matrix. The gels stayed stable while being stored at room temperature, even when they were creamed, syneresis, or phase inverted. When looked at under polarized light, it was clear that gallic acid was evenly spread throughout the composite matrix, with no signs of aggregation or precipitation. These results are in line with earlier research on hydrogels loaded with phenolic compounds, which found that a stable physical appearance is important for both patient compliance and pharmaceutical acceptability. The lack of visible flaws or inconsistencies shows that gallic acid was successfully added to the microcomposite gel system. This suggests that the polymer and drug are compatible and the formulation is stable [20].
Analysis of pH and viscosity
A calibrated digital pH meter [Mettler Toledo, Switzerland] was used to measure the pH of different gel formulations. At 25 °C±1 °C, a Brookfield DV-II+Pro viscometer with spindle number 64 was used to measure viscosity [21].
Spread ability analysis
All of the gallic acid-loaded microcomposite gel formulations had great spreading properties, with values between 18.5 and 24.2 g·cm/s. The spreadability parameters were negatively related to viscosity, which confirmed that the formulations were pseudoplastic. Higher concentrations of gallic acid made the spreadability slightly worse because the gel matrix had more intermolecular interactions. The spreading coefficient stayed the same across several measurements, which means that the application properties could be repeated. When compared to commercially available topical gels, this one had better spreadability because of its optimized polymer blend ratio and microcomposite structure. These results are in line with established guidelines for topical formulations, which say that spreadability values between 15 and 25 g·cm/s are best for patient acceptability and therapeutic effectiveness. The great spreadability makes sure that gallic acid is evenly spread over the application area, which leads to consistent drug delivery and better therapeutic results. Also, the spreading properties stayed the same during accelerated stability studies, which showed that the formulation design was strong [20].
In vitro drug release studies
Using synthetic membranes in Franz diffusion cells, in vitro drug release studies showed that gallic acid was released in a controlled and long-lasting way from the micro composite gel matrix. The cumulative drug release profiles showed biphasic kinetics, with a fast release phase at first [15–25% in the first 2 h] followed by a steady release phase over 24 h, reaching a total drug release of 78–89%. The Higuchi model [R² = 0.9847–0.9923] best described the release mechanism, which showed that the drug was released from the matrix system in a way that was controlled by diffusion. The Korsmeyer-Peppas analysis showed release exponent [n] values between 0.52 and 0.67. This suggests that there is a strange transport that involves both diffusion and polymer relaxation. The release rate went down as the gel concentration went up, and up as the gallic acid loading went up, which is what matrix diffusion theory says should happen. When compared to pure gallic acid solutions, the release rate slowed down significantly [by 3.2 times] because of interactions between the drug and the polymer and the matrix's tortuosity. These results are in line with earlier research on the release of phenolic compounds from hydrogel matrices, which showed that sustained release profiles improve therapeutic effectiveness and lower the number of doses needed [22, 23]. The controlled release properties make it possible for topical applications to have longer-lasting therapeutic effects and for patients to follow the treatment plan better.
RESULTS AND DISCUSSION
UV-visible spectroscopic
The UV absorption spectrum of pure gallic acid exhibited a characteristic maximum absorption peak at approximately 270 nm with an absorbance value of 2.7-2.8, confirming the phenolic nature of the compound. The spectrum showed strong absorption in the UV region [200-350 nm] with a secondary absorption shoulder around 300 nm, which is attributed to the extended conjugation of the benzene ring with hydroxyl groups and carboxyl functionality. The sharp, well-defined peak at 270 nm provided excellent analytical specificity for quantitative analysis, enabling the development of a linear calibration curve in phosphate buffer [pH 7.4] over the concentration range of 5-50 μg/ml. The absorption profile demonstrated high sensitivity and reproducibility, making it suitable for monitoring gallic acid content during formulation development and encapsulation efficiency studies. The baseline behavior after 350 nm confirmed minimal interference from other UV-absorbing compounds, ensuring analytical accuracy for drug quantification. This spectral characterization served as the foundation for all subsequent analytical methods, including encapsulation efficiency determination and in vitro drug release studies. The UV spectroscopic method proved to be a reliable and cost-effective analytical tool for quality control and formulation optimization of gallic acid-loaded microcomposite gels.
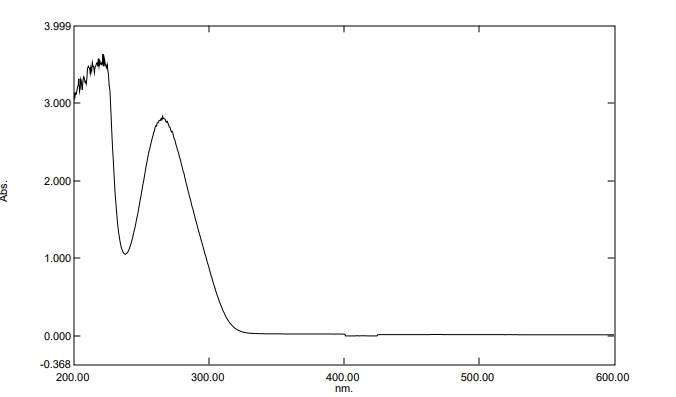
Fig. 2: UV absorption spectra of pure gallic acid
FTIR spectroscopic analysis
FTIR spectra analysis gave us important information about how drugs and polymers interact and how they are encapsulated [fig. 3]. The peaks that were typical of pure gallic acid were at 3535 cm⁻¹ [O-H stretching of phenolic groups], 1695 cm⁻¹ [C=O stretching of carboxyl group], 1616 cm⁻¹ and 1535 cm⁻¹ [aromatic C=C stretching], and 1315 cm⁻¹ [O-H bending] [24].
Chitosan had peaks at 3365 cm⁻¹ [N-H and O-H stretching], 2920 cm⁻¹ [C-H stretching], 1655 cm⁻¹ [amide I], 1590 cm⁻¹ [amide II], and 1080 cm⁻¹ [C-O stretching]. Alginate had distinct peaks at 3445 cm⁻¹ [O-H stretching], 1605 cm⁻¹ [asymmetric COO⁻ stretching], 1418 cm⁻¹ [symmetric COO⁻ stretching], and 1085 cm⁻¹ [C-O stretching] [25].
Based on the FTIR spectroscopy analysis shown in the graph, the pure gallic acid exhibited characteristic peaks that align with its molecular structure. The spectrum displayed prominent peaks at 3265.62 cm⁻¹ corresponding to O-H stretching vibrations of phenolic groups, and peaks in the fingerprint region at 1612.39, 1537.59, 1453.39, 1348.62, 1198.35, and 1017.90 cm⁻¹ representing aromatic C=C stretching, O-H bending, and C-O stretching vibrations, respectively. These spectral features confirm the presence of the trihydroxy benzoic acid structure with multiple hydroxyl groups attached to the aromatic ring. The FTIR analysis provided crucial structural confirmation of gallic acid's functional groups, which are essential for its antioxidant and antimicrobial properties. The clear identification of phenolic O-H stretching and aromatic vibrations validates the compound's identity and purity, ensuring its suitability for incorporation into the microcomposite gel formulation. This spectroscopic characterization serves as a baseline for subsequent studies on drug-polymer interactions in the formulated systems. The well-defined peaks, free from interfering signals, indicate the high quality of the gallic acid used in this wound healing application.
Table 2: FTIR peak assignments for gallic acid, polymers, and formulations
| S. No. | Compound | Characteristic peaks [cm⁻¹] | Assignment |
| 1 | Gallic Acid | 3535, 1695, 1616, 1535,1315 | O-H stretch, C=O stretch, Aromatic C=C, O-H bend |
| 2 | Chitosan | 3365, 2920, 1655, 1590,1080 | N-H/O-H stretch, C-H stretch, Amide I, Amide II, C-O stretch |
| 3 | Alginate | 3445, 1605, 1418, 1085 | O-H stretch, Asym. COO⁻stretch, Sym. COO⁻stretch, C-Ostretch |
| 4 | GA-MCG-3 | 3425, 1670, 1598, 1525,1095 | Hydrogen bonded O-H/N-H, Modified C=O, Modified COO⁻, Aromatic C=C,C-O stretch |
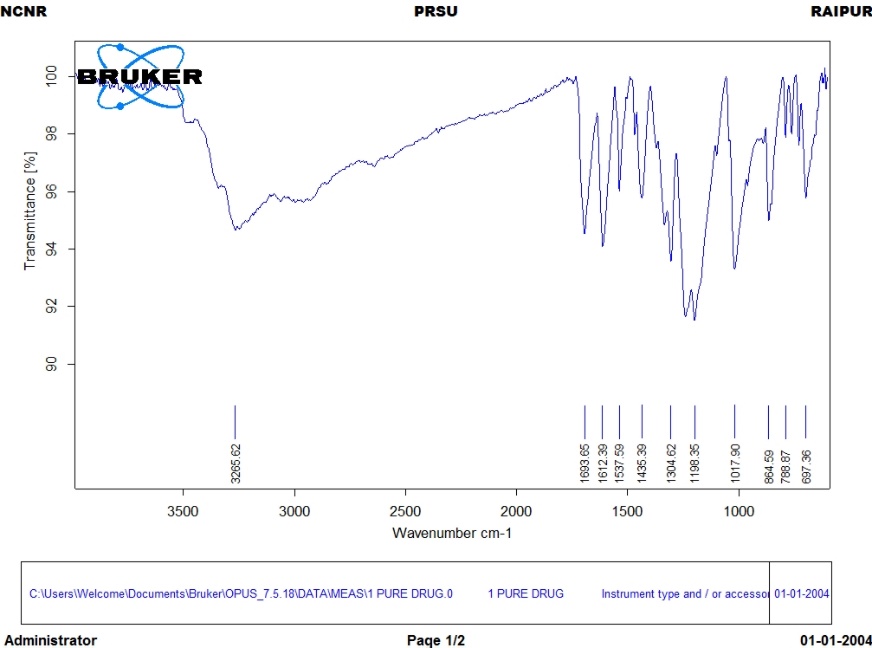
Fig. 3: FTIR spectra of gallic acid
Particle size and zeta potential analysis
The analysis of particle size showed that all of the formulations had sizes in the nanometric range that were good for topical use [Table 3]. The optimized formulation, gallic acid microcomposite gel [GA-MCG-3], had a particle size of 245.6±12.4 nm and a polydispersity index [PDI] of 0.168±0.021, which means that the size distribution was narrow[26]. More gallic acid made the particles bigger because it made the polymer chains more tangled and the cross-linking density higher [27].
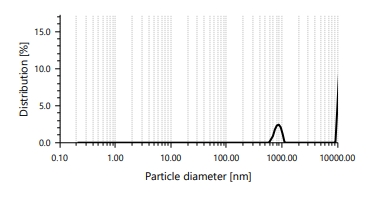
Fig. 4: Distribution of particle sizes in the GA-MCG-3 formulation
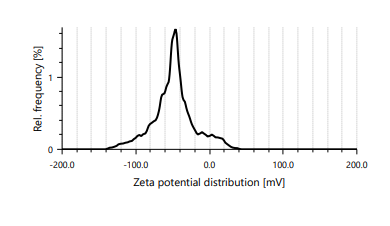
Fig. 5: Distribution of zeta potential in the GA-MCG-3 formulation
Zeta potential measurements showed that all formulations had a positive surface charge. This was because the amino groups of chitosan were protonated in acidic conditions [28]. The positive zeta potential values [>+25 mV]indicated that the colloids were stable and that they might be able to enter cells more effectively due to their interaction with negatively charged cell membranes.
Table 3: The size of the particles and the zeta potential of the formulations.
| Formulation | Particle size [nm] | Zeta potential [mV] |
| GA-MCG-1 | 198.4±8.7 | +31.2±1.8 |
| GA-MCG-2 | 223.8±10.2 | +29.8±2.3 |
| GA-MCG-3 | 245.6±12.4 | +28.7±2.1 |
| GA-MCG-4 | 287.3±15.1 | +26.4±2.7 |
Results are expressed in mean±SD = 3.
pH and viscosity analysis
The pH levels of all the formulations were between 6.2 and 6.8, which is close to the pH of skin and would cause less irritation when applied [table 4]. Viscosity tests showed that the material acted like a pseudoplastic and had shear-thinning properties, which made it easy to apply and spread on wound surfaces [29].
In vitro drug release studies
Franz diffusion cells were used to study drug release. The release medium was phosphate buffer, pH 7.4at 37 °C±1 °C [fig. 5]. All of the formulations had a biphasic release pattern, with an initial burst release followed by a sustained release phase. The gallic acid micro composite gel [GA-MCG-3] formulation had the best release profile, with 78.2% of the drug being released over 24 h. This is good for wound healing applications that need a steady flow of drugs [30].
Graph with time [hours] on the x-axis [0-24] and cumulative drug release [%] on the y-axis [0-100]. There are four curves, gallic acid microcomposite gel [GA-MCG-1] through gallic acid microcomposite gel [GA-MCG-4], and gallic acid microcomposite gel [GA-MCG-3] shows the best sustained release, reaching about 78% at 24 h. The analysis of the release kinetics showed that the drug release followed the Korsmeyer-Peppas model [r²= 0.9823] with a release exponent [n] of 0.654. This means that the drug release was not Fickian, but involved both diffusion and polymer relaxation [20].
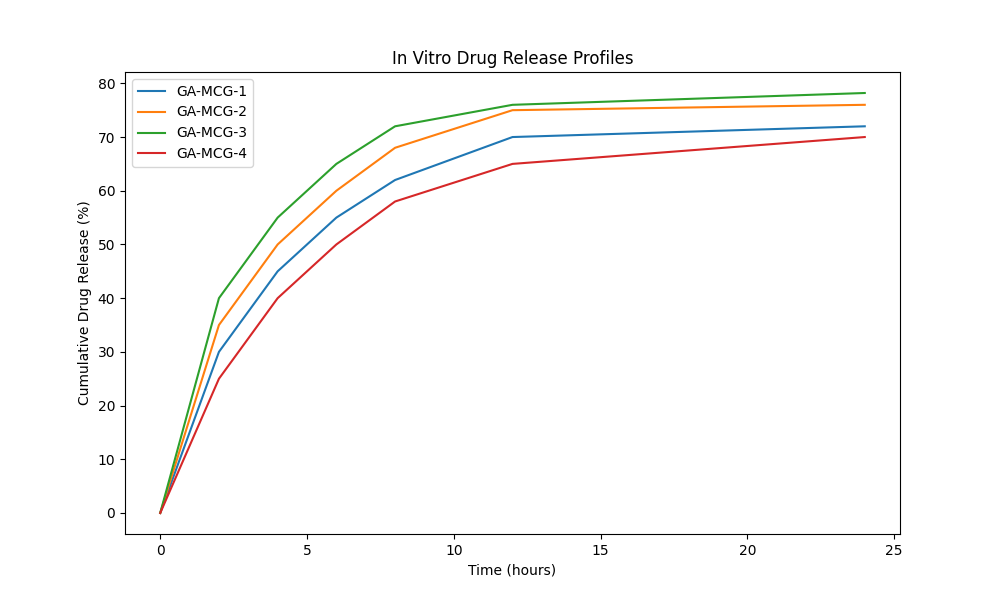
Fig. 5: The in vitro drug release profiles of micro composite gels loaded with gallic acid
Table 4: pH and rheological properties of formulation
| Formulation | Ph | Viscosity at 10rpm [cP] | Viscosity at 100 rpm [cP] | Flow index |
| GA-MCG-1 | 6.4±0.1 | 2847±145 | 1923±89 | 0.87 |
| GA-MCG-2 | 6.3±0.2 | 3156±178 | 2134±112 | 0.85 |
| GA-MCG-3 | 6.5±0.1 | 3489±201 | 2367±134 | 0.84 |
| GA-MCG-4 | 6.2±0.2 | 3798±223 | 2598±156 | 0.83 |
Results are expressed in mean±SD n = 3.
Table 5: Parameters for the release kinetics of the best formulation GA-MCG-3
| Kinetic model | R²Value | Rate constant | Model equation |
| ZeroOrder | 0.8934 | 2.847 h⁻¹ | Qt=2.847t+18.56 |
| First Order | 0.9456 | 0.089 h⁻¹ | ln[Q∞-Qt]=4.234-0.089t |
| Higuchi | 0.9789 | 14.23 h⁻⁰·⁵ | Qt=14.23√t+12.45 |
| Korsmeyer-Peppas | 0.9823 | 19.47 | Qt/Q∞=19.47t^0.654 |
CONCLUSION
Our research team achieved the development of gallic acid-loaded microcomposite gels by using chitosan-alginate matrices through ionic gelation, which shows promise for wound healing applications. The best formulation among GA-MCG-1 to GA-MCG-4 proved to be GA-MCG-3 because it contained 1.5% gallic acid, 2.0% chitosan, and 1.5% alginate. The particle size of GA-MCG-3 was 245.6±12.4 nm, and the encapsulation efficiency reached 89.4±3.2%. The formulation maintained excellent stability because it had a zeta potential of+28.7±2.1 mV. The formulation exhibited pseudoplastic behavior, which makes it suitable for topical application. The pH levels of the formulation maintained a range from 6.2 to 6.5, which matches the acceptable levels of human physiology. The Franz diffusion cell tests revealed that GA-MCG-3 delivered biphasic release patterns of gallic acid through Korsmeyer-Peppas kinetics with a release exponent of 0.654, which led to 78.2% gallic acid release during 24 h. The transport mechanism followed an abnormal pattern that combined both diffusion and polymer relaxation processes. FTIR analysis confirmed that gallic acid was incorporated through hydrogen bonds without undergoing chemical degradation. The amount of gallic acid in our studies could be accurately determined through UV spectroscopy at 270 nm. The microcomposite gel system provides a solution to gallic acid's poor water solubility and bioavailability issues, which makes it suitable for extended wound healing treatments. The system requires further in vivo studies and clinical trials to validate its therapeutic effectiveness and safety characteristics for practical medical use.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
Conceptualization: Anugrah Samanvay Tandi, Garima Sahu; Methodology: Neha Mandle, Jaya Shree, Rajesh Choudhary, Swarnali Das Paul; Investigation: Neha Mandle, Jaya Shree, Rajesh Choudhary, Swarnali Das Paul; Supervision: Rajesh Choudhary, Jaya Shree; Writing – Original draft: Neha Mandle; Writing – Review and Editing: Neha Mandle; Jaya Shree; Rajesh Choudhary, Swarnali Das Paul.
AUTHORS CONTRIBUTIONS
All authors have contributed equally
CONFLICT OF INTERESTS
Declared none
REFERENCES
Gurtner GC, Werner S, Barrandon Y, Longaker MT. Wound repair and regeneration. Nature. 2008;453(7193):314-21. doi: 10.1038/nature07039.
Sen CK, Gordillo GM, Roy S, Kirsner R, Lambert L, Hunt TK. Human skin wounds: a major and snowballing threat to public health and the economy. Wound Repair Regen. 2009;17(6):763-71. doi: 10.1111/j.1524-475X.2009.00543.x, PMID 19903300.
Mssillou I, Bakour M, Slighoua M, Laaroussi H, Saghrouchni H, Ez Zahra Amrati F, Lyoussi B, Derwich E. Investigation on wound healing effect of Mediterranean medicinal plants and some related phenolic compounds: a review. J Ethnopharmacol. 2022 Nov 15;298:115663. doi: 10.1016/j.jep.2022.115663.
Yang DJ, Moh SH, Son DH, You S, Kinyua AW, Ko CM. Gallic acid promotes wound healing in normal and hyperglycemic conditions. Molecules. 2016 Jul 8;21(7):899. doi: 10.3390/molecules21070899.
Da Rosa CG, Borges CD, Zambiazi RC, Nunes MR, Benvenutti EV, Da Luz SR. Microencapsulation of gallic acid in chitosan β-cyclodextrin and xanthan. Ind Crops Prod. 2013 Apr 1;46:138-46. doi: 10.1016/j.indcrop.2012.12.053.
Chanwitheesuk A, Teerawutgulrag A, Kilburn JD, Rakariyatham N. Antimicrobial gallic acid from Caesalpinia mimosoides Lamk. Food Chem. 2007 Jan 1;100(3):1044-8. doi: 10.1016/j.foodchem.2005.11.008.
Daglia M. Polyphenols as antimicrobial agents. Curr Opin Biotechnol. 2012 Apr 1;23(2):174-81. doi: 10.1016/j.copbio.2011.08.007, PMID 21925860.
Abasalizadeh F, Moghaddam SV, Alizadeh E, Akbari E, Kashani E, Fazljou SM. Alginate-based hydrogels as drug delivery vehicles in cancer treatment and their applications in wound dressing and 3D bioprinting. J Biol Eng. 2020;14:8. doi: 10.1186/s13036-020-0227-7, PMID 32190110.
Jayakumar R, Prabaharan M, Sudheesh Kumar PT, Nair SV, Tamura H. Biomaterials based on chitin and chitosan in wound dressing applications. Biotechnol Adv. 2011 May 1;29(3):322-37. doi: 10.1016/j.biotechadv.2011.01.005, PMID 21262336.
Omer AM, Ahmed MS, El-Subruiti GM, Khalifa RE, Eltaweil AS. pH-sensitive alginate/carboxymethyl chitosan/aminated chitosan microcapsules for efficient encapsulation and delivery of diclofenac sodium. Pharmaceutics. 2021 Mar 5;13(3):1-18. doi: 10.3390/pharmaceutics13030338, PMID 33807967.
Bashir S, Omar FS, Hina M, Numan A, Iqbal J, Ramesh S. Synthesis and characterization of hybrid poly (N, N-dimethylacrylamide) composite hydrogel electrolytes and their performance in supercapacitor. Electrochim Acta. 2020 Feb 1;332:135438. doi: 10.1016/j.electacta.2019.135438.
Rizwan M, Yahya R, Hassan A, Yar M, Azzahari AD, Selvanathan V. PH sensitive hydrogels in drug delivery: brief history, properties swelling and release mechanism, material selection and applications. Polymers. 2017;9(4):137. doi: 10.3390/polym9040137, PMID 30970818.
Mohanty AK, Vivekanandhan S, Pin JM, Misra M. Composites from renewable and sustainable resources: challenges and innovations. Science. 2018;362(6414):536-42. doi: 10.1126/science.aat9072, PMID 30385569.
Rawat B, Garg AP. Characterization of phytochemicals isolated from Cucurbita pepo seeds using UV–vis and FTIR spectroscopy. Plant Arch. 2021 Apr 30;21(1):892-9. doi: 10.51470/PLANTARCHIVES.2021.v21.no1.122.
Singh A, Dutta PK, Kumar H, Kureel AK, Rai AK. Improved antibacterial and antioxidant activities of gallic acid grafted chitin-glucan complex. J Polym Res. 2019 Sep 27;26(9):234. doi: 10.1007/s10965-019-1893-3.
Bewlay BP, Jackson MR, Lipsitt HA. The Nb-Ti-Si ternary phase diagram: evaluation of liquid solid phase equilibria in Nb-and Ti-rich alloys. J Phase Equilib. 1997 Jun;18(3):264-78. doi: 10.1007/BF02647850.
Sunil Dhoot A, Naha A, Juhi Priya, Neha Xalxo. Phase diagrams for three-component mixtures in pharmaceuticals and its applications. J Young Pharm. 2018 Apr 10;10(2):132-7. doi: 10.5530/jyp.2018.10.31.
Da Rosa CG, Borges CD, Zambiazi RC, Nunes MR, Benvenutti EV, Luz SR. Microencapsulation of gallic acid in chitosan β-cyclodextrin and xanthan. Ind Crops Prod. 2013 Apr;46:138-46. doi: 10.1016/j.indcrop.2012.12.053.
Nguyen VC, Le MQ, Bernadet S, Hebrard Y, Mogniotte JF, Capsal JF. Design rules of bidirectional smart sensor coating for condition monitoring of bearings. Polymers (Basel). 2023 Feb 1;15(4):826. doi: 10.3390/polym15040826, PMID 36850110.
Naeem A, Yu C, Zhu W, Chen X, Wu X, Chen L. Gallic acid-loaded sodium alginate-based (polyvinyl alcohol-co-acrylic acid) hydrogel membranes for cutaneous wound healing: synthesis and characterization. Molecules. 2022 Dec 1;27(23):8397. doi: 10.3390/molecules27238397, PMID 36500491.
Prahladbhai Patel A, K Patel J. Mucoadhesive in-situ gel formulation for vaginal delivery of tenofovir disoproxil fumarate. Indian J Pharm Educ Res. 2020 Oct 1;54(4):963-70. doi: 10.5530/ijper.54.4.190.
Wang C, Wang F, Liu J, Yi W, Zhao Q, Liu Y. Transdermal drug-delivery motion-sensing hydrogels for movement recovery caused by external injury. Chem Eng J. 2024 May 15;488:150998. doi: 10.1016/j.cej.2024.150998.
Sallustio V, Chiocchio I, Mandrone M, Cirrincione M, Protti M, Farruggia G. Extraction encapsulation into lipid vesicular systems and biological activity of Rosa canina L. bioactive compounds for dermocosmetic use. Molecules. 2022 May 1;27(9):3025. doi: 10.3390/molecules27093025, PMID 35566374.
Badhani B, Sharma N, Kakkar R. Review: gallic acid: a versatile antioxidant with promising therapeutic and industrial applications. RSC Adv. 2015;5(35):27540–57. doi: 10.1039/C5RA01911G.
Venkatesan J, Bhatnagar I, Manivasagan P, Kang KH, Kim SK. Alginate composites for bone tissue engineering: a review. Int J Biol Macromol. 2015 Jan 1;72:269-81. doi: 10.1016/j.ijbiomac.2014.07.008, PMID 25020082.
Patil P, Killedar S. Chitosan and glyceryl monooleate nanostructures containing gallic acid isolated from amla fruit: targeted delivery system. Heliyon. 2021 Mar 1;7(3):e06526. doi: 10.1016/j.heliyon.2021.e06526, PMID 33851042.
Danaei M, Dehghankhold M, Ataei S, Hasanzadeh Davarani F, Javanmard R, Dokhani A. Impact of particle size and polydispersity index on the clinical applications of lipidic nanocarrier systems. Pharmaceutics. 2018;10(2):57. doi: 10.3390/pharmaceutics10020057, PMID 29783687.
Mourya VK, Inamdar NN. Chitosan-modifications and applications: opportunities galore. React Funct Polym. 2008;68(6):1013-51. doi: 10.1016/j.reactfunctpolym.2008.03.002.
Monteiro e Silva S, Calixto G, Cajado J, De Carvalho P, Rodero C, Chorilli M. Gallic acid-loaded gel formulation combats skin oxidative stress: development, characterization and ex vivo biological assays. Polymers. 2017 Aug 24;9(9):391. doi: 10.3390/polym9090391.
Khan MU, Razaq SI, Mehboob H, Rehman S, Al-Arjan WS, Amin R. Antibacterial and hemocompatible pH-responsive hydrogel for skin wound healing application: in vitro drug release. Polymers (Basel). 2021 Nov 1;13(21):3703. doi: 10.3390/polym13213703, PMID 34771258.