
Int J Pharm Pharm Sci, Vol 18, Issue 2, 1-4Original Article
ANTIOXIDANT AND ANTI-INFLAMMATORY EFFECT OF GENISTEIN AGAINST PHARMACOLOGICAL EXPERIMENTAL MODELS
JHAKESHWAR PRASAD, RAJESH CHOUDHARY*, JAYA SHREE, SWARNALI DAS PAUL
Shri Shankaracharya College of Pharmaceutical Sciences, Shri Shankaracharya Professional University, Junwani-490020, Bhilai, Chhattisgarh, India
*Corresponding author: Rajesh Choudhary; *Email: rajesh080987@gmail.com
Received: 25 Nov 2025, Revised and Accepted: 26 Dec 2025
ABSTRAC
Objective: Inflammation is a biological response of vascularized tissue to harmful stimuli, including infection, damaged cells, or irritants, and is characterized by a complex, protective, non-specific response. Genistein, derived from soybeans by isoflavonoids of estrogenic and anti-inflammatory activities, recently attracted attention as an attractive drug candidate for the treatment or prevention of rheumatoid arthritis. The present investigation was carried out to explore the pharmacological role of genistein treatment in an experimentally induced inflammatory model with its antioxidant activity.
Methods: The antioxidant capacity was evaluated utilizing the 2-diphenyl-1-picrylhydrazyl (DPPH) assay method. The anti-inflammatory activity was also evaluated with the carrageenan-induced paw oedema model in Wistar rats. The research includes a normal group (vehicle control), a control group (carrageenan control), a standard group (celecoxib 50 mg/kg, p. o.), and test groups (T1: genistein 10 mg/kg, p. o., and T2: genistein 20 mg/kg, p. o.). Inflammation in experimental groups (except the normal group) was induced by a single dose injection of 0.1 ml of carrageenan (1% w/v in saline) at the subplantar region of the left hind paw after pretreatment with vehicle or drugs as per protocol. Paw edema was observed at different times after induction.
Results: The genistein showed dose-dependent antioxidant activity in the DPPH assay (inhibition of 83.63% at 100 µg/ml). In vivo, paw edema was inhibited by genistein to an extent comparable with the control group (P<0.05).
Conclusion: Genistein exerted potent anti-inflammatory and antioxidative activities in experimental models, which underlined its use as a nutraceutical for the adjuvant therapy of inflammatory diseases.
Keywords: Genistein, Antioxidant, Anti-inflammatory, Carrageenan, DPPH assay, Phytoestrogen, Nutraceutical
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijpps.2026v18i2.57618 Journal homepage: https://innovareacademics.in/journals/index.php/ijpps
INTRODUCTION
Inflammation is well-known as a nonspecific protective biological response of vascularized tissues to harmful stimuli such as pathogens, damaged cells, or irritants [1]. Inflammation may be associated with oxidative stress, and oxidative stress is implicated as one of the important mechanisms that has induced deleterious effects in different organs due to increased production of oxygen-free radicals [2]. As a process of wound healing, it is the earliest stage of tissue repair and is generally characterized by changes in blood vessels, mobilization of inflammatory cells (in particular neutrophils and macrophages), and secretion of chemical mediators that eliminate the damaging agent, remove necrotic tissue, and prepare the ground for tissue regeneration and repair. The condition affects approximately 1 percent of the global population and is more prevalent in women, who usually experience its onset between 30 and 60 years old, but has also been identified in men at a lower rate. Genetic risk factors, environmental triggers, and immune imbalance cumulatively trigger the development of the inflammatory cascades followed by the synthesis of pro-inflammatory cytokines such as TNF-α, IL1β, and IL-6 [3, 4].
Traditional anti-inflammatory agents, such as non-steroidal anti-inflammatory drugs (NSAIDs) and corticosteroids, have demonstrated efficacy but are hampered by clinically important adverse effects and therapeutic limitations. Adverse effects of NSAIDs include gastrointestinal bleeding, kidney injury, and cardiovascular risk; and during corticosteroid therapy, there is suppression of immunity, delayed wound healing, and systemic complications, including osteoporosis and adrenal suppression. In addition to this, long use of these medications can make them less effective while also more addictive, making it imperative for alternative treatments to be safer and more target-specific in the clinical setting [5]. This lack of effective therapy has resulted in an aggressive search for alternative agents that match or exceed the efficacy and safety profile of current treatments. Among natural product-derived agents, genistein, a phytoestrogenic isoflavone abundant in soybeans, has received much medicinal attention owing to its broad pharmacological effects as an anti-inflammatory, antioxidant, immunomodulatory, and bone-protective agent.
Genistein (4′, 5, 7-trihydroxyisoflavone) is a 17β-estradiol-mimetic compound that exhibits estrogenic activity by binding to the estrogen receptor (ER), particularly ERβ [6]. This interaction is the foundation of its immunomodulatory and bone-maintenance role, including in inflammatory disease. Besides its hormonal mimicry activity, genistein is also a tyrosine kinase inhibitor, suggesting that it might be exerting influence on the network of signaling pathways leading to the inflammatory and proliferative events associated with inflammation through molecules such as NF-κB, MAPKs, and JAK/STAT systems [7, 8].
In this regard, the development of treatment strategies based on functional foods and plant-derived substances has displayed an increasing trend in recent years. Soybean-based diets, which are isoflavone-like and genistein-rich, are associated with a lower incidence of degenerative and inflammatory diseases, including osteoporosis or rheumatoid arthritis. With the two-sided face of food and pharmacology, genistein is an excellent candidate to combine nutritional and treatments for inflammation [9].
MATERIALS AND METHODS
Chemicals and reagents
Ascorbic acid (Loba Chemie, Mumbai, Maharashtra, India), genistein (Tokyo Chemical Industries, Chennai, India), carrageenan (Loba Chemie, Mumbai, Maharashtra, India), and 2,2-diphenyl-1-picrylhydrazyl (HiMedia Laboratories Pvt. Ltd., Maharashtra, India) were procured from a departmental store. All chemicals and reagents employed in the study were of analytical grade.
DPPH assay
To determine DPPH radical scavenging activity, an in vitro standard antioxidant assay is used, with DPPH as a stable free radical. A stock solution of genistein is prepared in methanol at concentrations of 10-100 µg/ml. A 0.1 mmol methanolic solution of DPPH is made freshly and kept in the dark. A quantity of genistein solution is combined with an equal volume of DPPH solution in every test tube, followed by 30-minute incubation at room temperature (37 °C) in the dark for reaction. The absorbance is recorded at 515 nm using a UV-Vis spectrophotometer (UV-1780, Shimadzu, USA), against a blank. The maximum absorbance is established by a control that contains DPPH but with no genistein [10]. The percentage of inhibition against DPPH radicals was calculated as follows:

The value of Acontrol is the absorption of the DPPH solution without genistein, while Asample is the absorbance with genistein. This method allows us to find out the way genistein may combat free radicals by measuring how effectively it is doing so.
Experimental animals
Albino Wistar rats (150-200 g) of both sexes were procured from the Animal House facility, an institutional source, Shri Shankaracharya Professional University, Bhilai (C. G.), India. The protocol was reviewed by the Institution Animal Ethics Committee (IAEC) and approved in the meeting with approval no. SSPU/KIPS/IAEC/2024/007. Animals were acclimatized for 2 w before the experiments. They were housed on husk bedding in polypropylene cages. Aqua Guard water and a pellet diet were kept available to the animals. Animals were kept in standard environmental conditions, such as a 12:12 light/dark cycle with 60% relative humidity, as per CCSEA guidelines [11].
Experimental protocol
For the study design, an equal number of 30 animals is assigned to five experimental groups (n = 6 in each). The normal group was injected with saline (0.1 ml) at the sub-plantar region of the left hind paw, followed by a one-hour treatment of saline (10 ml/kg, p. o.). The control group was injected with 0.1 ml carrageenan (1% w/v in saline) in the subplantar region of the left hind paw and treated for one hour with saline (10 ml/kg, p. o.). The treatment groups received celecoxib 50 mg/kg, p. o. (standard group); genistein 10 mg/kg, p. o. (T1 group); and genistein 20 mg/kg, p. o. (T2 group). One hour after these treatments, animals received 0.1 ml carrageenan (1% w/v in saline) in the sub-plantar region of the left hind paw to induce inflammation [12-13]. The volume of the paw was measured at regular intervals post-injection, e. g., 0, 1, 2, 3, 4, and 24 h, using a vernier caliper. Paw volume elevation was computed as an indication of edema, and treatment groups were compared with the control group to assess the anti-inflammatory activity. The approach is reliable to assess the anti-inflammatory properties of pharmacological and biological compounds, as evidenced by significant inhibition of carrageenan-induced edema formation.
Statistical analysis
Two-way analysis of variance (ANOVA) followed by a post-hoc comparison with Bonferroni’s test was employed to analyze the data. Results were shown as mean±SEM, with p<0.05 being regarded as a significant difference. All calculations were performed using GraphPad Prism statistical software to ensure the reliability and reproducibility of experimental data.
RESULTS AND DISCUSSION
DPPH assay for genistein
Table 1 shows the DPPH radical scavenging activities of genistein in comparison with those of the standard antioxidant, ascorbic acid, at a concentration range from 10 to 100 µg/ml. In both compounds, there is a dose-dependent increase in values in % inhibition, with an increasingly stronger neutralization of free radical activity observed as the concentration increases. The point of 50% inhibition in the presence of genistein is found at a concentration of 41.59 µg/ml, and at a value of 26.97 µg/ml for ascorbic acid, but it does not exceed its maximum level. The mean±SEM (n = 3) data confirm the reproducibility of the measurements and evidence the importance of genistein as a modulator of oxidative stress, especially in products that demand milder antioxidant findings.
Table 1: DPPH radical scavenging activity (% Inhibition)
| Concentration (µg/ml) | Genistein (% inhibition) | Ascorbic acid (% inhibition) |
| 10 | 19.83±0.72 | 26.93±0.29 |
| 20 | 36.40±0.66 | 46.86±0.85 |
| 40 | 56.53±0.48 | 67.43±0.56 |
| 60 | 66.96±0.60 | 80.76±0.64 |
| 80 | 78.10±0.26 | 92.86±0.31 |
| 100 | 83.63±0.46 | 96.17±0.88 |
Value are expressed as mean±standard error of the mean (n = 3 replicates).
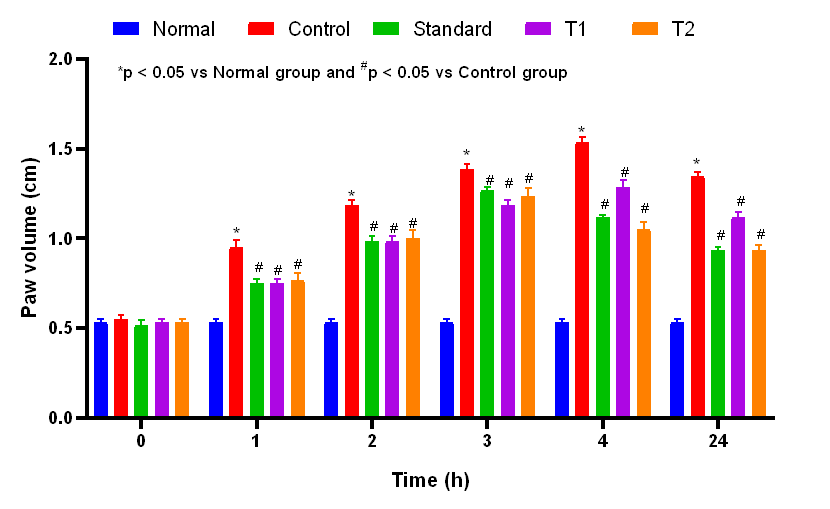
Fig. 1: Effect of genistein on the left hind paw volume of carrageenan-induced inflammatory rats. Data represented as mean±SEM; *p<0.05 vs. normal group; #p<0.05 vs. control group (two-way ANOVA followed by Bonferroni’s post hoc test). Normal group (saline only), control group (carrageenan+vehicle), standard group (carrageenan+celecoxib 50 mg/kg), test group 1 (carrageenan+genistein 10 mg/kg), and test group 2 (carrageenan+genistein 20 mg/kg)
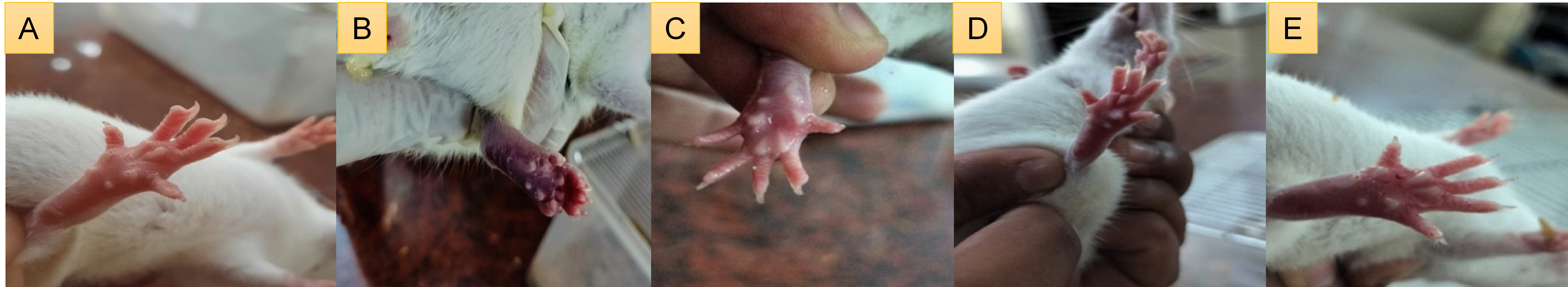
Fig. 2: Diagrammatic representation of rat paws 4 h after carrageenan injection. (A) Normal group (saline only); (B) Control group (carrageenan+vehicle); (C) Standard group (carrageenan+Celecoxib 50 mg/kg); (D) Test group-1 (carrageenan+Genistein 10 mg/kg); (E) Test group-2 (carrageenan+Genistein 20 mg/kg). Visual reduction in edema is evident in groups C, D, and E compared to group B
Anti-inflammatory activity of genistein in experimental animals
Clock time of recordings was at 0, 1, 2, 3, 4, and the following day (24 h) thereafter in five groups: Normal, Control, Standard T1, and T2. All tested groups presented similar paw volume (~0.533 to 0.550 cm) as inflammatory effusion at the baseline time (0 h). After carrageenan injection, the control group revealed a rise in edema over time until 4 h (1.533±0.033 cm) to confirm the inflammatory reaction process (fig. 1). On the other hand, the standard group (probably treated with a known anti-inflammatory drug) showed an evident decrease in edema and values that were also below those of the control, especially at 3 h and 4 h (1.266±0.021 and 1.116±0.016 cm, respectively). In T2, the attenuation of inflammation was slightly more pronounced compared to T1 at 4 h (1.050±0.042 cm versus T1: 1.283±0.040 cm). After 24 h, the standard (0.93±0.021 cm) and treatment groups (T1: 1.11±0.031 cm and T2: 0.93±0.033 cm) significantly (p<0.05) reduced paw edema compared to the control group (1.35±0.022 cm), indicating a continued anti-inflammatory effect. Fig. 2 shows the paw edema of the experimental pharmacological assay for anti-inflammatory activity using carrageenan-induced paw edema in rats. A stable sample size (n=6) and SEM between groups support the consistency of the data. Consequently, it is evident that both the tested treatments have significant anti-inflammatory activity and that T2 shows equivalent potency when compared with the standard drug.
DISCUSSION
Genistein has antioxidant activity observed in the DPPH radical scavenging assay. Improved free radicals’ neutralization (83.63% inhibition at 100 µg/ml). Since ascorbic acid exhibits stronger activity with an inhibition of 96.17%, the performance is more attractive, while bi-functional use in a diet and pharmacological function has potential for genistein. These findings are in congruence with earlier studies by Jomova and Alomar (2024) that showed the scavenging activity of genistein against ROS and its ability to up-regulate endogenous antioxidant enzymes, including SOD and CAT, which protected bone and cartilage tissues from oxidative injury [14]. Oxidative stress is an accepted contributor to the pathogenesis of inflammation, contributing to synovial hyperplasia, chondrocyte apoptosis, and matrix degradation. By lowering oxidative stress, genistein would potentially prevent joint structures and inflammation from damage.
In vivo, genistein showed a marked anti-inflammatory effect in the carrageenan-induced paw oedema test, a classic assay to evaluate acute inflammation [7]. The 20 mg/kg (T2) showed a significantly reduced paw volume as compared with the control group and a similar active reference anti-inflammatory drug, celecoxib. This is particularly worthy of note as concerns continue to be expressed over the gastrointestinal, renal, and cardiovascular effects of long-term NSAID use [15]. The anti-inflammatory activity of genistein is also evidenced in Zhang et al. (2012), who demonstrated that genistein was a potentiating factor for the treatment of inflammatory conditions like RA by suppressing the secretion of fibroblast-like synoviocyte (FLS) exosomes through regulation of the Rab27/nSMase2/Mfge8 axis [16]. This pathway is essential for the transmission of inflammatory signals and synovial hyperplasia, which are key in RA pathogenesis. Additional clarification about the mechanism underlying genistein’s anti-inflammatory effect has been demonstrated by Cheng et al. (2019), who reported that genistein inhibits angiogenesis in RA by blocking the IL-6/JAK2/STAT3/VEGF signal cascade [17]. The present study adds to this emerging picture by presenting strong evidence of genistein’s anti-inflammatory and antioxidant actions in a widely used animal model of acute inflammation. The observed paw volume reduction, especially at 20 mg/kg.
CONCLUSION
In conclusion, this study confirmed the pharmaceutical potential of genistein as both a dual-action antioxidant and anti-inflammatory in the rat model. It has prominent attenuation of carrageenan-induced paw edema and dose-dependent free radical scavenging activity. The study confirmed its anti-inflammatory activity at a dose of 20 mg/kg was robust compared to celecoxib, thus proposing it further as a nutraceutical co-adjuvant with low toxicity. These results provide a basis for the inclusion of genistein in autoimmune therapy, especially as an alternative plant-derived medicine to current medication for patients who demand safer remedies. In the future, more investigations need to be performed focusing on the improvement of bioavailability of genistein using novel delivery technologies such as nanoparticles, liposomes, and phytosomes, or others, to resolve its pharmacokinetic limitations. Elucidation of its mechanisms for modulating inflammatory signaling pathways, particularly NF-κB, JAK/STAT, and MAPK, could have further implications in understanding the immune regulatory functions. Clinical studies are needed to confirm the efficacy and safety in human patients, ideally stratified by estrogen receptor profiles for improved therapeutic outcomes.
FUNDING
No funding was received
AUTHORS CONTRIBUTIONS
Jhakeshwar Prashad was involved in investigation, data collection, and manuscript writing. Rajesh Choudhary was involved in conceptualization, methodology, supervision, and manuscript review and editing. Jaya Shree was involved in data analysis and manuscript writing, and Swarnali Das Paul was involved in conceptualization and supervision.
CONFLICT OF INTERESTS
The authors declared no conflict of interest
REFERENCES
Jahid M, Khan KU, Rehan-Ul-Haq ARS. Overview of rheumatoid arthritis and scientific understanding of the disease. Mediterr J Rheumatol. 2023 Aug;34(3):284-91. doi: 10.31138/mjr.20230801.oo, PMID 37941854.
Divya J, James Jainey P, Sneh P. Development and optimization of polymeric nanoparticles of glycyrrhizin: physicochemical characterization and antioxidant activity. Int J Appl Pharm. 2024 Jan;16(1):166-71. doi: 10.22159/ijap.2024v16i1.49164.
Shah P, Trivedi J, Shah P. Pathophysiology of rheumatoid arthritis. ICCRR. 2024 Jun 12;2(1):1-4. doi: 10.59657/2993-0855.brs.24.010.
Franco BB, Agilandeswari P, Karthik L. Computational screening of potent anti-inflammatory compounds for human mitogen-activated protein kinase: a comprehensive and combined in silico approach. Int J Curr Pharm Sci. 2024 Nov;16(6):21-32. doi: 10.22159/ijcpr.2024v16i6.6023.
Padjen I, Crnogaj MR, Anic B. Conventional disease-modifying agents in rheumatoid arthritis a review of their current use and role in treatment algorithms. Reumatologia. 2020 Dec;58(6):390-400. doi: 10.5114/reum.2020.101400, PMID 33456082.
Sharifi Rad J, Quispe C, Imran M, Rauf A, Nadeem M, Gondal TA. Genistein: an integrative overview of its mode of action pharmacological properties and health benefits. Oxid Med Cell Longev. 2021 Jul;2021:3268136. doi: 10.1155/2021/3268136, PMID 34336089.
Goh YX, Jalil J, Lam KW, Husain K, Premakumar CM. Genistein: a review on its anti-inflammatory properties. Front Pharmacol. 2022 Jan;13:820969. doi: 10.3389/fphar.2022.820969, PMID 35140617.
Duan W, Kuo IC, Selvarajan S, Chua KY, Bay BH, Wong WS. Antiinflammatory effects of genistein a tyrosine kinase inhibitor on a guinea pig model of asthma. Am J Respir Crit Care Med. 2003 Feb;167(2):185-92. doi: 10.1164/rccm.200205-420OC, PMID 12406820.
Soyata A, Hasanah AN, Rusdiana T. Isoflavones in soybean as a daily nutrient: the mechanisms of action and how they alter the pharmacokinetics of drugs. Turk J Pharm Sci. 2021 Dec 31;18(6):799-810. doi: 10.4274/tjps.galenos.2020.79106, PMID 34979737.
Suliman MN, Hamad AI, Ibrahim F. Valorization of antioxidant anticancer activities and phytochemical analysis of ethanolic extract of Euphorbia cuneata Vahl. and its effect on the expression of P53, RAF-1 and CASP3 genes. Asian J Pharm Clin Res. 2025 Mar;18(3):46-52. doi: 10.22159/ajpcr.2025v18i3.53908.
Devaki K, Beulah U, Akila G, Gopalakrishnan VK. Effect of aqueous extract of Passiflora edulis on biochemical and hematological parameters of Wistar albino rats. Toxicol Int. 2012 Jan;19(1):63-7. doi: 10.4103/0971-6580.94508, PMID 22736906.
Hafeez A, Jain U, Sajwan P, Srivastava S, Thakur A. Evaluation of carrageenan-induced anti-inflammatory activity of ethanolic extract of bark of Ficus virens Linn. in swiss albino mice. J Phytopharmacol. 2013;2(1-3):39-43. doi: 10.31254/phyto.2013.21307.
Dzoyem JP, McGaw LJ, Kuete V, Bakowsky U. Chapter 9 Anti-inflammatory and anti-nociceptive activities of African medicinal spices and vegetables. In: Academic Press, editor. Medicinal spices and vegetables from Africa. Amsterdam: Elsevier; 2017. p. 239-70. doi: 10.1016/B978-0-12-809286-6.00009-1.
Jomova K, Alomar SY, Alwasel SH, Nepovimova E, Kuca K, Valko M. Several lines of antioxidant defense against oxidative stress: antioxidant enzymes, nanomaterials with multiple enzyme-mimicking activities and low-molecular-weight antioxidants. Arch Toxicol. 2024 May;98(5):1323-67. doi: 10.1007/s00204-024-03696-4, PMID 38483584.
Sohail R, Mathew M, Patel KK, Reddy SA, Haider Z, Naria M. Effects of non-steroidal anti-inflammatory drugs (NSAIDs) and gastroprotective NSAIDs on the gastrointestinal tract: a narrative review. Cureus. 2023 Apr 3;15(4):e37080. doi: 10.7759/cureus.37080, PMID 37153279.
Zhang Y, Dong J, He P, Li W, Zhang Q, Li N. Genistein inhibit cytokines or growth factor-induced proliferation and transformation phenotype in fibroblast-like synoviocytes of rheumatoid arthritis. Inflammation. 2012 Feb;35(1):377-87. doi: 10.1007/s10753-011-9365-x, PMID 21792602.
Cheng WX, Huang H, Chen JH, Zhang TT, Zhu GY, Zheng ZT. Genistein inhibits angiogenesis developed during rheumatoid arthritis through the IL-6/JAK2/STAT3/VEGF signaling pathway. J Orthop Translat. 2020 May;22:92–100. doi: 10.1016/j. jot.2019.07.007.