
Int J Pharm Pharm Sci, Vol 18, Issue 3, 1-7Review Article
ARTIFICIAL INTELLIGENCE IN PHARMACEUTICAL REGULATORY AFFAIRS: REVOLUTIONIZING DRUG DEVELOPMENT, SUBMISSIONS AND REGULATORY COMPLIANCE
PATHIPATI SUNITHA, R. S. SAILESH, S. NAGALAKSHMI*
1Department of Pharmaceutics, Sri Ramachandra Faculty of Pharmacy, Sri Ramachandra Institute of Higher Education and Research (Deemed to be University), Porur, Chennai-600116, Tamil Nadu, India
*Corresponding author: S. Nagalakshmi; *Email: nagalakshmi.s@sriramachandra.edu.in
Received: 04 Dec 2025, Revised and Accepted: 22 Jan 2026
ABSTRAC
Objective of this review was to evaluate the evolution, applications, and regulatory implications of artificial intelligence technologies in pharmaceutical regulatory affairs, with emphasis on their influence on drug development, regulatory submissions, and global harmonization. This review examines published literature, regulatory authority documents, and international reports describing the integration of artificial intelligence into regulatory processes. Key technological approaches, including machine learning, natural language processing, and automated workflow systems, were analyzed for their contribution to data assessment, documentation, decision support, and pharmacovigilance activities. Challenges related to data integrity, ethical responsibility, transparency, system validation, and differences in global regulatory expectations were also assessed. Artificial intelligence was found to significantly improve regulatory efficiency by enabling rapid data processing, structured documentation, and predictive regulatory analysis that reduces manual workload and strengthens decision accuracy. Regulatory authorities in multiple regions have begun adopting advanced digital frameworks that support standardized data exchange, collaborative review mechanisms, and more robust monitoring of drug safety. Despite these advancements, concerns remain regarding fairness of algorithms, reliability of automated outputs, protection of confidential information, and the absence of unified international standards. Artificial intelligence technologies provide strong opportunities to streamline regulatory pathways, improve the consistency and reliability of regulatory decisions, and support broader global harmonization. Successful implementation requires proactive attention to ethical, technical, and regulatory challenges to ensure transparent, responsible, and effective integration within pharmaceutical regulatory affairs.
Keywords: Artificial intelligence, Pharmacovigilance, Global regulatory harmonization, Regulatory submissions, Pharmaceutical regulatory affairs
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijpps.2026v18i3.57700 Journal homepage: https://innovareacademics.in/journals/index.php/ijpps
INTRODUCTION
Pharmaceutical regulatory affairs (PRA) ensures that medications meet predetermined criteria for efficacy, safety, and quality throughout their lifecycle [1]. Events such as the thalidomide incident emphasized the need for robust regulatory frameworks to protect patients and support timely access to effective treatments [2]. Despite advancements in drug development, the process remains time-consuming and resource-intensive, requiring 12–15 y and costing USD 1–2.6 billion per approved product. Procedural inefficiencies continue to delay patient access to essential medications [3, 4]. Several international initiatives aim to address these constraints, including the AI-driven Clinical Evaluation and Safety Studies (ACCESS) Consortium, Project Orbis, patient-centric review approaches, and the use of real-world evidence (RWE) to improve regulatory performance and accelerate approvals [1, 5]. Regulatory dossiers have grown in size and complexity due to the increasing focus on biologics, precision medicine, and omics-based technologies, creating new challenges for global health authorities [4, 6].
Artificial intelligence (AI), which includes machine learning (ML) and deep learning (DL), is one of the emerging technologies that presents encouraging prospects for increasing regulatory efficiency [3, 6, 7]. AI can assist with a variety of regulatory tasks, including regulatory intelligence gathering, dossier preparation, submission management, and pharmacovigilance (PV), through automation, natural language processing (NLP), and predictive analytics [8, 9]. Agencies such as the European Medicines Agency (EMA), China's National Medical Products Administration (NMPA), and the U. S. Food and Drug Administration (FDA) have initiated pilot programs using these tools to modernize review processes and reduce compliance risks [6, 10]. However, important concerns remain regarding algorithmic explainability, data privacy, and global regulatory harmonization [4, 11]. Therefore, establishing transparent governance models, ethical oversight, and a digitally skilled workforce is essential to realizing the full potential of AI in PRA. These elements are crucial for enhancing patient access, speeding up approvals, and bolstering international regulatory compliance. This review was conducted through a structured literature search using PubMed, Scopus, Web of Science, and Google Scholar databases. Keywords such as artificial intelligence, pharmaceutical regulatory affairs, regulatory submissions, pharmacovigilance, machine learning, natural language processing, and digital regulatory systems were used individually and in combination. Literature published between 2014 and 2025 was prioritized to ensure contemporary relevance. In addition, regulatory guidelines, policy documents, and technical reports from the U. S. Food and Drug Administration (FDA), European Medicines Agency (EMA), International Council for Harmonisation (ICH), and World Health Organization (WHO) were reviewed.
AI’S Historical evolution
The development of AI can be divided into four major phases. The foundational era (1940s–1950s) began when Alan Turing posed the question, “Can machines think?” During this period, the theoretical foundations of AI were established, and the term “artificial intelligence” was formally introduced at the 1956 Dartmouth Conference. The next significant phase, known as the expert systems era (1960s–1980s), focused on building rule-based programs such as DENDRAL and MYCIN. Although these systems demonstrated early promise in problem-solving, their performance was limited by the computing power available at that time. The machine learning era (1980s–2000s) followed, during which AI progressed beyond fixed rules. Techniques such as early neural networks, decision trees, and support vector machines enabled computers to learn from data instead of relying solely on predefined instructions. This marked the beginning of data-driven classification and prediction. The most recent era-deep learning and generative AI-began in the 2000s. Advancements in large datasets, high-performance graphics processing units (GPUs), and architectures such as convolutional neural networks and transformer models have driven rapid progress. These developments led to modern innovations like AlphaGo and ChatGPT, which now influence diverse areas including regulatory science and healthcare [6].
Historical evolution of regulatory submissions
The regulatory submission process has undergone significant changes over the past few decades, transitioning from paper-based dossiers to fully digital and structured systems. The demand for faster drug approvals and the need for global harmonization have been major drivers of this shift. Early efforts to digitize regulatory workflows produced several pioneering systems, including the U. S. FDA’s Computer-Assisted New Drug Application (CANDA, 1985), Europe’s Drug Application Management and Organization System (DAMOS, 1989), and France’s Système d’Évaluation et de Documentation des Autorisations de Mise sur le Marché (SEDAMM, 1993). Although these platforms improved data management, they lacked interoperability across regulatory agencies and did not follow standardized formatting [12]. Collaborative initiatives, such as the Medical Electronic Review System (MERS, 1994) and the Management of New Submissions and Evaluations (MANSEV, 1997), further highlighted the importance of standardization. A major breakthrough occurred in 2003 with the introduction of the electronic Common Technical Document (eCTD), which established a structured, five-module XML-based format for submissions. After the U. S. FDA made eCTD mandatory in 2008, it rapidly became the global standard for regulatory communication [12, 13].

Fig. 1: Digital milestones in global regulatory submissions.
Key milestones in the evolution of electronic regulatory submissions, including the adoption of eCTD and transition from paper-based dossiers, synthesized from regulatory agency reports and published literature [1, 12, 13] (Created by authors)
To support this transition, the International Council for Harmonisation (ICH) introduced three key guidelines: M2 (electronic communication), M4 (CTD structure), and M8 (eCTD specifications). These guidelines reduced redundant assessments across agencies and accelerated the overall review process [13]. Despite these advances, several challenges remain, including fragmented data systems, time-consuming procedures, and country-specific submission requirements that complicate international coordination [14]. The FDA’s Regulated Product Submission (RPS) framework serves as the foundation for the latest development, eCTD Version 4.0. Its objectives include improving system interoperability, enabling two-way communication between applicants and regulators, and enhancing cross-referencing of previous submissions. Complementary International Council for Harmonisation (ICH) guidelines such as M4, E6 (R2), and Q8–Q11, along with U. S. regulatory pathways including the Investigational New Drug (IND), New Drug Application (NDA), and Abbreviated New Drug Application (ANDA), continue to reinforce the goal of a systematic, efficient, and globally harmonized review process [13].
AI in regulatory affairs
AI is transforming the pharmaceutical regulatory affairs landscape by automating complex processes, improving data analysis, and increasing submission efficiency. Intelligent systems are increasingly used for document preparation, data evaluation, regulatory tracking, and submission forecasting. For example, Amgen’s Quality Overall Summary (QOS) tool uses large language models to reduce Module 3 preparation time by more than 60%. Similarly, generative AI technologies can now create regulatory dossiers customized to regional requirements, reducing manual rework and strengthening global compliance. Predictive compliance algorithms are gaining traction for enhancing lifecycle management. These tools analyze historical data to identify potential regulatory risks before submission. Cloud-based platforms that support real-time data exchange and reliance-based review-such as Roche’s PAC Reliance Pilot and Accumulus Synergy-are redefining regulatory collaboration. International data standards, including those from Pharmaceutical Quality/Chemistry, Manufacturing, and Controls (PQ/CMC), the Society for Clinical Data Management (SCDM), ISO’s Identification of Medicinal Products (IDMP), and Health Level Seven’s Fast Healthcare Interoperability Resources (HL7-FHIR), further strengthen these efforts. Together, these frameworks enable modular submissions, faster approvals, and efficient data sharing, providing significant benefits for low-and middle-income countries (LMICs) [14]. The integration of AI, standardized data formats, and collaborative digital systems is transforming the regulatory submission process into a flexible, data-driven ecosystem. This shift supports faster approvals, improved compliance, and enhanced patient access to innovative therapies.
AI technologies shaping pharmaceutical regulatory affairs
Core AI technologies in regulatory affairs
AI, ML, DL, and advanced computational models are key technologies transforming regulatory operations. These tools support continuous compliance monitoring, predictive modeling, and workflow automation. In particular, automated text summarization and NLP have increased the efficiency of document review and pharmacovigilance workflows [15-17]. ML models use clinical trial registries and electronic health records to support risk profiling, classification, and predictive analytics, although challenges such as “black-box” decision-making and biased datasets remain [18, 19].
Supporting digital infrastructure for AI-enabled regulatory systems
Additionally, robotic process automation (RPA) reduces manual, repetitive tasks, including data entry, formatting, and submission organization, especially when integrated with blockchain and AI technologies [20-23]. The five Vs of big data-volume, velocity, variety, veracity, and value, enable the rapid analysis of large and complex datasets, supporting regulatory compliance and drug safety [24]. Cloud computing and blockchain further provide secure, traceable data management, support remote collaboration across global teams, and allow scalable submissions [25].
These technological advancements are shifting regulatory affairs from inflexible, document-heavy systems to adaptable, data-driven frameworks that strengthen patient safety, compliance, and transparency.
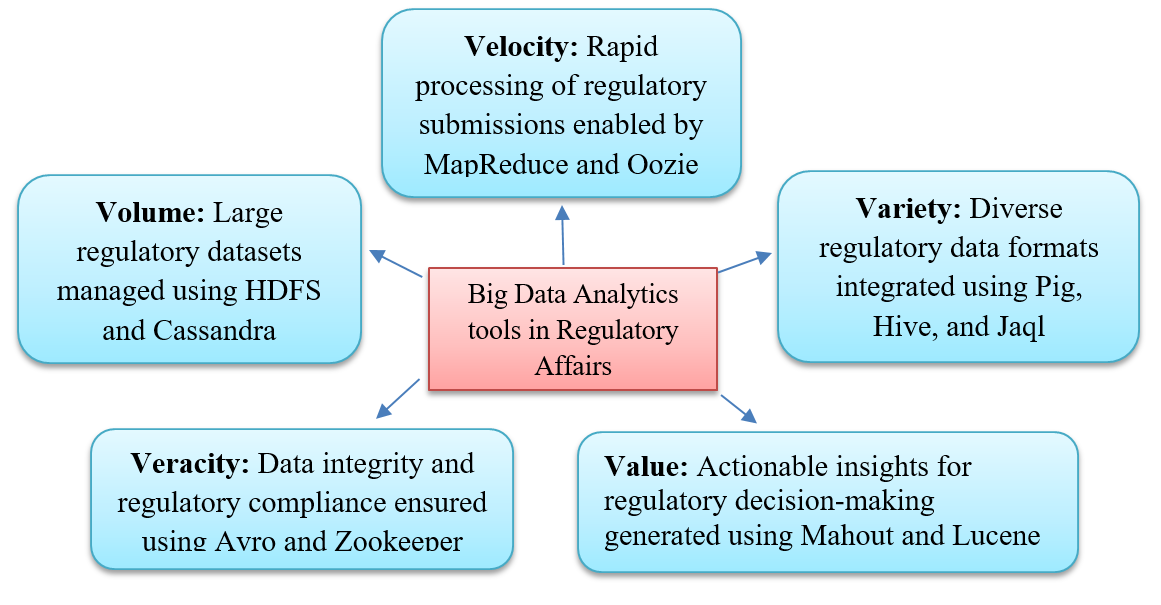
Fig. 2: Big data analytics tools in pharmaceutical regulatory affairs.
This fig. illustrates the five dimensions of big data-volume, velocity, variety, veracity, and value. These tools support regulatory submissions, pharmacovigilance signal detection, and compliance analytics (Created by authors)
AI in drug development–regulatory implications
AI is also reshaping drug development by accelerating discovery, improving clinical trials, and supporting faster regulatory approval. The adoption of artificial intelligence in healthcare and pharmaceutical regulatory systems has expanded rapidly in recent years due to increasing data complexity, demand for efficient decision-making, and digital transformation initiatives across regulatory agencies [3, 6, 11]. Conventional drug development typically requires 10–15 y and over USD 2 billion, with fewer than 12% of Phase I candidates reaching the market. In contrast, AI-enabled approaches have been shown to improve operational efficiency, reduce manual workload, and accelerate evidence generation and regulatory decision-making across the drug development and regulatory continuum [26, 27]. Integrating AI across the drug development and regulatory continuum, from target identification and preclinical design to dossier generation, enables pharmaceutical companies and regulators to promote faster, more transparent, and patient-focused innovation. Digital twins, DL, and ML technologies enhance biomarker discovery, dose selection, and patient stratification. Regulatory initiatives such as the FDA’s Physiologically-Based Pharmacokinetic (PBPK) Modeling Pilot under ICH M15, which has demonstrated the potential to optimize clinical trial design and reduce unnecessary patient exposure, demonstrate increasing regulatory openness to AI-based tools. Adoption is further supported by frameworks including the EMA Guidelines for Trustworthy AI, the FDA’s Real-World Evidence (RWE) Program, and international projects such as Consolidated Standards of Reporting Trials–Artificial Intelligence (CONSORT-AI), Standard Protocol Items: Recommendations for Interventional Trials–Artificial Intelligence (SPIRIT-AI), and the ACCESS Consortium. NLP contributes to improved Good Manufacturing Practice (GMP) adherence through predictive monitoring and supports efficient detection and assessment of adverse events in pharmacovigilance systems [26]. However, concerns related to ethical risks, inconsistent standards, and the opacity of “black-box” models underscore the need for standardized, transparent, and risk-based frameworks to support global integration [27]. Throughout the entire medication lifecycle, AI enhances safety, compliance, and predictive capabilities. Table 1 summarizes key AI techniques and their applications in pharmaceutical regulatory science [26].
Table 1: AI Tools across the drug lifecycle [26]
| Stage | AI Tools/Models | Applications |
| Preclinical | QSAR (DEREK, TOPKAT), PBPK (SimCYP, Gastro Plus) | Toxicity prediction, dose optimization |
| Clinical | ML trial prediction models, Digital twins, IoT sensors | Recruitment, trial simulation, monitoring |
| Regulatory Submissions | NLP (Synchrogenix), RPA (UiPath) | Dossier preparation, Automated gap analysis, consistency checks, and regulatory validation |
| Postmarket | WEB-RADR project tools, HSA web crawlers, ML ADR models | Pharmacovigilance, safety signal detection |
| Manufacturing | ANN models | Formulation optimization, quality control |
| Repurposing | ML-based collaborative filtering, EHR mining | New indications for existing drugs |
This table summarizes representative artificial intelligence tools and their applications in pharmaceutical regulatory affairs and is not intended to be exhaustive
Abbreviations: QSAR–Quantitative Structure–Activity Relationship; DEREK–Deductive Estimation of Risk from Existing Knowledge; TOPKAT–Toxicity Prediction by Komputer Assisted Technology; SimCYP–Simulation of Clinical Pharmacokinetics; GastroPlus–GastroPlus PBPK Modeling and Simulation Software; IoT–Internet of Things; Synchrogenix–Synchrogenix Regulatory Solutions (Certara); UiPath–UiPath Automation Platform; WEB-RADR–Web-Recognizing Adverse Drug Reactions; HSA–Health Sciences Authority; ANN–Artificial Neural Network.
AI in dossier preparation and submission
To modernize regulatory submissions, the biopharmaceutical industry increasingly uses cloud technologies, structured content and data management (SCDM), and artificial intelligence (AI). Standardized models such as ISO’s Identification of Medicinal Products (IDMP), PQ/CMC, and HL7-FHIR facilitate harmonized international data exchange. AI technologies including ML, NLP, RPA, predictive analytics, and large language models (LLMs)-enable automated lifecycle tracking, compliance checks, and metadata tagging. Companies such as Novartis and Pfizer have reported the use of artificial intelligence–enabled regulatory intelligence and predictive analytics to manage evolving regulatory requirements, while Roche has implemented an LLM-assisted Quality Overall Summary (QOS) authoring tool that reduces Module 3 preparation time by more than 60% [14, 28]. Cloud-based solutions, including Accumulus Synergy and Roche’s PAC Reliance Pilot, support real-time, multi-tenant submissions and reliance-based approvals, offering particular benefits for LMICs. Despite these advancements, challenges related to data security, ethics, standardization, and infrastructure persist, requiring phased and regulatory-compliant adoption [14, 28].
AI in compliance and post-market surveillance
AI enhances post-market surveillance (PMS) in addition to traditional systems such as Med Watch (FDA’s adverse event reporting program), Eudra Vigilance (EMA’s safety monitoring database), and Periodic Safety Update Reports (PSURs) [29]. AI tools-including ML, NLP, deep learning, and computer vision-automate analysis, detect anomalies, and enable proactive risk assessment. Although the study by Reniewicz et al. focuses on in vitro diagnostic assays, the artificial intelligence and machine-learning methodologies applied to post-market surveillance-such as automated signal detection, risk classification, and safety monitoring-are directly transferable to pharmaceutical pharmacovigilance systems and regulatory safety assessment frameworks [30]. By integrating data from wearables, social media, electronic health records (EHRs), and adverse event (AE) reporting systems, AI supports real-time monitoring and early signal detection. PV platforms such as the Sentinel System, Information Visualization Platform (InfoViP), Infer BERT (an AI-based inference tool) and FAERS classifiers strengthen safety evaluation. International tools-including WHO Drug Koda (WHO drug classification tool), Sweden's PhaVAI (AI-driven pharmacovigilance system), and Swiss medic’s LiSA (safety assessment platform), highlight global adoption [6, 29]. AI-enabled platforms like Huma. AI, an AI-driven pharmacovigilance tool, significantly reduce review timelines-from weeks to hours-and increase detection accuracy by up to tenfold. However, challenges persist regarding algorithmic bias, privacy, interpretability, and data quality. Future developments will center on risk-based frameworks for global patient safety, automated pharmacovigilance, and integration with the AI-Internet of Things (AI–IoT) [29].
Global regulatory landscape of AI in pharmaceutical regulatory affairs
Global regulatory frameworks are progressively incorporating tools such as AI and ML to enhance productivity, decision-making, and patient safety [6, 30].
United States
The USFDA integrates AI through initiatives such as the IT Strategy 2024–2027; TMAP, DMAP, EMAP, and CMAP (Therapeutic, Device, Evaluation, and Compliance Mapping Programs) for regulatory modernization; ISTAND for AI and non-animal testing methodologies; BEST (Biologics Effectiveness and Safety) for adverse event monitoring; and Sentinel for post-market surveillance. These programs support applications such as dose optimization, clinical trial design, regulatory document development, and ML-enabled device oversight [30].
Europe
Through the Big Data Workplan, the Multi-Annual AI Workplan, and Regulatory Science 2025, the EMA and national regulatory organizations, such as the Federal Institute for Drugs and Medical Devices (BfArM), the Paul Ehrlich Institute, and Swiss medic, actively promote the implementation of artificial intelligence (AI). A risk-based framework has been established under the European Union’s (EU) AI Act:
Unacceptable risk: Applications that are prohibited.
Low/minimal risk: Optional guidance with post-market monitoring.
High risk: Subject to stringent compliance and conformity evaluation [2].
Asia and beyond
Japan’s Pharmaceuticals and Medical Devices Agency (PMDA) promotes AI research but lacks a unified regulatory framework. China’s NMPA incorporates AI, big data, blockchain, and IoT across the drug lifecycle [30]. African and international organizations aim for harmonization through ICH M15, CONSORT-AI, SPIRIT-AI, and the EMA Ethics Guidelines [26, 30].
Workshops and industry examples
AI applications in pharmacovigilance, production, development, and drug discovery were highlighted at the 2024 FDA–Clinical Trials Transformation Initiative (CTTI) workshop. Companies adopting AI include Pfizer (immune modeling), AstraZeneca (target discovery), Merck and Bayer (drug repurposing), GSK (COPD treatments), Benevolent AI (COVID-19), Bristol Myers Squibb (CYP450 metabolism), and Taisho in collaboration with in silico (senescence-targeted medicines) [27].
Benefits of AI integration
AI significantly enhances efficiency, accuracy, and consistency in pharmaceutical regulatory affairs by automating dossier preparation, regulatory intelligence, and compliance monitoring [6, 31]. NLP enables rapid review and summarization of extensive regulatory documentation, while predictive analytics facilitates early identification of regulatory risks and submission deficiencies. In addition, cloud-based platforms integrated with blockchain technology improve data traceability, audit readiness, and lifecycle management across global regulatory submissions [14, 28]. Roche has implemented a large language model–assisted Quality Overall Summary (QOS) authoring tool that reduced Module 3 preparation time by more than 60%, while enhancing internal consistency and regulatory readiness across multiple regions [14, 28]. Similarly, AI-driven pharmacovigilance platforms such as Huma. AI have demonstrated the ability to reduce adverse event review timelines from weeks to hours and significantly improve safety signal detection accuracy, thereby strengthening post-market surveillance systems [29]. AI also supports personalized medicine, adaptive clinical trial design, QSAR modeling, and drug repurposing. Regulatory agencies increasingly recognize these advantages, as reflected in initiatives such as the FDA’s PBPK Modeling Pilot under ICH M15, which has enabled optimized clinical trial design and reduced unnecessary patient exposure [26, 27]. Evidence from Innovare journals further supports the role of artificial intelligence in improving regulatory documentation quality, compliance tracking, and pharmacovigilance efficiency, particularly in emerging and resource-limited regulatory environments [33]. Explainable artificial intelligence (XAI) frameworks further enhance transparency and accountability, ensuring that AI functions as a decision-support tool rather than a substitute for expert regulatory judgment [31].
Challenges, risks, and ethical concerns
Despite its significant advantages, the integration of artificial intelligence into pharmaceutical regulatory affairs presents several challenges related to algorithmic transparency, data quality, system interoperability, and ethical governance. Many AI systems function as “black boxes,” making it difficult for regulators to interpret, validate, or justify automated outputs. Consequently, regulatory authorities emphasize the need for explainability, validation, and continuous human oversight to ensure regulatory acceptability and patient safety [6, 31]. Although AI-enabled PBPK modeling and clinical trial prediction tools demonstrate strong regulatory potential, inconsistent data inputs and the absence of standardized validation frameworks across regions have limited their widespread adoption [27]. Similarly, AI-based pharmacovigilance systems trained on biased or incomplete datasets may underreport adverse events in specific populations, underscoring the importance of diverse, high-quality training data and continuous post-deployment monitoring [29, 31].
Ethical concerns-including data privacy, informed consent, algorithmic bias, accountability, and data ownership-remain central barriers to AI adoption. Variability in global regulatory standards further complicates harmonization, particularly for multinational submissions. Studies published in Innovare journals highlight challenges related to data standardization, regulatory acceptance, and workforce readiness during AI implementation in pharmaceutical regulatory operations, emphasizing the need for validated systems and sustained human oversight to ensure compliance and patient safety [32]. Addressing these challenges requires adaptive governance models, independent auditing mechanisms, lifecycle monitoring, and workforce training that bridge regulatory science and data analytics. Sustained public and regulatory trust will depend on transparent governance structures that ensure AI enhances-rather than undermines-regulatory integrity and patient safety [31].
Future directions in regulatory affairs and AI in drug development
Emerging technologies, including AI, ML, digital twins, and structured data ecosystems, enable in silico trials, automated submissions, and real-time compliance monitoring. These tools have the potential to transform drug development [5, 34]. Flexible regulatory frameworks are needed to accommodate rapidly evolving therapies such as digital therapeutics, CRISPR-based gene editing, mRNA vaccines, and cell and gene therapies [5]. Future drug development will rely more on patient-centered models that integrate genomic data, RWE, and patient-reported outcomes [5, 34]. Greater international alignment can reduce duplicative work and accelerate access to innovative therapies globally [5]. AI-driven regulatory science must be guided by principles of ethics, transparency, and patient safety. AI-enabled wearables and smart health technologies will facilitate continuous monitoring, personalized dosing, and improved treatment adherence. Regulatory professionals will increasingly require expertise in data interpretation, digital literacy, and adaptive leadership [5, 34]. Horizon scanning, supply chain resilience, and sustainability will shape future regulatory frameworks. Early adopters of AI are likely to gain advantages such as more efficient clinical trials, faster approvals, and stronger research and development (RandD) pipelines [34].
CONCLUSION
AI is redefining pharmaceutical regulatory affairs by increasing efficiency in dossier preparation, compliance monitoring, pharmacovigilance, and patient safety [6, 26, 31]. Regulatory agencies are transitioning from document-based workflows to data-driven, intelligent systems, offering opportunities for improved consistency, transparency, and accelerated patient access [6, 34]. However, challenges such as inconsistent data standards, opaque algorithms, and ethical concerns remain. Realizing AI’s full potential will require robust governance, flexible regulatory pathways, skilled workforces, and international collaboration [31, 34]. Emerging evidence from Asian pharmaceutical regulatory environments supports the growing role of artificial intelligence in regulatory decision-making, compliance management, and patient safety assurance [32, 35].
LIST OF ABBREVIATIONS
ACCESS–AI-driven Clinical Evaluation and Safety Studies Consortium, AE–Adverse Event, AI–Artificial Intelligence, AI–IoT–Artificial Intelligence–Internet of Things, ANDA–Abbreviated New Drug Application, ANN–Artificial Neural Network, BEST–Biologics Effectiveness and Safety, BfArM–Federal Institute for Drugs and Medical Devices, BLA–Biologics License Application, CANDA–Computer-Assisted New Drug Application, CMC–Chemistry, Manufacturing, and Controls, CONSORT-AI–Consolidated Standards of Reporting Trials–Artificial Intelligence, COPD–Chronic Obstructive Pulmonary Disease, CRISPR–Clustered Regularly Interspaced Short Palindromic Repeats, CTD–Common Technical Document, CTTI–Clinical Trials Transformation Initiative, DAMOS–Drug Application Management and Organization System, DL–Deep Learning, DMAP–Device Mapping Program, DMF–Drug Master File, eCTD – Electronic Common Technical Document, EHR–Electronic Health Record, EMA–European Medicines Agency, EMAP–Evaluation Mapping Program, EU–European Union, FAERS–FDA Adverse Event Reporting System, FDA–Food and Drug Administration, FHIR–Fast Healthcare Interoperability Resources, GMP–Good Manufacturing Practice, GPU–Graphics Processing Unit, HL7–Health Level Seven, HL7-FHIR–Health Level Seven–Fast Healthcare Interoperability Resources, HSA–Health Sciences Authority, ICH–International Council for Harmonisation, IDMP–Identification of Medicinal Products, IND–Investigational New Drug, Info ViP–Information Visualization Platform, IoT–Internet of Things, ISO–International Organization for Standardization, ISTAND–Innovative Science and Technology Approaches for New Drugs, LLM–Large Language Model, LMICs–Low-and Middle-Income Countries, MANSEV–Management of New Submissions and Evaluations, MERS–Medical Electronic Review System, ML–Machine Learning, NDA–New Drug Application, NLP–Natural Language Processing, NMPA–National Medical Products Administration, PBPK–Physiologically Based Pharmacokinetic, PMDA–Pharmaceuticals and Medical Devices Agency, PMS–Post-Market Surveillance, PQ/CMC–Pharmaceutical Quality/Chemistry, Manufacturing, and Controls, PSUR–Periodic Safety Update Report, PV–Pharmacovigilance
QOS–Quality Overall Summary, R and D–Research and Development, RPA–Robotic Process Automation, RPS–Regulated Product Submission, RWE–Real-World Evidence, SCDM–Structured Content and Data Management, SEDAMM–Système d’Évaluation et de Documentation des Autorisations de Mise sur le Marché, SPIRIT-AI–Standard Protocol Items: Recommendations for Interventional Trials–Artificial Intelligence, TMAP–Therapeutic Mapping Program, USFDA–United States Food and Drug Administration, WEB-RADR–Web-Recognizing Adverse Drug Reactions, WHO–World Health Organization, WHO Drug Koda–World Health Organization Drug Dictionary (Koda Tool), XAI–Explainable Artificial Intelligence
AUTHORS CONTRIBUTIONS
Pathipati Sunitha: Conceived and designed the review, conducted an extensive literature search, analyzed and synthesized the relevant scientific and regulatory data, and drafted the initial manuscript. R. S. Sailesh: Contributed to the critical review of the literature, assisted in organizing and structuring the manuscript, and supported refinement of the regulatory concepts and interpretations. S. Nagalakshmi: Provided academic guidance, critically reviewed and edited the manuscript for scientific accuracy and regulatory relevance, ensured compliance with journal requirements, and approved the final version for submission.
CONFLICT OF INTERESTS
The authors declare no conflict of interest.
REFERENCES
Huma T, Peng Z. Introduction to regulatory affairs and different regulatory bodies for pharmaceutical products and impact of digitalization on regulatory affairs. Pharmacol Pharm. 2023;14(11):463-77. doi: 10.4236/pp.2023.1411030.
D Singh V. The ever-expanding role of regulatory affairs. IJSR. 2024;13(2):847-52. doi: 10.21275/SR24209205014.
Patil RS, Kulkarni SB, Gaikwad VL. Artificial intelligence in pharmaceutical regulatory affairs. Drug Discov Today. 2023;28(9):103700. doi: 10.1016/j.drudis.2023.103700, PMID 37442291.
Maddela S. Exploring AI’s impact on drug development and regulatory affairs. Indo Am J Pharm Biosci. 2022;20(2):97-105.
Chisholm O, Critchley H. Future directions in regulatory affairs. Front Med (Lausanne). 2023;9:1082384. doi: 10.3389/fmed.2022.1082384, PMID 36698838.
Fu L, Jia G, Liu Z, Pang X, Cui Y. The Applications and advances of artificial intelligence in drug regulation: a global perspective. Acta Pharm Sin B. 2025;15(1):1-14. doi: 10.1016/j.apsb.2024.11.006, PMID 40041903.
Huanbutta K, Burapapadh K, Kraisit P, Sriamornsak P, Ganokratanaa T, Suwanpitak K. Artificial intelligence-driven pharmaceutical industry: a paradigm shift in drug discovery formulation development manufacturing quality control and post-market surveillance. Eur J Pharm Sci. 2024;203:106938. doi: 10.1016/j.ejps.2024.106938, PMID 39419129.
Shelke S. Navigating drug regulatory affairs: an analytical review of compliance in the pharmaceutical industry. J Drug Deliv Ther. 2024;14(2):1-2.
Chandrasekaran K, Kumar TM, Balamuralidhara B. Artificial intelligence in pharmaceutical regulatory affairs: an introduction. Asian J Biol Life Sci. 2025;14(2):235-9. doi: 10.5530/ajbls.20251167.
Sharma S, Mathure D, Dingankar S, Dhapte Pawar V. In pursuit of software solutions for pharmaceutical regulatory affairs: insights and trends. Ann Pharm Fr. 2025;83(6):1053-61. doi: 10.1016/j.pharma.2025.05.005, PMID 40403839.
Madhuria N, Saxena V, Singh SP, Sharma G, Begum MS. Role of digitalization and artificial intelligence in streamlining pharmaceutical regulatory affairs: opportunities and challenges. Front Health Inform. 2024;13(3):1-12.
Ahammad N, Reddy N, Nagabhushanam MV, Ramakrishna B. Challenges faced during eCTD and CTD filing procedures for USFDA and Canada. J Drug Deliv Ther. 2019;9(4S):673-9. doi: 10.22270/jddt.v9i4-s.3334.
Ross C. The history of electronic regulatory submissions technologies: a focus on eCTD (electronic common technical document) and its challenges and benefits. Athens, GA: University of Georgia; 2013. Available from: https://getd.libs.uga.edu/pdfs/ross_charnelle_201008_ms.pdf.
Ahluwalia K, Abernathy MJ, Algorri M, Cauchon NS, Perico Norred NM, Youssef RY. The Future of regulatory filings: digitalization. AAPS Open. 2025;11(1):9. doi: 10.1186/s41120-025-00113-7.
Zade N, Mate G, Kishor K, Rane N, Jete M. NLP based automated text summarization and translation: a comprehensive analysis. In: 2024 2nd International Conference on Sustainable Computing and Smart Systems (ICSCSS). India: IEEE; 2024 Jul 10. p. 528-31. doi: 10.1109/ICSCSS60660.2024.10624907.
Jayatilleke N, Weerasinghe R, Senanayake N. Advancements in natural language processing for automatic text summarization. In: 2024 4th International Conference on Computer Systems (ICCS). Sri Lanka: IEEE; 2024 Sep 20. p. 74-84. doi: 10.1109/ICCS62594.2024.10795848.
Haque MM, Pervin S, Begum Z. Literature review of automatic single-document text summarization using NLP. Int J Innov Appl Stud. 2013;3(3):857-65.
Black JE, Kueper JK, Williamson TS. An Introduction to machine learning for classification and prediction. Fam Pract. 2023;40(1):200-4. doi: 10.1093/fampra/cmac104, PMID 36181463.
Badawy M, Ramadan N, Hefny HA. Healthcare predictive analytics using machine learning and deep learning techniques: a survey. J Electr Syst Inf Technol. 2023;10(1):40. doi: 10.1186/s43067-023-00108-y.
Radke AM, Dang MT, Tan A. Using robotic process automation (RPA) to enhance item master data maintenance process. Log Forum. 2020;16(1):129-40. doi: 10.17270/J.LOG.2020.380.
Huang F, Vasarhelyi MA. Applying robotic process automation (RPA) in auditing: a framework. Int J Acc Inf Syst. 2019;35:100433. doi: 10.1016/j.accinf.2019.100433.
Hofmann P, Samp C, Urbach N. Robotic process automation. Electron Markets. 2020;30(1):99-106. doi: 10.1007/s12525-019-00365-8.
Madakam S, Holmukhe RM, Jaiswal DK. Future digital workforce: robotic process automation. J Inf Syst Technol Manag. 2019;16:e201916001. doi: 10.4301/S1807-1775201916001.
Raghupathi W, Raghupathi V. Big data analytics in healthcare: promise and potential. Health Inf Sci Syst. 2014;2(1):3. doi: 10.1186/2047-2501-2-3, PMID 25825667.
Venkatesh R, Ahad HA, Sreedhara A, Gowda SR, Krishnappa C, Nagesh PH. Innovative cloud solutions reshaping healthcare and pharma industries with enhanced efficiency, collaboration and patient-centric paradigms. Asian J Pharm Res. 2025;15(2):163-70. doi: 10.52711/2231-5691.2025.00027.
Zhang K, Yang X, Wang Y, Yu Y, Huang N, Li G. Artificial intelligence in drug development. Nat Med. 2025;31(1):45-59. doi: 10.1038/s41591-024-03434-4, PMID 39833407.
Mirakhori F, Niazi SK. Harnessing the AI/ML in drug and biological products discovery and development: the regulatory perspective. Pharmaceuticals (Basel). 2025;18(1):47. doi: 10.3390/ph18010047, PMID 39861110.
Venna SR. AI and automation in regulatory operations: the future of eCTD submissions. Indo Am J Pharm Biosci. 2023;21(2):33-42.
Khinvasara T, Tzenios N, Shankar A. Post-market surveillance of medical devices using AI. J Compl Altern Med Res. 2024;25(7):108-22. doi: 10.9734/jocamr/2024/v25i7552.
Reniewicz J, Suryaprakash V, Kowalczyk J, Blacha A, Kostello G, Tan H. Artificial intelligence / machine-learning tool for post-market surveillance of in vitro diagnostic assays. N Biotechnol. 2024;79:82-90. doi: 10.1016/j.nbt.2023.11.005, PMID 38040287.
Gude S, Gude YS. The synergy of artificial intelligence and machine learning in revolutionizing pharmaceutical regulatory affairs. Translational and Regulatory Sciences. 2024;6(2):37-45. doi: 10.33611/trs.2024-005.
Muppalla A, Maddi BD, Maddi NV. Artificial intelligence in regulatory compliance: transforming pharmaceutical and healthcare documentation. Int J Drug Reg Affairs. 2025;13(2):73-80. doi: 10.22270/ijdra.v13i2.764.
Rao P, Srinivas R, Kiranmai G. Application of artificial intelligence in pharmaceutical regulatory affairs and compliance management. Asian J Pharm Clin Res. 2023;16(8):45-50.
Bose A. Regulatory initiatives for artificial intelligence applications: regulatory writing implications. Med Writ. 2023;32(3):12-5. doi: 10.56012/kdyv1106.
Jasmine P, Ibrahim A, Siddiq S, Dhanurshra A, Nirmala M, Shakir Basha SS. Transformative applications of artificial intelligence in healthcare and pharmaceutical innovation. Int J Appl Pharm. 2025;17(3):101-11. doi: 10.22159/ijap.2025v17i3.52719.