
Int J Pharm Pharm Sci, Vol 18, Issue 4, 1-6Review Article
TRADITIONAL ANTIDIABETIC PLANTS IN EIGHT WEST AFRICAN COUNTRIES: SYSTEMATIC REVIEW OF ETHNOBOTANICAL STUDIES AND PRIORITY SPECIES
ABDULAI TURAY¹* , OLAYINKA TAIWO ASEKUN², MOSHOOD OLUSOLA AKINLEYE³
, OLAYINKA TAIWO ASEKUN², MOSHOOD OLUSOLA AKINLEYE³
¹Drug Discovery and Development, University of Lagos, Lagos, Nigeria. ²Faculty of Physical and Earth Sciences, University of Lagos, Lagos State, Nigeria. ³Faculty of Pharmacy, University of Lagos, Lagos State, Nigeria
*Corresponding author: Abdulai Turay; *Email: abdulai.turay@usl.edu.sl
Received: 21 Dec 2025, Revised and Accepted: 13 Feb 2026
ABSTRAC
Diabetes mellitus is rising across West Africa, where barriers to sustained access to conventional care and strong cultural acceptability of traditional medicine drive widespread use of plant-based remedies. Ethnobotanical evidence is dispersed across countries and often reported with variable botanical verification and quantitative prioritization, limiting cross-country comparison and selection of candidate species for validation and safety monitoring. Objective of the study was to systematically synthesise ethnobotanical or ethnopharmacological studies reporting medicinal plants used to manage diabetes mellitus or hyperglycaemia in eight West African countries, and summarise commonly cited species, plant parts, preparation methods, and routes of administration. This review was conducted and reported in line with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidance. Searches of PubMed and Google Scholar were complemented by targeted journal or publisher website searches and backward reference screening (last search: 18 December 2025). Primary ethnobotanical or ethnopharmacological studies from eight West African countries reporting ≥1 plant used for diabetes or hyperglycaemia were eligible (English or French). Records were screened in two stages (title or abstract, then full text). Extracted data included country or setting, respondent type, sample size (if reported), antidiabetic species, plant parts, preparation or administration patterns, and priority species (e. g., highest relative frequency of citation where reported). The search yielded 512 records; after de-duplication (92 removed), 420 records were screened and 12 studies met inclusion criteria (8 countries: Nigeria, Ghana, Togo, Benin, Côte d’Ivoire, Guinea, Senegal, Sierra Leone). Across studies, a total of 8 recurrently reported antidiabetic medicinal plant species were identified. Leaves were the most commonly reported plant part and decoction was the predominant preparation method, with oral administration most frequently described. Recurrently reported candidate species across multiple settings included Azadirachta indica, Momordica charantia, Moringa oleifera, Phyllanthus amarus, Khaya senegalensis, Garcinia kola, Citrus aurantifolia, and Tetrapleura tetraptera. Among these, Moringa oleifera was reported in 8 of the 12 studies and Azadirachta indica in 7 of 12 studies, indicating their frequent citation across countries. Where quantitative prioritization was reported, Citrus aurantifolia was highlighted as the highest-ranked species in Benin (RFC = 0.21). In conclusion, the diabetes ethnomedicine of eight West African countries is characterised by reliance on leaf-based aqueous preparations, mainly decoctions or infusions, administered orally, and a recurring shortlist of priority species. Future studies should strengthen reporting (voucher specimens, quantitative indices, dosing details) and link priority plants to toxicovigilance or pharmacovigilance and staged experimental or clinical validation.
Keywords: Diabetes mellitus, Ethnobotany, Ethnopharmacology, Medicinal plants, West Africa, Traditional medicine, PRISMA 2020
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijpps.2026v18i4.57871 Journal homepage: https://innovareacademics.in/journals/index.php/ijpps
INTRODUCTION
Diabetes mellitus (DM) is a chronic metabolic disorder defined by persistent hyperglycaemia resulting from impaired insulin secretion, impaired insulin action, or both, and it is a major driver of microvascular (e. g., retinopathy, nephropathy, neuropathy) and macrovascular (e. g., cardiovascular disease, stroke) complications when poorly controlled [1]. The global burden of diabetes continues to rise, with hundreds of millions of adults living with the condition and a substantial proportion remaining undiagnosed, especially in low-and middle-income settings where screening coverage, continuity of care, and affordability of medicines can be limited [2]. In West Africa, rapid urbanization, dietary transitions, physical inactivity, and population growth are contributing to increasing DM prevalence alongside persistent health-system constraints, creating a dual challenge of expanding need and limited capacity for sustained, affordable long-term management [2].
Traditional medicine remains a central component of healthcare-seeking behaviour across many African settings and is frequently used for chronic diseases, including DM. Regional policy documents note that traditional medicine is widely relied upon for primary healthcare in Africa, supported by cultural acceptability, local availability, and cost considerations [3]. At the same time, access barriers to essential medicines persist across parts of the region, which can further encourage reliance on plant-based therapies when conventional treatment is unavailable, unaffordable, or perceived as less culturally aligned [4]. Many herbal medicines have demonstrated antidiabetic activity; for example, Cassia auriculata, Gymnema sylvestre, Syzygium cumini, Trigonella foenum-graecum, and Cinnamomum zeylanicum are widely cited for their hypoglycemic effects and even pomegranate (Punica granatum) leaf extract has shown significant anti-diabetic activity in experimental models [4–6].
Within West African communities, medicinal plants used for diabetes management are commonly identified through ethnobotanical knowledge systems and documented in ethnobotanical or ethnopharmacological studies. However, the evidence base is often heterogeneous across countries and districts, differing in sampling approaches, respondent types, botanical identification methods (including whether voucher specimens are deposited), and the use of quantitative indices (e. g., frequency of citation, use value, informant consensus). This variability complicates cross-country comparison and makes it difficult to prioritise candidate species for laboratory validation, clinical evaluation, and public-health guidance [9].
Beyond effectiveness, safety considerations are critical. Herbal medicines can produce adverse reactions, vary in composition due to differences in harvesting or processing, and may interact with prescribed antidiabetic medicines, raising concerns about hypoglycaemia risk, altered drug exposure, and delayed presentation for care [10, 11].
These risks strengthen the case for synthesizing ethnobotanical evidence in a structured way that supports rational prioritization of species while highlighting gaps in safety reporting and the need for linked pharmacovigilance or toxicovigilance efforts [10, 11].
Whereas large compilations exist in other contexts (for example, 61 antidiabetic plant species documented in Limpopo Province, South Africa; 143 species compiled in Bangladesh; and a recent pan-African review covering the continent), no prior synthesis has focused specifically on West Africa. The present review therefore, provides a PRISMA 2020-guided analysis of ethnomedicinal diabetes plants from eight West African countries, highlighting recurring priority species and key knowledge gaps for validation and safety monitoring [7–9].
MATERIALS AND METHODS
Study design and reporting
This systematic review was conducted and reported in accordance with the PRISMA 2020 guidance to ensure transparent identification, selection, and synthesis of evidence [10]. The primary aim was to synthesise ethnobotanical or ethnopharmacological studies reporting medicinal plants used for the management of DM or hyperglycaemia in eight West African countries, and to summarise commonly cited species, plant parts, preparation methods, and administration patterns. A PRISMA 2020 checklist for this review is provided in Supplementary file 1.
Search strategy
A structured search strategy was developed using key concepts related to diabetes and ethnobotany. Searches were conducted in PubMed and Google Scholar and complemented by targeted searches of relevant journal or publisher websites and backward reference screening of included papers. The last search was conducted on 18 December 2025. Search terms covered diabetes-related keywords (“diabetes”, “hyperglyc*”, “antidiabetic”), ethnomedicine terms (“ethnobotanical”, “ethnopharmacological”, “traditional medicine”), and geographic terms specifying West African countries (e. g., Nigeria, Ghana, Togo, Benin, Senegal, Guinea, Côte d’Ivoire, and Sierra Leone). An example search string used in PubMed or Google Scholar was: (“diabetes” or “hyperglyc*” or “antidiabetic”) and (ethnobotanical or ethnopharmacological OR “traditional medicine”) and (Nigeria or Ghana or Togo or Benin or Senegal or Guinea or “Côte d’Ivoire” or “Ivory Coast” or “Sierra Leone”).
Eligibility criteria
Studies were eligible if they were primary ethnobotanical or ethnopharmacological studies conducted in a West African country and explicitly reported at least one plant species used to treat diabetes mellitus or hyperglycaemia, with full text accessible in English or French. Review articles were excluded, as were studies that did not report diabetes-or hyperglycaemia-related use, studies conducted outside West Africa, and records lacking sufficient methodological detail to confirm primary data collection.
Study selection
The study selection process was conducted in two stages consistent with PRISMA 2020 reporting principles [8]. Records were screened for relevance at title or abstract level, followed by full-text assessment against the eligibility criteria. Reasons for full-text exclusion were documented (e. g., not DM-related, not a primary ethnobotanical study, or not conducted in West Africa). Where screening decisions differed, disagreements were resolved by discussion and consensus.
Data extraction
A standardised data extraction template was used to ensure consistency across included studies. Extracted variables included study characteristics (country, setting, respondent type, and sample size when reported), ethnobotanical outcomes (number of antidiabetic species reported, plant parts used, preparation methods, and route of administration), priority species (based on author-reported indicators such as highest citation or highest relative frequency of citation [RFC] where available), and reporting features (evidence of botanical verification such as voucher specimen documentation and the presence or absence of quantitative ethnobotanical indices).
Data analysis and presentation
Given heterogeneity in study design and reporting, a meta-analysis was not undertaken. Findings were synthesised using descriptive tabulation (distribution of studies by country; summary of plant parts used, preparation methods, and routes of administration; listing of commonly reported or prioritized species) and narrative synthesis (identification of cross-country patterns such as reliance on leaves and decoctions, and description of recurrent priority species and country-specific prominence where quantitative indices were reported). Results were presented in structured tables and narrative summaries to enhance clarity and reproducibility.
Quality appraisal and risk of bias
Given the absence of a standardized ethnobotanical risk-of-bias tool, a pragmatic quality checklist was applied to evaluate the completeness and reliability of reporting across included ethnobotanical studies. The checklist focused on clarity of sampling strategy and study population; botanical identification procedures, including voucher specimen documentation; transparency of data collection methods; reporting of quantitative ethnobotanical indices (e. g., RFC or use value) when applicable; and completeness of plant-use reporting (parts used, preparation, and administration). This appraisal was used to contextualize confidence in the evidence rather than to exclude studies.
Ethics
Ethical approval was not required because this review used only publicly available published literature. All procedures followed accepted standards for responsible evidence synthesis and reporting [8].
RESULTS
Study selection
The database searches (PubMed and Google Scholar), supplemented by targeted journal or publisher website searches and backward reference screening, identified a total of 512 records (480 from databases and 32 from other methods). After removing 92 duplicates, 420 records were screened by title or abstract, of which 360 were excluded. Subsequently, 60 reports were sought for retrieval; 4 could not be retrieved. A total of 56 full-text reports were assessed for eligibility, and 44 were excluded with reasons (not West Africa, n = 8; not DM or hyperglycaemia-related, n = 17; not primary ethnobotanical or ethnopharmacological study, n = 12; insufficient methodological detail, n = 7). Overall, 12 studies met the inclusion criteria and were included in the final review (fig. 1).
Characteristics of included studies
A total of 12 eligible ethnobotanical or ethnopharmacological studies were included from eight West African countries: Nigeria, Ghana, Togo, Benin, Côte d’Ivoire, Guinea, Senegal, and Sierra Leone. Studies varied substantially in reporting completeness: some reported respondent numbers and detailed ethnobotanical indices (e. g., RFC), while others provided inventories without quantitative prioritization metrics.
Country coverage of included studies (n=12; 8 countries): Nigeria (1), Ghana (2), Togo (2), Benin (2), Côte d’Ivoire (2), Senegal (1), Guinea (1) and Sierra Leone (1) [13–24].
Quality appraisal and risk of bias
Using the pragmatic reporting-quality checklist described in the Methods section, the most common limitations across the included studies were:
Incomplete reporting of sampling strategy and sample size (several studies listed NR for respondent numbers).
Inconsistent botanical verification reporting (voucher specimen documentation was not consistently stated across extracted summaries).
Variable use of quantitative ethnobotanical indices (RFC or use value and similar indices were reported in some studies, but absent or unclear in others).
Incomplete dosing or administration details (frequency and duration of use were often not standardised or not fully reported).
Synthesis of findings
Across the included studies, leaves were the most commonly reported plant part and decoction was the predominant preparation method, with oral administration frequently reported when routes were specified. Where quantitative indicators were available, they highlighted country-specific prominence of certain species (e. g., RFC reporting in Benin) [13-15, 17].
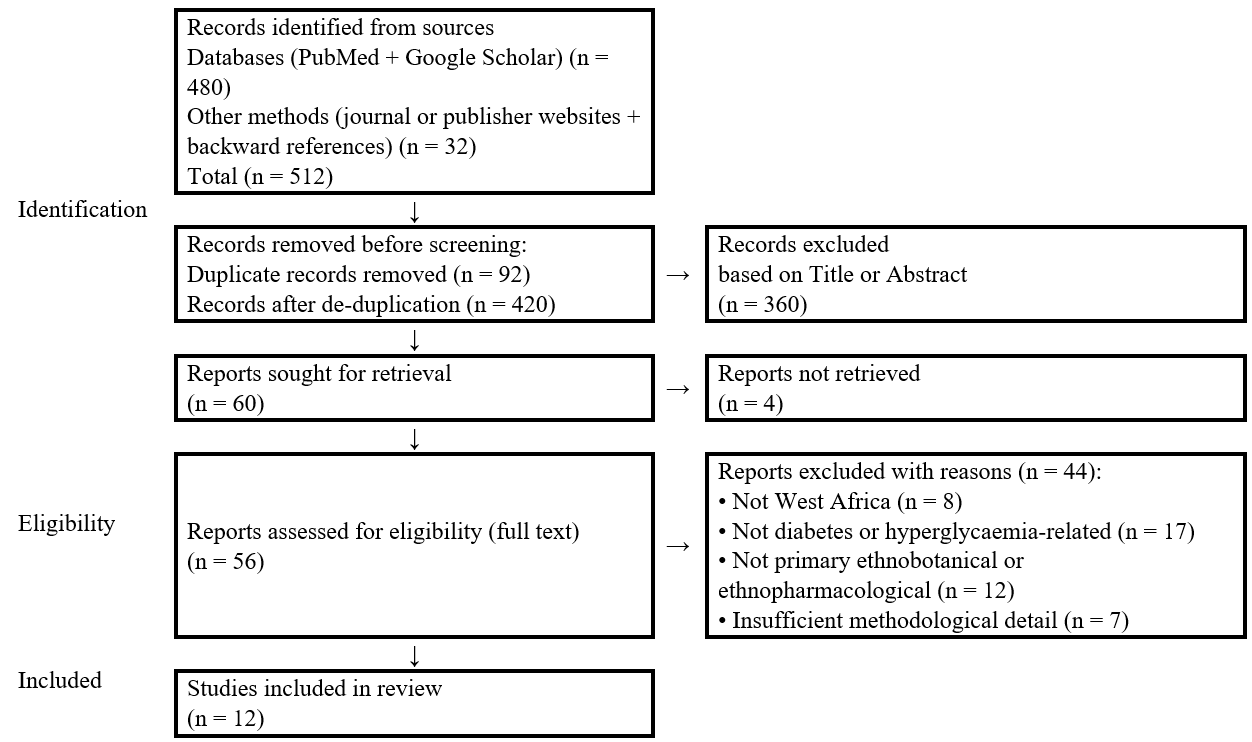
Fig. 1: PRISMA 2020 flow diagram of study selection for the systematic review
Table 1: Characteristics of included studies (n = 12)
| Country | Study (year) | Setting/participants | Sample size | Key notes (parts or prep; priority plants) |
| Nigeria | Abo et al., 2008 [13] | South-West Nigeria; ethnobotanical survey | NR | Antidiabetic inventory reported. |
| Ghana | Asase and Yohonu, 2016 [14] | Dangme West District; traditional healers | NR | Documented antidiabetic plants and use patterns. |
| Ghana | Asafo-Agyei et al., 2023 [15] | Herbalists (Ghana) | NR | Reported plants used for diabetes (and hypertension) and preparation patterns. |
| Togo | Karou et al., 2011 [16] | Central Region; traditional medicine | NR | Plants used for diabetes and hypertension in traditional medicine. |
| Togo | Holaly et al., 2015 [17] | Maritime Region; traditional healers | 164 respondents | High diversity of species and recipes; oral administration common. |
| Benin | Lawin et al., 2015 [18] | Sudano-Guinean zone; practitioners or sellers or farmers | 126 respondents | Leaves most used (27%); decoction common (53%); Citrus aurantifolia highest RFC (0.21). |
| Benin | Lalèyè et al., 2015 [19] | Multiple locations; healers or traders or farmers | 254 interviews | Leaves or roots or bark predominant; decoction and infusion common; indices reported (e. g., similarity measures). |
| Côte d’Ivoire | Diomandé et al., 2022 [20] | Abidjan (Adjamé markets); plant sellers | 40 sellers | Fabaceae/Euphorbiaceae prominent; Tetrapleura tetraptera frequently recommended; leaves and decoction common. |
| Côte d’Ivoire | N’guessan et al., 2009 [21] | Agboville; traditional healers | NR | Semi-structured interviews; decoction commonly reported. |
| Senegal | Dièye et al., 2008 [22] | Senegal; diabetes management context | NR | Reported medicinal plants used for diabetes management. |
| Guinea | Diallo et al., 2012 [23] | Coastal lowlands; traditional medicine | NR | Ethnobotanical investigation of diabetes management in Guinean traditional medicine. |
| Sierra Leone | Adepoju et al., 2023 [24] | Sierra Leone; ethnobotanical survey | NR | Reported antidiabetic plants used locally. |
NR = Not reported; RFC = Relative Frequency of Citation.
Table 2: Cross-study ethnobotanical patterns (as reported in included studies)
| Domain | Dominant pattern | No. of studies (n=12) | Evidence or examples from included studies |
| Plant parts used | Leaves most commonly used | 8 | Reported across studies where plant parts were described. |
| Preparation method | Decoction predominates; infusion also common | 7 | Decoction was frequently described in included studies reporting preparation methods. |
| Administration route | Mainly oral | 10 | Oral administration predominated across studies reporting route of administration. |
| Quantitative prioritization | Indices inconsistently reported | 2 | Only a minority of studies reported comparable quantitative indices (e. g., RFC). |
Table 3: Table 3: Priority antidiabetic plant species identified from included studies (n=12): recurrent and single‑study mentions.
| Plant species | Summary note | Countries reported in | Key source(s) among included studies |
| Azadirachta indica | Recurrently reported as an antidiabetic remedy across West African ethnobotanical literature. | Benin, Ghana, Guinea, Nigeria, Sierra Leone, Togo | [13-17, 19, 23, 24] |
| Momordica charantia | Frequently cited for diabetes or hyperglycaemia management across settings. | Ghana, Guinea, Senegal, Sierra Leone, Togo | [14–17, 22–24] |
| Moringa oleifera | Commonly listed among antidiabetic plants across multiple countries. | Benin, Ghana, Guinea, Senegal, Sierra Leone, Togo | [14, 15, 17–19, 22–24] |
| Phyllanthus amarus | Recurrently documented in diabetes-related ethnomedicinal use. | Ghana, Guinea, Nigeria, Sierra Leone, Togo | [13–15, 17, 23, 24] |
| Khaya senegalensis | Repeatedly reported for diabetes management in regional ethnobotanical sources. | Benin, Ghana, Guinea, Senegal, Sierra Leone, Togo | [14–17, 19, 22–24] |
| Garcinia kola | Reported in multiple contexts as part of diabetes-related traditional remedies. | Ghana, Nigeria, Senegal, Sierra Leone, Togo | [13–17, 22, 24] |
| Citrus aurantifolia | Highlighted as a priority plant where indices were reported (e. g., RFC in Benin). | Benin | [18, 19] |
| Tetrapleura tetraptera | Frequently recommended in market/healer settings (e. g., Côte d’Ivoire). | Côte d’Ivoire | [20, 21] |
| Secamone afzelii | Single‑study mention (reported in 1 included study). | Nigeria | [13] |
| Curculigo pilosa | Single‑study mention (reported in 1 included study). | Nigeria | [13] |
| Gladiolus psittaccinus | Single‑study mention (reported in 1 included study). | Nigeria | [13] |
| Abrus precatorius | Single‑study mention (reported in 1 included study). | Nigeria | [13] |
Key source(s) indicate included primary studies in this review that reported the plant as used for diabetes or hyperglycaemia.
Frequently recurring antidiabetic species across West African settings
A recurring set of antidiabetic species was reported across multiple West African settings in the included literature. Because many studies did not consistently publish comparable quantitative indices, recurrence is presented as cross-study presence rather than pooled frequency.
Results summary statement
Overall, the included evidence (n = 12 studies; 8 countries) indicates that West African diabetes ethnomedicine is characterised by strong reliance on leaf-based preparations, predominantly decoctions or infusions, administered mainly orally, with a recurring set of priority species (Azadirachta indica, Momordica charantia, Moringa oleifera, Phyllanthus amarus, Khaya senegalensis, Garcinia kola, Citrus aurantifolia, Tetrapleura tetraptera) observed across multiple studies.
DISCUSSION
A total of 12 priority antidiabetic plant species were identified from the included studies (8 countries): 8 recurrently reported species (≥2 studies) and 4 single‑study mentions (1 study each) [13–24].
Table 3 summarises the 12 priority antidiabetic species and their supporting primary studies. Recurrence across included studies showed that Azadirachta indica was reported in 9 studies; Moringa oleifera and Khaya senegalensis in 8 studies each; Momordica charantia and Garcinia kola in 7 studies each; and Phyllanthus amarus in 6 studies. Citrus aurantifolia and Tetrapleura tetraptera were each reported in 2 studies, while Secamone afzelii, Curculigo pilosa, Gladiolus psittaccinus and Abrus precatorius were each reported in 1 study [13–24].
These findings indicate substantial cross‑country consistency in the use of a core set of antidiabetic species, alongside several country‑specific (single‑study) species that may warrant further pharmacological investigation [13–24].
Notably, the recurrence analysis highlights Azadirachta indica as the most frequently reported species (9/12 studies), followed by Moringa oleifera and Khaya senegalensis (8/12 studies each) [13–24].
This systematic review synthesised ethnobotanical or ethnopharmacological evidence on medicinal plants used for DM or hyperglycaemia management across West Africa. Despite heterogeneity in study designs and reporting, several cross-cutting patterns emerged: widespread reliance on leaf-based remedies; predominance of water-based preparations, particularly decoctions (and, to a lesser extent, infusions); mainly oral administration; and recurrence of a relatively small set of candidate species across multiple countries.
The predominance of leaves and aqueous preparations is consistent with practical and ecological realities of traditional healthcare practice. Leaves are relatively accessible, renewable (harvesting can be less destructive than bark or root collection), and compatible with household-level preparation using water as the extraction medium. From a quality assurance perspective, however, variability in leaf age, harvesting season, drying conditions, and storage can affect phytochemical composition and therefore the consistency of exposure. World Health Organization (WHO) guidance emphasizes that correct botanical identification and basic quality control steps (identity, purity, and detection of contaminants or adulterants) are foundational prerequisites before standardization or pharmacological evaluation [21].
Leaves can often be harvested sustainably without destroying the plant, and they are frequently rich in secondary metabolites (e. g., flavonoids, tannins, alkaloids) that may contribute to bioactivity. In addition, leaf-based remedies prepared as decoctions or infusions align with culturally familiar, low-cost household preparation practices, which may reinforce their preference in many West African settings [5].
A key implication of this review is that cross-study recurrence can support prioritization when quantitative ethnobotanical indices are inconsistently reported. Nevertheless, recurrence is not equivalent to clinical effectiveness. Frequent citation may also reflect local availability, market circulation, cultural salience, or transboundary knowledge diffusion rather than therapeutic benefit. Where available, quantitative indices such as RFC, use value, and informant consensus factor help distinguish highly salient remedies within a defined study population and support transparent prioritization [23, 24]. Future reviews would be strengthened if primary studies more consistently reported comparable indices and the underlying denominators (e. g., number of informants and number of use-reports).
Botanical verification is fundamental for the credibility and downstream utility of ethnobotanical evidence. Misidentification, synonym confusion, and incomplete taxonomic reporting can undermine reproducibility and may lead to incorrect attribution of pharmacological activity or toxicity. Accordingly, voucher specimen deposition and reporting of voucher numbers and herbarium repositories should be considered minimum reporting standards for ethnobotanical studies intended to inform further laboratory and clinical work [21].
Safety considerations are particularly important in diabetes, where concurrent use of plant remedies and conventional antidiabetic medicines is common. This may increase the risk of additive hypoglycaemia and or clinically meaningful herb–drug interactions. Evidence from pharmacovigilance and interaction research indicates that herbal products can alter drug exposure via effects on absorption, drug-metabolizing enzymes, and transporters, and that adverse reactions to herbal medicines are often under-recognised and under-reported [6, 7]. WHO has issued specific guidance on integrating herbal medicines into pharmacovigilance systems, including case reporting, causality assessment, and the need for product identification and traceability to support signal detection [22]. These considerations reinforce the need to link highly cited antidiabetic plants to structured toxicovigilance and pharmacovigilance activities in West Africa, particularly where herbal products are widely used.
From a translational standpoint, the recurrent plants identified in this review, such as Azadirachta indica, Momordica charantia, Moringa oleifera, Phyllanthus amarus, Khaya senegalensis, Garcinia kola, Citrus aurantifolia and Tetrapleura tetraptera, represent immediate priorities for targeted phytochemical, pharmacological and safety evaluation. In addition, single‑study species (Secamone afzelii, Curculigo pilosa, Gladiolus psittaccinus and Abrus precatorius) may reflect locally important remedies but require confirmation in additional settings before being prioritised [13–24].
Finally, this review underscores gaps in geographic coverage and reporting completeness. Only eight West African countries were represented among the 12 included studies, suggesting that the published ethnobotanical record may under-represent several Economic Community of West African States (ECOWAS) countries and may be influenced by database indexing and retrieval constraints. Expanding evidence synthesis using additional databases and country repositories, and strengthening primary study reporting (sampling, voucher specimens, indices, and dosing detail), would improve cross-country comparability and the utility of ethnobotanical evidence for health research and policy.
Recommendations
Standardise ethnobotanical reporting: Primary studies should clearly report sampling strategy, respondent characteristics, interview tools, and quantitative indices (e. g., RFC, use value, and informant consensus) to enable comparison and prioritization [23, 24].
Strengthen botanical verification: Deposit voucher specimens and report voucher numbers, herbarium location, and accepted taxonomic names for all cited species to improve reproducibility and reduce misidentification risk [21].
Improve preparation and dosing documentation: Report preparation parameters (plant part mass, solvent volume, boiling or steeping time), administration frequency, and duration of use to support experimental replication and risk assessment.
Link priority plants to safety monitoring: Integrate commonly used antidiabetic plants and products into national pharmacovigilance systems, including structured adverse reaction reporting and case investigation [22].
Promote quality assurance of herbal products: Apply identity, purity, and contaminant testing (e. g., microbial load, heavy metals, pesticide residues, and adulterants) as recommended in WHO quality-control guidance [21].
Prioritise staged pharmacological validation: Use recurrence and any available quantitative indices to select candidates for mechanistic studies and preclinical evaluation, with transparent criteria and preregistered protocols where feasible [8].
Support responsible integration into care pathways: Strengthen patient counselling on concurrent use of herbal and conventional antidiabetic medicines to reduce interaction-related risks [7].
Encourage regional collaboration: Establish multi-country research networks and shared repositories (voucher specimens, datasets, and standard operating procedures) to harmonize methods and accelerate comparative learning across West Africa.
LIMITATIONS
This review has several limitations. First, the search relied primarily on PubMed and Google Scholar plus targeted website and reference screening; relevant studies indexed only in other databases (e. g., Scopus, Web of Science, African Journals Online, and country repositories) may have been missed. Second, although English and French full texts were eligible, indexing and retrieval constraints may still have under-represented Francophone or Lusophone outputs. Third, heterogeneity in study designs and reporting, particularly incomplete reporting of sample sizes, voucher specimens, indices, and dosing, limited the feasibility of pooled quantitative synthesis and reduced cross-study comparability. Fourth, the pragmatic checklist appraisal was used to contextualize confidence in reporting completeness rather than to exclude studies; therefore, the certainty of specific plant-priority rankings remains constrained by the quality of primary reporting. Finally, ethnobotanical studies may underreport medicinal plant use because traditional healers sometimes guard knowledge or are reluctant to disclose all information, which may result in omission of some antidiabetic remedies from the published literature and, consequently, from this review.
Future research
Future work should: (i) expand country coverage to include more ECOWAS states and under-represented settings; (ii) adopt harmonized ethnobotanical protocols, including explicit calculation and reporting of quantitative indices and voucher specimen documentation [23, 24, 21]; (iii) develop standard operating procedures for preparation parameters that reflect local practice while enabling laboratory replication; (iv) evaluate safety through toxicological screening, contaminant profiling, and prospective adverse reaction monitoring in line with WHO herbal pharmacovigilance guidance [22]; and (v) conduct clinically relevant studies (including interaction assessments) for prioritized species only after standardization and safety evaluation.
CONCLUSION
This review identified 12 eligible ethnobotanical or ethnopharmacological studies from eight West African countries, indicating consistent reliance on leaf-based, water-prepared remedies, predominantly decoctions or infusions, administered mainly orally. A recurring set of candidate antidiabetic species was observed across multiple settings, providing a practical shortlist for prioritized standardisation and validation. Strengthening primary-study reporting (sampling, voucher specimens, quantitative indices, and dosing detail) and integrating safety monitoring for commonly used herbal antidiabetic remedies are critical steps to improve the reproducibility, comparability, and public-health value of ethnobotanical evidence in West Africa.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
Abdulai Turay conceived the study, performed searches, screening, data extraction, synthesis, and drafted the manuscript. Olayinka Taiwo Asekun contributed to methodological refinement, supervision, and critical revision. Moshood Olusola Akinleye contributed to supervision, scientific guidance, and critical revision. All authors reviewed and approved the final manuscript.
CONFLICT OF INTERESTS
The authors declare no conflict of interest.
AI-use disclosure
AI-use disclosure: No AI-generated text was used in the original manuscript. This revision was prepared with the assistance of an AI language model (ChatGPT) for editing purposes, under the authors’ direction, with all final content reviewed and approved by the authors.
REFERENCES
American Diabetes Association Professional Practice Committee. Diagnosis and classification of diabetes: standards of care in diabetes-2024. Diabetes Care. 2024;47(Suppl 1):S20–42. doi: 10.2337/dc24-S002, PMID 38078589.
International Diabetes Federation. Diabetes facts and figures. Brussels, Belgium: International Diabetes Federation; 2025. Available from: https://idf.org/about-diabetes/diabetes-facts‑figures/. [Last accessed on 18 Dec 2025].
World Health Organization Regional Office for Africa. Guidelines for registration of traditional medicines in the WHO African region. Brazzaville, Republic of Congo: World Health Organization Regional Office for Africa; 2010. Available from: https://iris.who.int/handle/10665/336176.
World Health Organization Regional Office for Africa. African Traditional Medicine Day 2022. Brazzaville, Republic of Congo: World Health Organization Regional Office for Africa; 2022 Aug 31. Available from: https://www.afro.who.int/regional-director/speeches-messages/african-traditional-medicine-day-2022. [Last accessed on 18 Dec 2025].
Rahul G, Nandhakumar E. A systematic review of five herbal ingredients for the management of diabetes mellitus. Asian J Pharm Clin Res. 2022;15(12):18-23. doi: 10.22159/ajpcr.2022.v15i12.45427.
Palanisamy V, Muthumanikandan A, Balakrishnan S. In vitro anti-diabetic activity of Punica granatum Linn leaf extract. Asian J Pharm Clin Res. 2023;16(2):15-7. doi: 10.22159/ajpcr.2023.v16i2.44964.
Semenya SS, Maroyi A. A review of plants used against diabetes mellitus by Bapedi and Vhavenda ethnic groups in the Limpopo province, South Africa. Asian J Pharm Clin Res. 2019;12(10):44-50. doi: 10.22159/ajpcr.2019.v12i10.34993.
Rahman MM, Uddin MJ, Reza AS, Tareq AM, Emran TB, Simal Gandara J. Ethnomedicinal value of antidiabetic plants in Bangladesh: a comprehensive review. Plants (Basel). 2021;10(4):729. doi: 10.3390/plants10040729, PMID 33918026.
Frimpong EK, Thembane N, Hlatshwayo S, Ngcobo M, Gqaleni N. Indigenous medicinal plants used in the management of diabetes in Africa: 5 y (2019-2024) in perspective. Plants (Basel). 2024;13(14):1898. doi: 10.3390/plants13141898, PMID 39065425.
Ekor M. The growing use of herbal medicines: issues relating to adverse reactions and challenges in monitoring safety. Front Pharmacol. 2014;4:177. doi: 10.3389/fphar.2013.00177, PMID 24454289.
Izzo AA, Ernst E. Interactions between herbal medicines and prescribed drugs: an updated systematic review. Drugs. 2009;69(13):1777-98. doi: 10.2165/11317010-000000000-00000, PMID 19719333.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. doi: 10.1136/bmj.n71, PMID 33782057.
Abo KA, Fred-Jaiyesimi AA, Jaiyesimi AE. Ethnobotanical studies of medicinal plants used in the management of diabetes mellitus in South Western Nigeria. J Ethnopharmacol. 2008;115(1):67-71. doi: 10.1016/j.jep.2007.09.005, PMID 17950547.
Asase A, Yohonu DT. Ethnobotanical study of herbal medicines for management of diabetes mellitus in dangme west district of southern Ghana. J Herb Med. 2016;6(4):204-9. doi: 10.1016/j.hermed.2016.07.002.
Asafo-Agyei T, Appau Y, Barimah KB, Asase A. Medicinal plants used for management of diabetes and hypertension in Ghana. Heliyon. 2023;9(12):e22977. doi: 10.1016/j.heliyon.2023.e22977, PMID 38076168.
Karou SD, Tchacondo T, Djikpo Tchibozo MA, Abdoul Rahaman S, Anani K, Koudouvo K. Ethnobotanical study of medicinal plants used in the management of diabetes mellitus and hypertension in the central region of Togo. Pharm Biol. 2011;49(12):1286-97. doi: 10.3109/13880209.2011.621959, PMID 22077164.
Holaly GE, Simplice KD, Gnoula C, Agbodeka K, Anani K, Tchacondo T, Agbonon A, Batawila K, Simpore J. Etude ethnobotanique des plantes utilisees dans le traitement du diabete dans la medecine traditionnelle de la region Maritime du Togo. Pan Afr Med J. 2015;20:437. doi: 10.11604/pamj.2015.20.437.5660, PMID 26309469.
Lawin IF, Laleye FO, Agbani OP, Assogbadjo AE. Ethnobotanical assessment of the plant species used in the treatment of diabetes in the Sudano-Guinean zone of Benin. J Anim Plant Sci. 2015;26(3):4108-23.
Laleye FO, Mensah S, Assogbadjo AE, Ahissou H. Diversity knowledge and use of plants in traditional treatment of diabetes in the Republic of Benin. Ethnobot Res App. 2015;14:231-57. doi: 10.17348/era.14.0.231-257.
Diomande R, Konkon NG, Kpan WB, Abeda ZH. Ethnobotanical study of medicinal plants sold by women in Adjame markets, southern Cote d’Ivoire. Int J Appl Res. 2022;8(2):389-98.
N’Guessan K, Kouassi K, Kouadio K, Ake-Assi L. Ethnobotanical study of plants used to treat diabetes in traditional medicine by Abbey and Krobou peoples of Agboville (Cote d’Ivoire). Am J Sci Res. 2009;4:45-58.
Dieye AM, Sarr A, Diop SN, Ndiaye M, Sy GY, Diarra M. Medicinal plants and the treatment of diabetes in Senegal: survey with patients. Fundam Clin Pharmacol. 2008;22(2):211-6. doi: 10.1111/j.1472-8206.2007.00563.x, PMID 18205810.
Diallo A, Traore MS, Keita SM, Balde MA, Keita A, Camara M. Management of diabetes in Guinean traditional medicine: an ethnobotanical investigation in the coastal lowlands. J Ethnopharmacol. 2012;144(2):353-61. doi: 10.1016/j.jep.2012.09.020, PMID 23006605.
Adepoju AO, Amusa MO, Samson AO, Paul-Chima UO. Ethnopharmacological survey on medicinal plants utilization in Freetown, Sierra Leone. RPS Pharm Pharmacol Rep. 2023;2(2):rqad019. doi: 10.1093/rpsppr/rqad019.