
Int J Pharm Pharm Sci, Vol 18, Issue 2, 13-16Case Study
FIXED DRUG ERUPTION AFTER AN OVER-THE-COUNTER AYURVEDIC SUPPLEMENT (GOOD HEALTH) CONTAINING WITHANIASOMNIFERA AND ECLIPTA ALBA: A CASE REPORT
SNEHASHIS SINGHA*, ANOOP KUMAR
Department of Pharmacology and Therapeutics, King George’s Medical University (KGMU), Lucknow, Uttar Pradesh, India
*Corresponding author: Snehashis Singha; *Email: dieformanu2208@gmail.com
Received: 02 Oct 2025, Revised and Accepted: 02 Jan 2026
ABSTRAC
Fixed drug eruption (FDE) is a distinctive cutaneous adverse drug reaction that recurs at the same anatomical sites upon re-exposure to the offending agent. Although most cases are triggered by antibiotics and non-steroidal anti-inflammatory drugs, herbal and polyherbal formulations are increasingly being recognized as potential culprits. We report a 45 y old female who developed multiple erythematous to hyperpigmented patches on the trunk and extremities following the use of an over-the-counter Ayurvedic supplement, Good Health, containing Withania somnifera (Ashwagandha), Eclipta alba (Bhringraj), and other ingredients. The lesions subsided after discontinuation of the supplement and topical application of mometasone 0.1 % cream but reappeared at identical sites upon re-exposure, confirming the diagnosis of FDE. Causality assessment using WHO-UMC and Naranjo criteria categorized the event as “probable.” Confirmatory patch testing was not performed; hence, the exact culprit herb remains uncertain. This case highlights that even widely used herbal preparations can precipitate fixed drug eruptions and underscores the importance of herbal pharmacovigilance and patient counselling regarding over-the-counter Ayurvedic products.
Keywords: Fixed drug eruption, Ashwagandha, Bhringraj, Herbal medicine, Ayurvedic supplement, Drug hypersensitivity, Adverse drug reaction
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijpps.2026v18i2.57048 Journal homepage: https://innovareacademics.in/journals/index.php/ijpps
INTRODUCTION
Fixed drug eruption (FDE) is a recurrent, well-defined cutaneous adverse drug reaction characterized by the reappearance of lesions at identical sites upon re-exposure to the offending agent. Although most FDEs are attributed to antibiotics and non-steroidal anti-inflammatory drugs, herbal and polyherbal formulations are emerging as potential culprits due to their increasing use and complex composition.
The coronavirus disease-2019 (COVID-19) pandemic has significantly increased global psychological stress, anxiety, and depression [1, 2]. Prolonged stress can adversely affect both physical and mental health, contributing to metabolic, cardiovascular, endocrinological, and neuropsychiatric disorders [3–5]. Adaptogens are herbal extracts that enhance the ability of the body to resist stress and maintain homeostasis without causing dependency [6].
Among adaptogens, Withania somnifera (Ashwagandha) holds a prominent place in Ayurvedic medicine for promoting vigor, vitality, and overall health [7]. Its pharmacologically active withanolides exhibit anxiolytic, antidepressant, and neuroprotective properties in both experimental and clinical studies [8-12]. However, despite its therapeutic reputation, recent reports indicate that even Ashwagandha-containing supplements can rarely cause adverse reactions, including hepatic injury and cutaneous eruptions.
Good Health is a commercially available polyherbal Ayurvedic supplement that combines Withania somnifera(Ashwagandha) and Eclipta alba (Bhringraj) as key ingredients. Ashwagandha is recognized for its adaptogenic and immunomodulatory benefits, while Bhringraj has traditional applications in liver protection, anti-inflammatory activity, and hair health [13]. Other components such as Shatavari (Asparagus racemosus), Yashtimadhu (Glycyrrhiza glabra), and Vidarikand (Pueraria tuberosa), along with purified Shilajit and Swarna bhasma, contribute to its restorative and tonic effects.
Here, we report a rare case of FDE temporally associated with the use of Good Health, highlighting the diagnostic challenges posed by polyherbal formulations and emphasizing the need for vigilant herbal pharmacovigilance.

Fig. 1: Withaniasomnifera (Ashwagandha) plant. Roots and leaves of Withaniasomnifera (ashwagandha), a medicinal shrub widely used in Ayurvedic formulations for its adaptogenic and immunomodulatory properties. Photo credit: Oregon’s Wild Harvest (licensed for educational use; image source: National Center for Complementary and Integrative Health. Bethesda (MD): NCCIH. Available from: https://www.nccih.nih.gov/health/ashwagandha)
Case presentation
A 25 y old male presented to the dermatology outpatient department with complaints of recurrent, well-demarcated hyperpigmented patches and papules over the trunk and peripheral body sites. The lesions, varying in shape and size, were erythematous, non-pruritic, and painless. He reported that the first episode occurred in July 2021, approximately six months after initiating daily use of an over-the-counter Ayurvedic supplement, Good Health, consumed for gym-related vitality since January 2021. The eruptions resolved within a few weeks of discontinuing the supplement, leaving residual hyperpigmentation. In October 2021, after resuming the same supplement, the patient experienced recurrence of lesions at identical sites, followed by similar episodes in December 2021 and February 2022 upon re-exposure. There wasno associated fever, mucosal involvement, or systemic symptoms. He denied the use of any other allopathic or herbal medications during this period. Personal and family histories were unremarkable, and no prior drug allergies were reported. The temporal pattern of recurrence after re-exposure strongly suggested a diagnosis of fixed drug eruption (FDE) secondary to the polyherbal supplement.
Management involved permanent discontinuation of the supplement, application of topical mometasone 0.1 % cream twice daily, and oral antihistamines for symptomatic relief. Lesions resolved completely within 10 d, leaving residual hyperpigmentation. No recurrence was observed during a six-month follow-up period after avoidance (table 1. Composition of the supplement, their roles and potential dermatological evidence) (table 2 Timeline of the events).

Fig. 2: Eclipta alba (Bhringraj) plant. Leaves of Eclipta alba (bhringraj), an herb known for immunomodulatory and hepatoprotective activity and a constituent of the reported polyherbal supplement [15]
Table 1: Composition of the supplement (Good health capsule)
| Ingredient | Common name | Declared role/claim on label | Potential dermatologic relevance |
| Withaniasomnifera | Ashwagandha | Stress/adaptogen | Prior single-agent FDE case reported; HILI reports exist [14] |
| Eclipta alba | Bhringraj | Hepatoprotective/hair | Limited human ADR data; theoretical immunomodulatory activity [15] |
| Asparagus racemosus | Shatavari | General tonic | Sparse cutaneous ADR data. |
| Glycyrrhiza glabra | Yashtimadhu | Anti-inflammatory | Can alter cortisol metabolism; rare hypersensitivity. |
| Pueraria tuberosa | Vidarikand | Rejuvenative | Limited ADR data. |
| Purified Shilajit | — | Stamina/tonic | Adulteration concerns reported in literature. |
| Swarna bhasma | — | Rasayana | Variability in preparation; safety depends on standardization. |
Clinical examination and diagnosis
On physical examination, multiple well-defined erythematous and hyperpigmented patches and papules of varying shapes and sizes were observed across the trunk and peripheral body parts, with no signs of active inflammation or infection. The lesions were non-tender and asymptomatic, without mucosal involvement. The patient reported that these lesions consistently appeared after taking the Good Health Capsule and gradually subsided upon discontinuation, only to recur at the same sites with each re-exposure. Given this history of repeated episodes and the characteristic presentation, a diagnosis of fixed drug eruption (FDE) was strongly considered [16] (fig. 3). FDE is a well-recognized dermatologic manifestation of drug hypersensitivity, often presenting as recurrent erythematous or hyperpigmented lesions at the same site of drug exposure. In this case, the use of Good Health Capsule was suspected as the causative agent.
Management and outcome
The primary intervention involved discontinuing the suspected offending agent, Good Health Capsule. The patient was advised to stop taking the supplement, and treatment with Mometasone 0.1% creamwas prescribed for local application at night for three weekEach time the patient experienced a recurrence after re-exposure to the supplement, the lesions followed a similar pattern, appearing at the same sites and gradually subsiding with discontinuation and topical treatment. Over multiple episodes, the same treatment approach led to resolution, with significant improvement in pigmentation and lesion count. During follow-up, the patient reported no long-term complications, but recurrences occurred whenever the supplement was resumed (table 2).
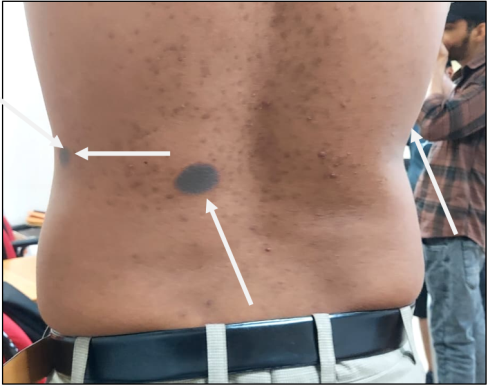
Fig. 3: Drug eruption rashes with FDE patches on the back and legs, recurring as dark red lesions (depicted by arrows), often leaving post-inflammatory hyperpigmentation
Table 2: Timeline of exposure and events
| Date | Event description |
| January 2021 | Began taking Good Health supplement daily for general wellness. |
| July 2021 | First appearance of erythematous–hyperpigmented lesions; supplement stopped → lesions resolved. |
| October 2021 | Restarted supplement → recurrence of lesions at identical sites. |
| December 2021 | Second recurrence following re-exposure. |
| February 2022 | Third recurrence with same morphology and distribution. |
| March 2022 | Supplement permanently discontinued; treated with mometasone 0.1 % cream → recovery. |
| September 2022 | Six-month follow-up — no recurrence observed. |
DISCUSSION
FDE represents a unique type of cutaneous ADR characterized by recurrence of lesions at the same anatomical site after re-exposure to the culprit agent. The pathogenesis involves cytotoxic T-cell-mediated immune responses to drug-derived antigens retained in resident epidermal memory cells [17]. Diagnosis is based on clinical morphology, history of re-exposure, and exclusion of alternative causes. Polyherbal formulations pose unique challenges for ADR attribution because of multiple active constituents and possible contaminants [18, 19]. Ashwagandha (Withania somnifera) has been associated with FDE in a prior report by Sehgal et al. (2012) [20], and hepatotoxicity cases have also been documented [21]. Eclipta alba (Bhringraj), while traditionally used for hepatoprotection and hair growth, has immunomodulatory and bioactive constituents that may provoke idiosyncratic reactions. Hence, in a polyherbal matrix like Good Health, specific culprit identification is difficult and causality should remain probabilistic.
Previous literature shows that herbal FDE is rare but increasingly recognized, with reported triggers including Tinospora cordifolia, Aloe vera, and ashwagandha supplements [22–24]. Unregulated marketing and absence of robust post-marketing surveillance contribute to under-reporting. Therefore, this case adds valuable clinical evidence to the limited database of herbal-related FDE.
CONCLUSION
This case highlights that even widely used Ayurvedic and polyherbal supplements can provoke fixed drug eruption (FDE) in susceptible individuals. Because such formulations contain multiple bioactive components, specific culprit identification often remains uncertain without confirmatory testing such as patch tests or supervised rechallenge. Clinicians should maintain a high index of suspicion for herbal-related adverse drug reactions (ADRs) and should document product brand, batch, and full ingredient composition in every suspected case. Application of standardized causality assessment tools—such as the WHO-UMC system and the Naranjo algorithm—is essential to classify the likelihood of association. All suspected herbal-associated ADRs should be reported to the national pharmacovigilance and AYUSH-pharmacovigilance programs to improve safety signal detection and protect public health. Public awareness campaigns emphasizing the potential adverse effects of self-prescribed “natural” or “immunity-boosting” products are equally vital to ensure the safe and rational use of herbal medicines.
ACKNOWLEDGEMENT
We sincerely thank Prof. Amod Kumar Sachan (Coordinator, PvPI), Prof. Anuradha Nischal (Deputy Coordinator, PvPI), and Mr. Anoop Kumar (Research Scientist – Senior Pharmacovigilance Associate), ADR Monitoring Centre, Department of Pharmacology and Therapeutics, King George’s Medical University, Lucknow, for their invaluable guidance and support during the publication of this case report. It was an honour to work under their mentorship and benefit from their expertise.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
CONFLICT OF INTERESTS
Declared none
REFERENCES
COVID-19 Mental Disorders Collaborators. Global prevalence and burden of depressive and anxiety disorders in 204 countries and territories in 2020 due to the COVID-19 pandemic. Lancet. 2021 Nov;398(10312):1700-12. doi: 10.1016/S0140-6736(21)02143-7, PMID 34634250.
McGinty EE, Presskreischer R, Han H, Barry CL. Psychological distress and loneliness reported by US adults in 2018 and April 2020. JAMA. 2020 Jul 7;324(1):93-4. doi: 10.1001/jama.2020.9740, PMID 32492088.
Chandrasekhar K, Kapoor J, Anishetty S. A prospective randomized double-blind placebo-controlled study of safety and efficacy of a high-concentration full-spectrum extract of ashwagandha root in reducing stress and anxiety in adults. Indian J Psychol Med. 2012 Jul 1;34(3):255-62. doi: 10.4103/0253-7176.106022, PMID 23439798.
Kyrou I, Tsigos C. Chronic stress visceral obesity and gonadal dysfunction. Hormones (Athens). 2008 Oct 1;7(4):287-93. doi: 10.14310/horm.2002.1209, PMID 19121989.
Tafet GE, Nemeroff CB. The links between stress and depression: psychoneuroendocrinological, genetic and environmental interactions. J Neuropsychiatry Clin Neurosci. 2016 Apr;28(2):77-88. doi: 10.1176/appi.neuropsych.15030053, PMID 26548654.
Panossian A, Wikman G. Evidence-based efficacy of adaptogens in fatigue and molecular mechanisms related to their stress-protective activity. Curr Clin Pharmacol. 2009 Sep 1;4(3):198-219. doi: 10.2174/157488409789375311, PMID 19500070.
Kulkarni SK, Dhir A. Withania somnifera: an Indian ginseng. Prog Neuropsychopharmacol Biol Psychiatry. 2008 Jul;32(5):1093-105. doi: 10.1016/j.pnpbp.2007.09.011.
Singh G, Sharma PK, Dudhe R, Singh S. Biological activities of withaniasomnifera. Scholars Research Library Annals of Biological Research. 2010;1(3):56-63.
Mirjalili MH, Moyano E, Bonfill M, Cusido RM, Palazon J. Steroidal lactones from Withania somnifera, an ancient plant for novel medicine. Molecules. 2009 Jul 3;14(7):2373-93. doi: 10.3390/molecules14072373, PMID 19633611.
Bhattacharya SK, Bhattacharya A, Sairam K, Ghosal S. Anxiolytic-antidepressant activity of withania somnifera glycowithanolides: an experimental study. Phytomedicine. 2000 Dec;7(6):463-9. doi: 10.1016/S0944-7113(00)80030-6, PMID 11194174.
Bhattacharya SK, Muruganandam AV. Adaptogenic activity of Withaniasomnifera: an experimental study using a rat model of chronic stress. Pharmacol Biochem Behav. 2003 Jun;75(3):547-55. doi: 10.1016/s0091-3057(03)00110-2.
Udayakumar R, Kasthurirengan S, Vasudevan A, Mariashibu TS, Rayan JJ, Choi CW. Antioxidant effect of dietary supplement Withania somnifera L. reduce blood glucose levels in alloxan-induced diabetic rats. Plant Foods Hum Nutr. 2010 Jun 26;65(2):91-8. doi: 10.1007/s11130-009-0146-8, PMID 20186490.
Jahan R, Al-Nahain A, Majumder S, Rahmatullah M. Ethnopharmacological significance of Eclipta alba (L.) Hassk. (Asteraceae). Int Sch Res Not. 2014 Oct 29;2014:1-22. doi: 10.1155/2014/385969.
Sehgal VN, Verma P, Bhattacharya SN. Fixed-drug eruption caused by ashwagandha (Withania somnifera): a widely used Ayurvedic drug. Skinmed. 2012;10(1):48-9. PMID 22324179.
Kumar V, Das BK, Swain HS, Chowdhury H, Roy S, Bera AK. Immunomodulatory potency of eclipta alba (bhringaraj) leaf extract in heteropneustes fossilis against oomycete pathogen aphanomyces invadans. J Fungi (Basel). 2023 Jan 21;9(2):142. doi: 10.3390/jof9020142, PMID 36836257.
Sehgal VN, Verma P, Bhattacharya SN. Fixed-drug eruption caused by ashwagandha (Withania somnifera): a widely used Ayurvedic drug. Skinmed. 2012;10(1):48-9. PMID 22324179.
Valeyrie Allanore L, Sassolas B, Roujeau JC. Drug-induced cutaneous adverse reactions. N Engl J Med. 2020;383:2499-512.
Ekor M. The growing use of herbal medicines: issues relating to adverse reactions and challenges in monitoring safety. Front Pharmacol. 2014;4:177. doi: 10.3389/fphar.2013.00177, PMID 24454289.
Posadzki P, Watson L, Ernst E. Adverse events associated with herbal medicines: an overview of systematic reviews. Br J Clin Pharmacol. 2013;75(6):1119-32.
Kumar V, Das BK, Swain HS, Chowdhury H, Roy S, Bera AK. Immunomodulatory potency of Eclipta alba (bhringaraj) leaf extract in Heteropneustes fossilis against oomycete pathogen aphanomyces invadans. J Fungi (Basel). 2023;9(2):142. doi: 10.3390/jof9020142, PMID 36836257.
Teschke R, Eickhoff A. Ashwagandha-induced liver injury: a series of case reports and literature review. Hepatol Commun. 2023;7(10):219-25. doi: 10.1097/HC9.0000000000000270.
Mukhopadhyay AK. Adverse drug reactions to adverse drug eruptions: a brief historical account from hammurabi (C.B.C. 1810-1750) to Morrow (A.D.1846 -1913). Indian Dermatol Online J. 2020;11(5):872-3. doi: 10.4103/idoj.IDOJ_228_20, PMID 33235876.
Agarwal S, Kaur J, Singh S. Cutaneous adverse reactions associated with herbal supplements: an emerging challenge. Clin Exp Dermatol. 2021;46:1298-301.
Teschke R, Eickhoff A, Wolff A. Pharmacovigilance of herbal medicines: safety signal identification and challenges. Br J Clin Pharmacol. 2023;89:412-30.